Glucocorticoid sensor
Here are the parts sent to the registry:
Name table
| Part Name
| Registry Number
|
| E1
| BBa_K1144001
|
| E2
| BBa_K1144002
|
| E3
| BBa_K1144003
|
| E4
| BBa_K1144004
|
| E1GF
| BBa_K1144005
|
| E2GF
| BBa_K1144006
|
| E3GF
| BBa_K1144007
|
| E4GF
| BBa_K1144008
|
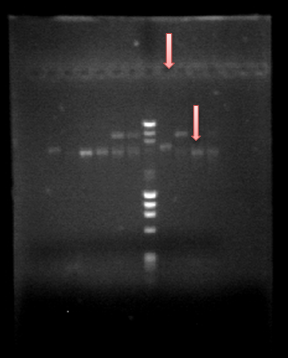
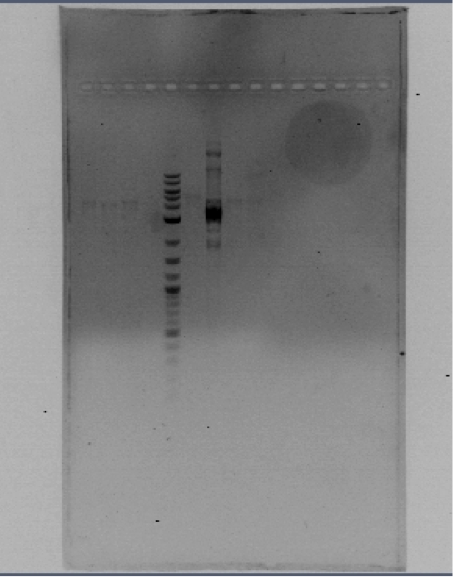
You can see the confirmation gels of the parts below:
PCR confirmation: E1GF-E1GF(2)-E2GF-E2GF(2)-WM-E3GF-E3GF(2)-E4GF-E4GF(2) 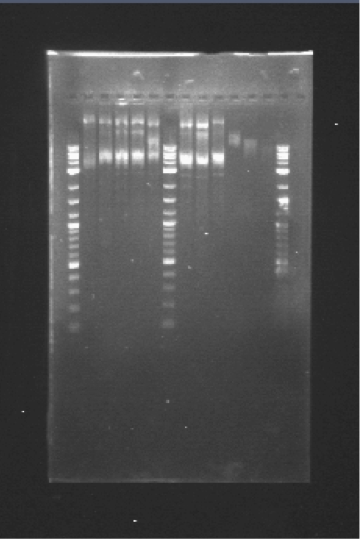
Co-transformation: WM-E1GF2-E2GF-E3GF1-E3GF2-E4GF-WM- E3GF3-E4GF2-E2GF2 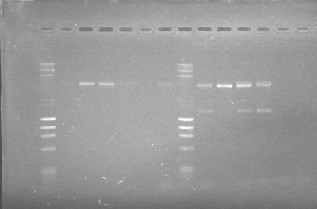
PCR confirmation of the parts cloned in the pSB1C3 backbone using the specifically designed primers 15 and 31 (see primers)
Characterization of reporters
To assess the strength of our promoters when induced with Dexamethasone, we performed a fluorometric assay using mCherry as our reporter.
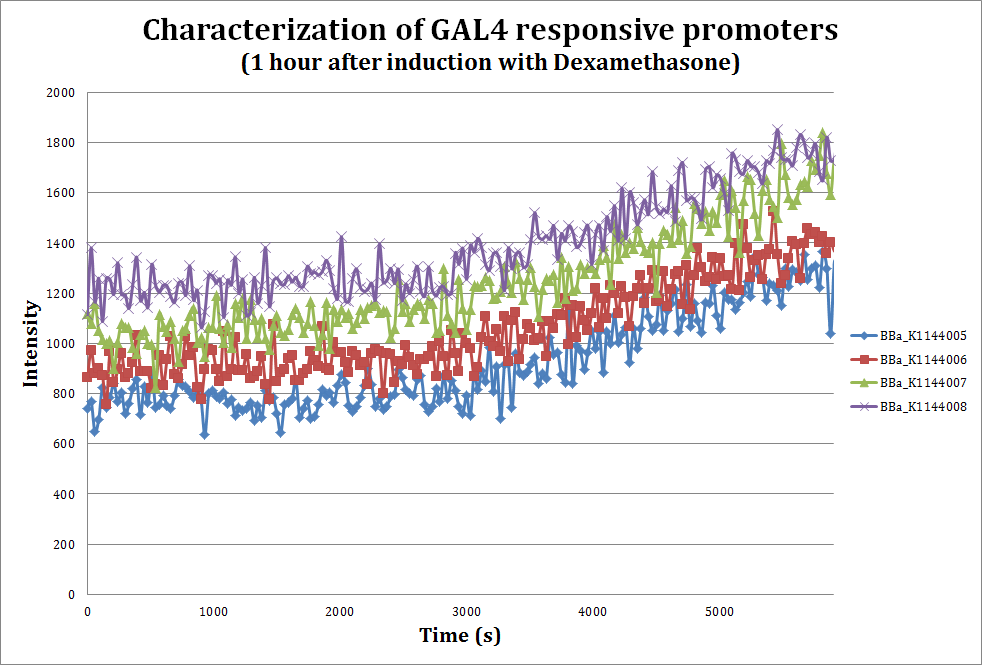
