Team:Goettingen/Team/Reporter
From 2013.igem.org
(→Riboswitch reporter system (part BBa_K1045002):) |
(→DarR reporter system (part BBa_K1045017):) |
||
| Line 53: | Line 53: | ||
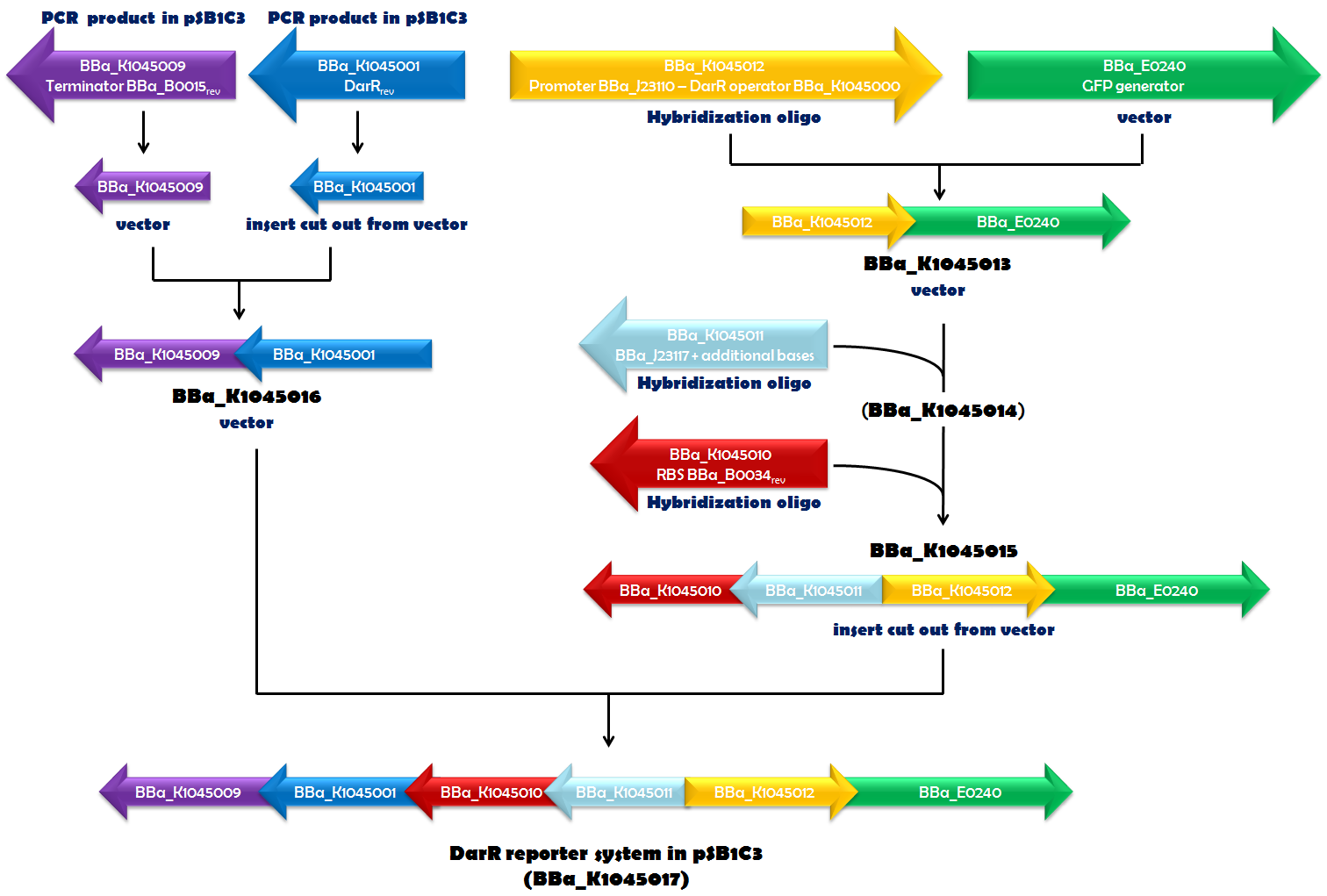
Recently, in ''Mycobacterium smegmatis'', a transcriptional repressor (DarR) was identified. It can bind to a specific DNA sequence, called the DarR operator. The binding efficiency of DarR is strongly enhanced by c-di-AMP. Since DarR is a c-di-AMP sensor, we intended to use it for our reporter system. We cloned DarR into pSB1C3 ([http://parts.igem.org/Part:BBa_K1045001 BBa_K1045001]) and also constructed the operator as a Biobrick ([http://parts.igem.org/Part:BBa_K1045000 BBa_K1045000]). We placed the DarR operator between a strong promoter ([http://parts.igem.org/Part:BBa_J23110 BBa_J23110]) and the GFP generator [http://parts.igem.org/Part:BBa_E0240 BBa_E0240]. In the very same plasmid but oriented in the opposite direction, we assembled a DarR expression unit. DarR expression was driven by a weak promoter based on [http://parts.igem.org/Part:BBa_J23117 BBa_J23117] and terminated by [http://parts.igem.org/Part:BBa_K1045009 BBa_K1045009]. This terminator is based on [http://parts.igem.org/Part:BBa_B0015 BBa_B0015], which is part of [http://parts.igem.org/Part:BBa_E0240 BBa_E0240], as well, and should ensure that the transcription of DarR and GFP did not influence each other. The RBS [http://parts.igem.org/Part:BBa_K1045010 BBa_K1045010] (derived from [http://parts.igem.org/Part:BBa_B0034 BBa_B0034]) was used as an RBS for DarR mRNA translation. Assembly of all those parts (Fig. 1.1) required a complex cloning process which finally resulted in part [http://parts.igem.org/Part:BBa_K1045017 BBa_K1045017], the DarR reporter system (Fig. 1.2). E. coli was then transformed with this construct to obtain the in vivo screening system for antibiotics directed against c-di-AMP. | Recently, in ''Mycobacterium smegmatis'', a transcriptional repressor (DarR) was identified. It can bind to a specific DNA sequence, called the DarR operator. The binding efficiency of DarR is strongly enhanced by c-di-AMP. Since DarR is a c-di-AMP sensor, we intended to use it for our reporter system. We cloned DarR into