Template:Kyoto/Notebook/Sep 27
From 2013.igem.org
(Difference between revisions)
(→Gel Extraction) |
(→Sep 27) |
||
| Line 312: | Line 312: | ||
|} | |} | ||
</div> | </div> | ||
| + | |||
| + | |||
| + | ===Plating=== | ||
| + | <div class="experiment"> | ||
| + | <span class="author">Hirano</span> | ||
| + | </div> | ||
| + | {| class="wikitable" | ||
| + | !Sample||Use plate | ||
| + | |- | ||
| + | |9/26 GGA2 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA3 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA4||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA5 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA6 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA7 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA11 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA312||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA13 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA14||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA15 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA20||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA23 ||LB(CP) | ||
| + | |- | ||
| + | |9/26 GGA24||LB(CP) | ||
| + | |- | ||
| + | |9/26 Ptet+RBS-GFP-DT ||LB(CP) | ||
| + | |} | ||
| + | </div> | ||
| + | incubate 37°C | ||
| + | |||
| + | ===Restriction Enzyme Digestion=== | ||
| + | <div class="experiment"> | ||
| + | <span class="author">Kojima</span> | ||
| + | {| class="wikitable" | ||
| + | !||9/24 pSB1C3||EcoRI||SpeI||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||11µL||1µL||1µL||3µL||14µL||30µL | ||
| + | |- | ||
| + | |NC||0.5µL||0µL||0µL||1µL||8.5µL||10µL | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | !||9/24 Pcon-pT181attenuator-aptamer12-1R-DT||EcoRI||SpeI||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||8.7µL||1µL||1µL||3µL||16.3µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||0µL||1µL||8.6µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/24 pT181attenuator||EcoRI||SpeI||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||3.1µL||1µL||1µL||3µL||21.9µL||30µL | ||
| + | |- | ||
| + | |NC||0.2µL||0µL||0µL||1µL||8.8µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||8/17 RBS-GFP-DT||EcoRI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |1cut||16.4µL||1µL||3µL||3µL||6.6µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||1µL||1µL||7.6µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/17 pSB4K5||EcoRI||PstI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||8.5µL||1µL||1µL||3µL||3µL||13.5µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||0µL||1µL||1µL||7.6µL||10µL | ||
| + | |} | ||
| + | |||
| + | {| class="wikitable" | ||
| + | !||9/14 pSB4K5||EcoRI||PstI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||7.3µL||1µL||1µL||3µL||3µL||14.7µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||0µL||1µL||1µL||7.6µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/22 Pcon-pT181attenuator-DT||EcoRI||SpeI ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||5.4µL||1µL||1µL||3µL||19.6µL||30µL | ||
| + | |- | ||