Unsaturated curve where we see how the mCherry reporter appears after the addition of 10 uM dexamethasone, a glucocorticoid. Emmision intensity was measured at 607nm after excitation at 586nm
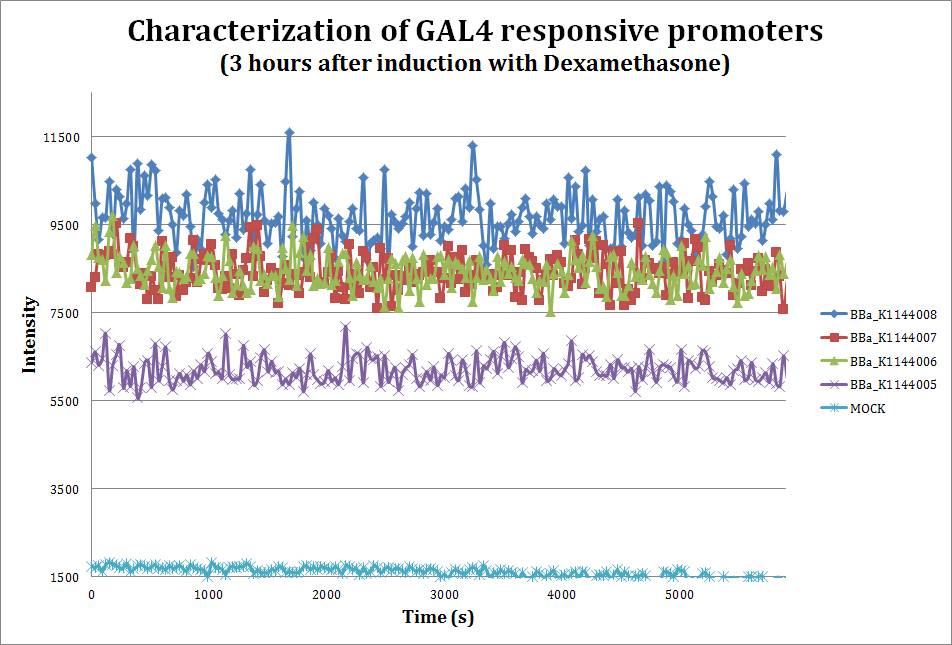
Saturated curve for fluorescence. After three hours of the addition of 10 uM dexamethasone the reporter (mCherry) reaches its highest signal. We can see the different strengths depending on the number of UAS boxes. Emmision intensity was measured at 607nm after excitation at 586nm
We also decided to visually inspect our induced transformants. Here two of the images taken using a epifluorescent microscopy with a TRITC filter.
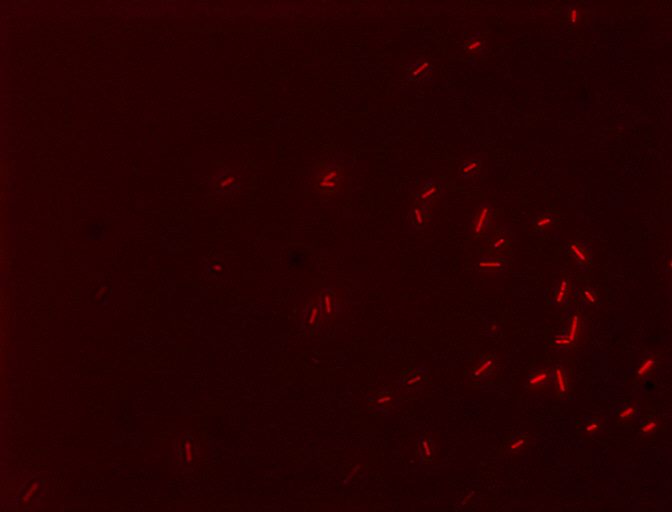
Cells with the E2GF part (BBa_K1144006) showing their fluorescent reporter, mCherry, after induction with 10 uM dexamethasone! The filter used was TRITC 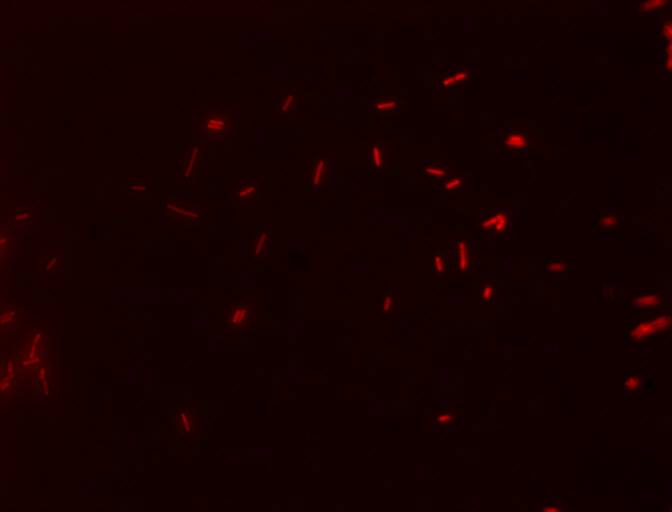
Cells with the E4GF part (BBa_K1144008) showing their fluorescent reporter, mCherry, after induction with 10 uM dexamethasone! The filter used was TRITC 2
 "
"