pSB1C3 ([http://parts.igem.org/Part:BBa_K1045001 BBa_K1045001]) and also constructed the operator as a Biobrick ([http://parts.igem.org/Part:BBa_K1045000 BBa_K1045000]). We placed the DarR operator between a strong promoter ([http://parts.igem.org/Part:BBa_J23110 BBa_J23110]) and the GFP generator [http://parts.igem.org/Part:BBa_E0240 BBa_E0240]. In the very same plasmid but oriented in the opposite direction, we assembled a DarR expression unit. DarR expression was driven by a weak promoter based on [http://parts.igem.org/Part:BBa_J23117 BBa_J23117] and terminated by [http://parts.igem.org/Part:BBa_K1045009 BBa_K1045009]. This terminator is based on [http://parts.igem.org/Part:BBa_B0015 BBa_B0015], which is part of [http://parts.igem.org/Part:BBa_E0240 BBa_E0240], as well, and should ensure that the transcription of DarR and GFP did not influence each other. The RBS [http://parts.igem.org/Part:BBa_K1045010 BBa_K1045010] (derived from [http://parts.igem.org/Part:BBa_B0034 BBa_B0034]) was used as an RBS for DarR mRNA translation. Assembly of all those parts (Fig. 1.1) required a complex cloning process which finally resulted in part [http://parts.igem.org/Part:BBa_K1045017 BBa_K1045017], the DarR reporter system (Fig. 1.2). E. coli was then transformed with this construct to obtain the in vivo screening system for antibiotics directed against c-di-AMP. | ||
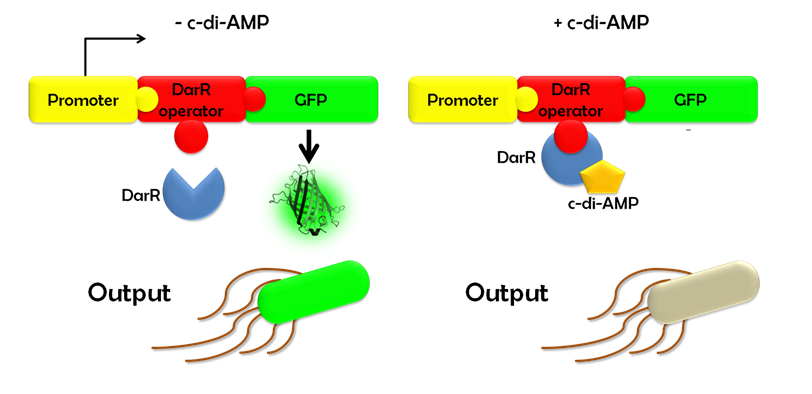
| - | Ideally, this c-di-AMP-sensing system works in the following way (Fig. 1.3): Without c-di-AMP, DarR is not bound to the DarR operator, so GFP is expressed resulting in green fluorescing cells. With c-di-AMP, DarR can bind to its binding sequence, repressing gfp transcription, leading to non-fluorescent cells. In the same way, the system might react to compounds similar to c-di-AMP. It can therefore be used to screen for compounds similar to c-di-AMP. It can be used in a large throughput scale, as well. This way, the DarR reporter system will make it possible to find inhibitors and competitors which might be used as antibiotics. | + | Ideally, this c-di-AMP-sensing system works in the following way (Fig. 1.3): Without c-di-AMP, DarR is not bound to the DarR operator, so GFP is expressed resulting in green fluorescing