Team:Washington/GENERAL BASIC PROTOCOL
From 2013.igem.org
'
Feel free to add to this, but be brief. If you make changes, make a comment as to why.
Ask if before you do some lab task if you haven’t done it before.
Please clean up after yourself, and make the stations a little bit better each time use use them.
If everyone does this we can keep a functional and safe lab environment.
General lab information:
Most dangerous:
Fire, Autoclave (think hole in wall here), Chemicals, UV, microwaving media bottles
Be careful. Follow guidelines.
Lab Etiquette:
Put everything (especially common supplies, and all of the iGEM supplies are common) back where you found it.
Clean up after your self at the end of lab. Make sure someone wipe down the bench after you used it.
If you borrow dishware from the Baker Lab because we are out, make sure you replace it when finished and put our dishes in to be cleaned.
No gloves in outside of lab to prevent contamination of door handles. One glove is okay for carrying items.
Read the safety manual for the lab or take the online 15 minute EH&S safety training for more information.
Sterile Technique:
Use bunsen flame for sterilization, especially for media.
Pay attention to what contacts pipette tips (don’t touch to inside of bottle when aliquoting from 1L Bottles).
What To Keep Cold:
#1) ALL ENZYMES! Make sure these get back into the freezer. Leaving them out degrades them. Transport on ice or in freezer block.
#2) Antibiotics. Same as above. Fridge is OK for temporary storage (1 week - 1 month).
#3) Glycerol Stocks (possibly the most important, but usually the least problematic). High concentration of glycerol is toxic to cells and prolonged exposure to it kills them at RT, do not leave out.
Antibiotics:
Ap - Ampicillin
Cb - Carbenecillin (we use carb in this lab)
*Ampicillin and Carbenecillin can be used interchangeably
Km - Kanamycin
Cm - Chloramphenical (Chlor)
Sp - Spectinomycin
New aliquots are not needed every time unless degradation/contamination is suspected.
Shaker:
Place tubes on a slant.
Depress cap only to the first stop.
Gels:
Do not touch the running buffer while in operation, it can cause a shock.
Use pipette and containers labeled SYBR Safe and avoid contact with dye.
Keep gel area clean.
Seal the gel rig and wait for gel to cool to prevent leakage through mold pieces.
Transfer gel from casting box to box with electrodes (they look the same).
Add buffer to top of gel to make removal of comb easier.
Reuse TAE buffer a max 2-3 times or until foaming.
SYBR concentration is 10,000X. This means 5uls for 50mls of agarose.
30-45 seconds per 50mLs
Run at 100-150 volts. Lower for better resolution, higher for faster (but also can melt gel).
Use 3ul loading dye on parafilm + 5uls of sample.
Use 5ul of DNA ladder or 3ul to conserve stock.
Media:
Swirling under cool water can be used to speed cooling. Be careful not to cool to much, when bubbles cease to dart toward surface or you can hold the flask for a few seconds.
Do not fill more than half way.
Autoclave:
Get help with this! If you haven’t done this before it can be dangerous.
Sign up beforehand.
Use autoclave tape. And use foil to to prevent loose lid from popping off.
Do NOT screw lid on all the way. One method is to screw lid only one full turn.
Make sure door is firmly shut! (very important) Make sure it is closed completely, the hinges are loose and the left door can get cocked sideways. Push forcefully. Tighten “firmly,” enough so it is not easy to undo but it is not heroically tight. Be careful with the door. Wait a few minutes before leaving to be sure door is not leaking.
Read instructions posted and press buttons for programs.
Make sure pressure is 0 psi before opening! Open door slowly.
Use autoclavable tray (for more than one item)
Be careful when opening from steam. Steam burns.
Please label before to avoid mix ups. With contents, date and initials.
Bring hot mits with you. (Remember bring back to lab).
Push reset when finished.
Settings are different for left and right machines. Liquids (use the 45min cycle). Gravity to dry.
15 minutes to cool down.
Weighing:
Make sure to place used measuring utensils into dirty dish bin.
CLEAN UP after yourself or we will be nixed from using the chem room.
Postage Scale:
Add weighboat before zeroing.
Label and reuse weighboat if possible (store in iGEM lab).
UW iGEM Tips:
Use master mixes whenever possible to decrease pipetting and small volumes (especially for ligation)
DNA Ladder - 1kb NEB:
Dye is 6X so add 200ul of dye to 200ul of ladder plus 800uls of DI H2O.
OR, if you want a quantitative ladder add 166uls dye to 200uls ladder plus 634uls DI H2O.
PCR:
In general when running a PCR to leave a PCR tube rack in front of the machine with a label for whomever pulls the tubes out (or they can easily get lost). If you put a pcr in, pull them out the next day so the machine doesn't keep running.