| + | |NC||0.3µL||0µL||0µL||1µL||8.7µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||8/7 RBS-GFP-DT 1||XbaI||PstI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||8.1µL||1µL||1µL||3µL||3µL||13.9µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||0µL||1µL||1µL||7.6µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/26 RBS-GFP-DT 1||XbaI ||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |1cut||8.1µL||1µL||3µL||3µL||14.9µL||30µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/16 Pλ-luxI||EcoRI||PstI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||14.7µL||1µL||1µL||3µL||3µL||7.3µL||30µL | ||
| + | |- | ||
| + | |NC||0.7µL||0µL||0µL||1µL||1µL||7.3µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/21 Ptet||EcoRI||SpeI ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||14.2µL||1µL||1µL||3µL||10.8µL||30µL | ||
| + | |- | ||
| + | |NC||0.7µL||0µL||0µL||1µL||8.3µL||10µL | ||
| + | |} | ||
| + | {| class="wikitable" | ||
| + | !||9/24 Pcon-pT181attenuator-aptamer12-1R-DT||XbaI||PstI||BSA ||buffer||MilliQ||total | ||
| + | |- | ||
| + | |2cuts||8.7µL||1µL||1µL||3µL||3µL||13.3µL||30µL | ||
| + | |- | ||
| + | |NC||0.4µL||0µL||0µL||1µL||1µL||7.6µL||10µL | ||
| + | |} | ||
| + | |||
| + | </div> | ||
| + | |||
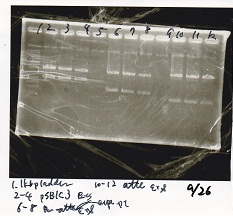
| + | ===Electrophoresis=== | ||
| + | <div class="experiment"> | ||
| + | <span class="author">Kojima</span> | ||
| + | {| class="wikitable" | ||
| + | !Lane||Sample|| Enzyme1|| Enzyme2 | ||
| + | |- | ||
| + | |1||pSB1C3||EcoRI||SpeI | ||
| + | |- | ||
| + | |2||pSB1C3||--||-- | ||
| + | |- | ||
| + | |3||Pcon-pT181attenuator-aptamer12-1R-DT||EcoRI||SpeI | ||
| + | |- | ||
| + | |4||Pcon-pT181attenuator-aptamer12-1R-DT||--||-- | ||
| + | |- | ||
| + | |5||1kb ladder||--||-- | ||
| + | |- | ||
| + | |6||pT181attenuator||EcoRI||SpeI | ||
| + | |- | ||
| + | |7||pT181attenuator||--||-- | ||
| + | |- | ||
| + | |8||RBS-GFP-DT||EcoRI||-- | ||
| + | |- | ||
| + | |9||RBS-GFP-DT||--||-- | ||
| + | |} | ||
| + | [[File:IgkuxxxxBa 1.jpg]]<br> | ||
Revision as of 07:08, 27 September 2013
Contents |
Sep 27
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 2 | pSB1C3 | EcoRI+SpeI |
| 3 | ||
| 4 | ||
| 6 | Pcon-attenuator-aptmer-DT | EcoRI+SpeI |
| 7 | ||
| 8 | ||
| 10 | Pcon-attenuator-aptmer-DT | EcoRI+SpeI |
| 11 | ||
| 12 |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| Pcon-attenuator-DT(EcoRI+SpeI) | 6.8 | 1.98 | 0.43 |
| pT181-attenuator(EcoRI+SpeI) | 7.5 | 1.84 | 0.06 |
| pSB1C3(EcoRI+SpeI) | 17.7 | 1.97 | 0.71 |
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 3 | pSB4K5 | EcoRI+PstI |
| 4 | pSB4K5 | EcoRI+PstI |
| 5 | pSB4K5 | EcoRI+PstI |
| 7 | pSB6A1 | EcoRI+PstI |
| 8 | pSB6A1 | EcoRI+PstI |
| 9 | pSB6A1 | EcoRI+PstI |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| (´ω`) | |||
| (ΦωΦ^) |
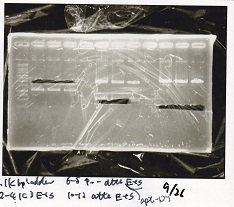
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | pSB4K5 | EcoRI | PstI |
| 2 | pSB4K5 | - | - |
| 3 | 1kbp ladder | - | - |
| 4 | Pcon-attenuator | EcoRI | SpeI |
| 5 | Pcon-attenuator | - | - |
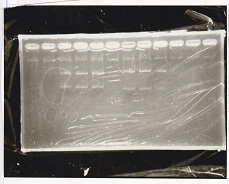
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | RBS-GFP-DT | XbaI | PstI |
| 2 | RBS-GFP-DT | - | - |
| 3 | RBS-GFP-DT | EcoRI | XbaI |
| 4 | 1kbp ladder | - | - |
| 5 | Ptet | EcoRI | SpeI |
| 6 | Ptet | - | - |