cells. With c-di-AMP, DarR can bind to its binding sequence, repressing ''gfp'' transcription, leading to non-fluorescent cells. In the same way, the system might react to compounds similar to c-di-AMP. It can therefore be used to screen for compounds similar to c-di-AMP. It can be used in a large throughput scale, as well. This way, the DarR reporter system will make it possible to find inhibitors and competitors which might be used as antibiotics. |
<html><a href="https://static.igem.org/mediawiki/2013/e/ea/Goe-rt-cloningSteps.png" target="_blank"><img src="https://static.igem.org/mediawiki/2013/e/ea/Goe-rt-cloningSteps.png" width="750"/></a></html> | <html><a href="https://static.igem.org/mediawiki/2013/e/ea/Goe-rt-cloningSteps.png" target="_blank"><img src="https://static.igem.org/mediawiki/2013/e/ea/Goe-rt-cloningSteps.png" width="750"/></a></html> | ||
| Line 75: | Line 75: | ||
</html> | </html> | ||
<br /><br /> | <br /><br /> | ||
| + | |||
==Riboswitch reporter system ([http://parts.igem.org/Part:BBa_K1045002 part BBa_K1045002]):== | ==Riboswitch reporter system ([http://parts.igem.org/Part:BBa_K1045002 part BBa_K1045002]):== | ||
The ''ydaO'' gene from'' B. subtilis'' is regulated by c-di-AMP. This control is mediated by a c-di-AMP-binding riboswitch, as we knew from a congress. Structurally, a riboswitch is a RNA-sequence. Functionally, it is a simple switch. It has two mutually exclusive states: ON and OFF. Depending on the circumstances, it can only be in one of the states. The ''ydaO''-riboswitch is dependent on c-di-AMP; whenever it is present, the riboswitch will be turned OFF, meaning that the gene controlled by it cannot be read. | The ''ydaO'' gene from'' B. subtilis'' is regulated by c-di-AMP. This control is mediated by a c-di-AMP-binding riboswitch, as we knew from a congress. Structurally, a riboswitch is a RNA-sequence. Functionally, it is a simple switch. It has two mutually exclusive states: ON and OFF. Depending on the circumstances, it can only be in one of the states. The ''ydaO''-riboswitch is dependent on c-di-AMP; whenever it is present, the riboswitch will be turned OFF, meaning that the gene controlled by it cannot be read. | ||
Revision as of 09:40, 1 October 2013
 "
"