Don’t use Tm printed on tube, use Tm of annealing region.
Run multiple PCR reactions simultaneously w/ different parameters if you are unsure about the efficiency of certain reagents
Labeling Gels:
Here are a few examples:
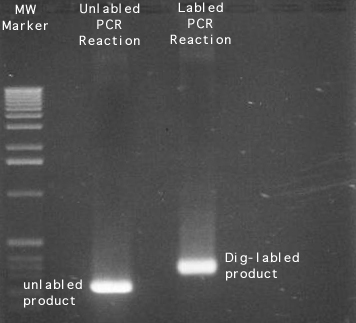
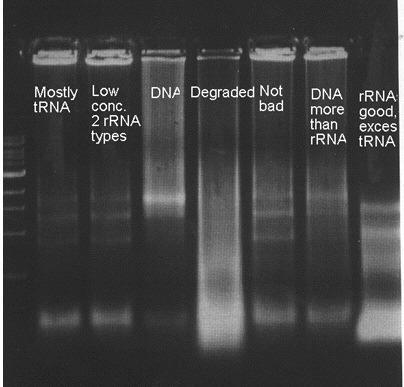
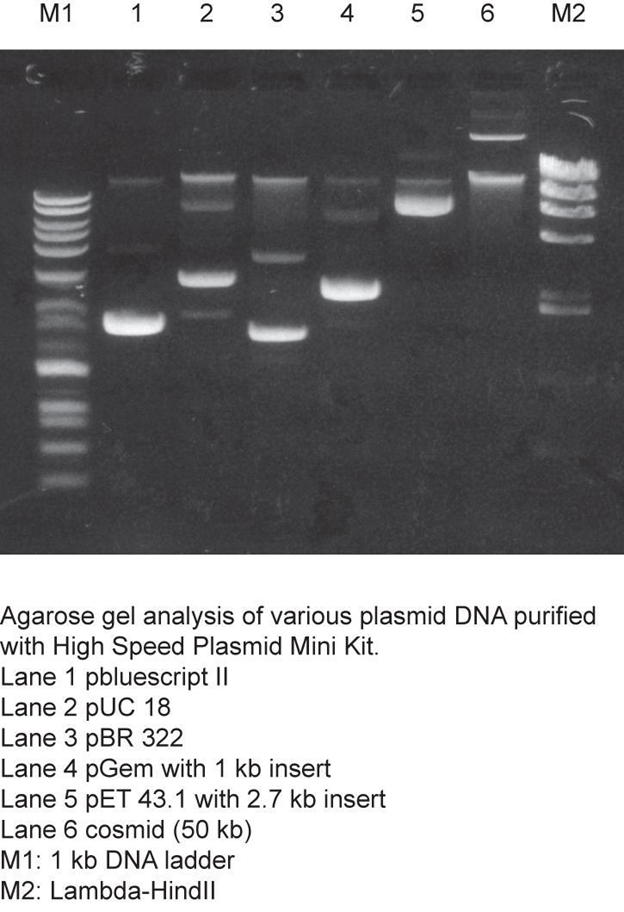
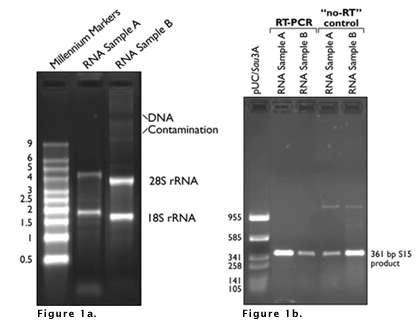
Making Solutions:
Introduction to lab Techniques - Faculty web pages
Digestion:
Use HF enzymes and XbaI all compatible in CutSmart. Digest overnight for further cutting.
Streak plate:
Use much less cells than expected.
Ligation:
Use ligation buffer aliquots in PCR tubes.
Heat inactivation increases transformation efficiency by around 10 fold.
Parafilm:
Cut across width and stretch. For several plates press against benchtop to hold one side and wrap around.
Screening:
Use primers specific to insert on multiple samples to test if any are insert.
Gel:
20ng of DNA is visible on a gel with ETBr
40-50ng of DNA is visible on a gel with SYBR
Sterilize/ Dispose of Cell Cultures:
10% Bleach for 30 minutes. down Drain (REMOVE AGAR)
Centrifuge:
Make sure samples are balanced with H20 if necessary.
Make sure the lid is on securely.
Wait for the centrifuge to reach full speed without loud sounds (and press stop if you hear any) before leaving.
Miniprep and PCR Purification:
Can run sample through column twice to increase binding.
Eluting in lower volume increases DNA concentration.
Eluting 2 times with half the total elution volume increases elution yield (15uls 2x is great)
 "
"