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 100bp ladder | -- |
| 3 | Ptet | EcoRI+SpeI |
| 4 | Ptet | EcoRI+SpeI |
| 5 | Ptet | EcoRI+SpeI |
File:Igku xxbeforexx.xxx
File:Igku xxafterxx.xxx
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 3 | Pcon-antisense-aptamer-DT | XbaI+PstI |
| 4 | Pcon-antisense-aptamer-DT | XbaI+PstI |
| 5 | Pcon-antisense-aptamer-DT | XbaI+PstI |
File:Igku xxbeforexx.xxx
File:Igku xxafterxx.xxx
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 3 | RBS-GFP-DT | EcoRI+XbaI |
| 4 | RBS-GFP-DT | EcoRI+XbaI |
| 5 | RBS-GFP-DT | EcoRI+XbaI |
File:Igku xxbeforexx.xxx
File:Igku xxafterxx.xxx
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kb ladder | -- |
| 2 | Pcon-attenuator | EcoRI+SpeI |
| 3 | Pcon-attenuator | EcoRI+SpeI |
| 4 | Pcon-attenuator | EcoRI+SpeI |
| 6 | pSB4K5 | EcoRI+PstI |
| 7 | pSB4K5 | EcoRI+PstI |
| 8 | pSB4K5 | EcoRI+PstI |
| 10 | RBS-GFP-DT | XbaI+PstI |
| 11 | RBS-GFP-DT | XbaI+PstI |
| 12 | RBS-GFP-DT | XbaI+PstI |
File:Igku xxbeforexx.xxx
File:Igku xxafterxx.xxx
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| (´ω`) | |||
| (ΦωΦ^) |
Liquid Culture
| Sample | medium |
|---|---|
| Pcon-GFP | Amp |
| RBS-GFP-DT | CP |
| Pcon-attenuator | Amp |
| Pcon-attenuator-aptamer-DT | Amp |
| Pcon-tetR-DT | Amp |
Ligation
| state | Vector | Inserter | Ligation High ver.2 | ||
|---|---|---|---|---|---|
| experiment | 9/17 pSB1C3 (EcoRI+SpeI) 10.0ng/µL | 10µL | 9/26 pT181 attenuator(EcoRI+SpeI) 7.5ng/µL | 1.3µL | 3.5 µL |
| experiment | 9/25 DT (EcoRI+XbaI) 42.8ng/µL | 2.3µL | 9/22 Pcon-pT181 antisense(EcoRI+SpeI) 46.7ng/µL | 1.5µL | 1.9 µL |
| experiment | 9/27 pSB4K5(EcoRI+PstI) 12.9ng/µL | 7.8µL | 9/10 Pcon-attenuator(EcoRI+SpeI) 13.8ng/µL | 8.0µL | 3.5 µL |
| experiment | 9/27 pSB4K5(EcoRI+PstI) 12.9ng/µL | 7.8µL | 9/25 RBS-GFP-DT(XbaI+PstI) 13.9ng/µL | 12µL | 3.5 µL |
| experiment | 9/8 pSB4K5 (EcoRI+SpeI) 17.6ng/µL | xx µL | 8/21 Pcon-GFP-DT(EcoRI+SpeI) 11ng/µL | 12.6µL | xx µL |
Liquid Culture
| Sample | medium |
|---|---|
| Pcon-spinach-DT | plusgrow Amp |
| Pcon-tetRaptamer-DT | plusgrow Amp |
| Pcon-antisense-spinach-DT | plusgrow Amp |
| spinach-DT | plusgrow CP |
RNA Extraction
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| 9/26 Pcon-GFP-DT | 423.0 | 1.83 | 1.46 |
| 9/26 Pcon-GFP-DT K | 298.7 | 1.82 | 1.50 |
Electrophoresis
Miniprep
| DNA |
|---|
| Pcon-aptamer-DT |
| Pcon-spinach-DT |
| Pcon-antisense |
| Pcon-tetR-DT |
| Pcon-attenuator-DT |
| Pcon-antisense-spinach-DT |
Transformation
| Name1 | Name2 | Sample1(µL) | Sample2(µL) | Competent Cells(µL) | Total(µL) |
|---|---|---|---|---|---|
| Pcon-attenuator+RBS-GFP-DT(4K5) | Pcon-attenuator-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-attenuator+RBS-GFP-DT(4K5) | Pcon-antisense-spinach-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-attenuator+RBS-GFP-DT(4K5) | Pcon-tetR aptamer-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-RBS-GFP-DT(4K5) | Pcon-attenuator-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-RBS-GFP-DT(4K5) | Pcon-antisense-spinach-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-RBS-GFP-DT(4K5) | Pcon-tetR aptamer-DT(1A2) | 2 | 1 | 30 | 33 |
| Pcon-attenuator+RBS-GFP-DT(4K5) | -- | 2 | -- | 20 | 22 |
| Pcon-RBS-GFP-DT(4K5) | -- | 2 | -- | 20 | 22 |
| attenuator(1C3) | -- | 2 | -- | 20 | 22 |
Plating
| Sample | Use plate |
|---|---|
| 9/26 GGA2 | LB(CP) |
| 9/26 GGA3 | LB(CP) |
| 9/26 GGA4 | LB(CP) |
| 9/26 GGA5 | LB(CP) |
| 9/26 GGA6 | LB(CP) |
| 9/26 GGA7 | LB(CP) |
| 9/26 GGA11 | LB(CP) |
| 9/26 GGA312 | LB(CP) |
| 9/26 GGA13 | LB(CP) |
| 9/26 GGA14 | LB(CP) |
| 9/26 GGA15 | LB(CP) |
| 9/26 GGA20 | LB(CP) |
| 9/26 GGA23 | LB(CP) |
| 9/26 GGA24 | LB(CP) |
| 9/26 Ptet+RBS-GFP-DT | LB(CP) |
incubate 37°C
Restriction Enzyme Digestion
| 9/24 pSB1C3 | EcoRI | SpeI | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 2cuts | 11µL | 1µL | 1µL | 3µL | 14µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 1µL | 8.5µL | 10µL |
| 9/24 Pcon-pT181attenuator-aptamer12-1R-DT | EcoRI | SpeI | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 2cuts | 8.7µL | 1µL | 1µL | 3µL | 16.3µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 8.6µL | 10µL |
| 9/24 pT181attenuator | EcoRI | SpeI | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 2cuts | 3.1µL | 1µL | 1µL | 3µL | 21.9µL | 30µL |
| NC | 0.2µL | 0µL | 0µL | 1µL | 8.8µL | 10µL |
| 8/17 RBS-GFP-DT | EcoRI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 1cut | 16.4µL | 1µL | 3µL | 3µL | 6.6µL | 30µL |
| NC | 0.4µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
| 9/17 pSB4K5 | EcoRI | PstI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 8.5µL | 1µL | 1µL | 3µL | 3µL | 13.5µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
| 9/14 pSB4K5 | EcoRI | PstI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 7.3µL | 1µL | 1µL | 3µL | 3µL | 14.7µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
| 9/22 Pcon-pT181attenuator-DT | EcoRI | SpeI | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 2cuts | 5.4µL | 1µL | 1µL | 3µL | 19.6µL | 30µL |
| NC | 0.3µL | 0µL | 0µL | 1µL | 8.7µL | 10µL |
| 8/7 RBS-GFP-DT 1 | XbaI | PstI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 8.1µL | 1µL | 1µL | 3µL | 3µL | 13.9µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
| 9/26 RBS-GFP-DT 1 | XbaI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 1cut | 8.1µL | 1µL | 3µL | 3µL | 14.9µL | 30µL |
| 9/16 Pλ-luxI | EcoRI | PstI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 14.7µL | 1µL | 1µL | 3µL | 3µL | 7.3µL | 30µL |
| NC | 0.7µL | 0µL | 0µL | 1µL | 1µL | 7.3µL | 10µL |
| 9/21 Ptet | EcoRI | SpeI | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|
| 2cuts | 14.2µL | 1µL | 1µL | 3µL | 10.8µL | 30µL |
| NC | 0.7µL | 0µL | 0µL | 1µL | 8.3µL | 10µL |
| 9/24 Pcon-pT181attenuator-aptamer12-1R-DT | XbaI | PstI | BSA | buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 8.7µL | 1µL | 1µL | 3µL | 3µL | 13.3µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
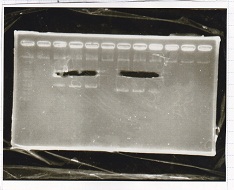
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | pSB1C3 | EcoRI | SpeI |
| 2 | pSB1C3 | -- | -- |
| 3 | Pcon-pT181attenuator-aptamer12-1R-DT | EcoRI | SpeI |
| 4 | Pcon-pT181attenuator-aptamer12-1R-DT | -- | -- |
| 5 | 1kb ladder | -- | -- |
| 6 | pT181attenuator | EcoRI | SpeI |
| 7 | pT181attenuator | -- | -- |
| 8 | RBS-GFP-DT | EcoRI | -- |
| 9 | RBS-GFP-DT | -- | -- |
 "
"