Team:Heidelberg/Templates/Del week13 OP
From 2013.igem.org
(Difference between revisions)
(→Restriction digest of fragment FS_22 to FS_13s; 2.7 kb; 21-07-2013 with EcoRI-HF) |
(→Restriction digest from FS_22 to FS_13; 2.7 kb; 25-07-2013 with EcoRI-HF) |
||
| (One intermediate revision not shown) | |||
| Line 131: | Line 131: | ||
==30-07-2013== | ==30-07-2013== | ||
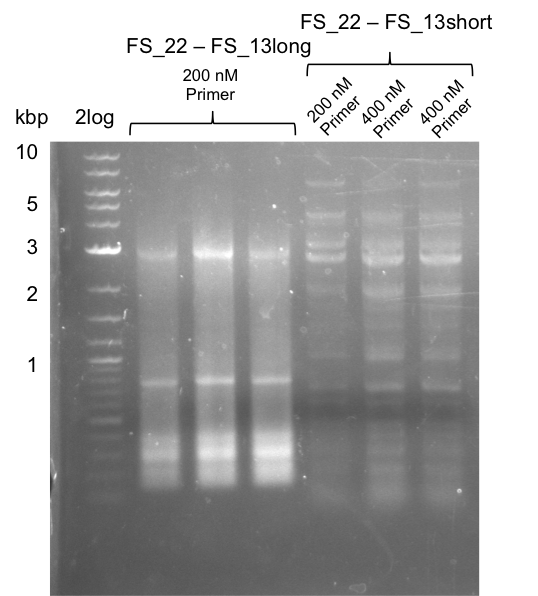
| - | ===Re-PCR from FS_22 to FS_13; 2.7 kb; | + | ===Re-PCR from FS_22 to FS_13; 2.7 kb; 19-07-2013=== |
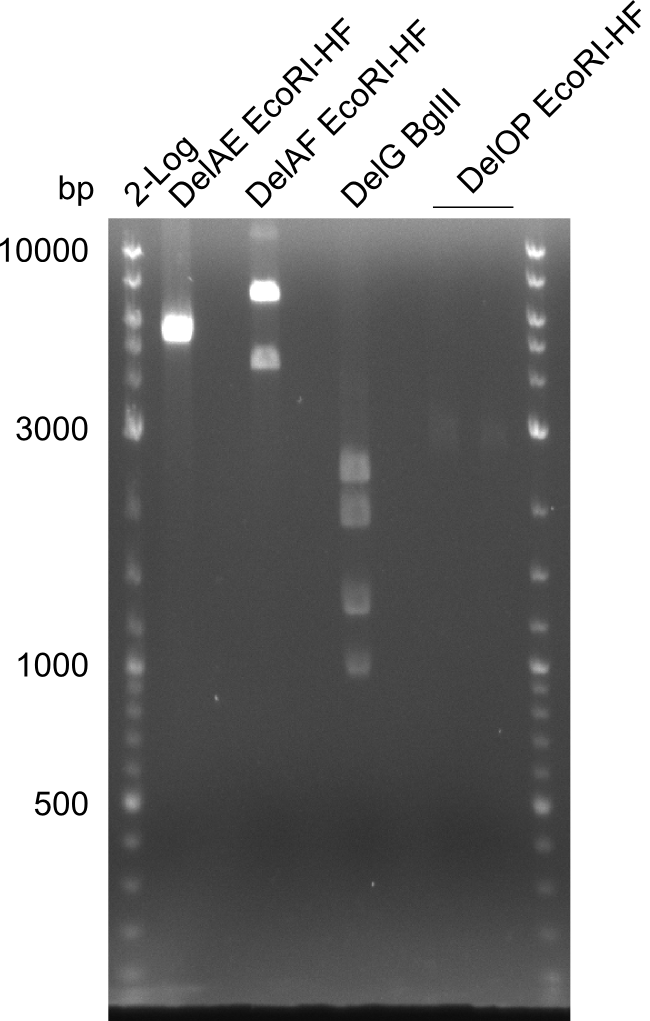
[[File:Heidelberg_20130730 3x 22-13long opecoR1 log2 Tyr 3 13 15besch.png|150px|thumb|Re-PCR OP, Restriction digest OP with EcoRI; run at 100 V, 0.8 % gel (TAE)]] | [[File:Heidelberg_20130730 3x 22-13long opecoR1 log2 Tyr 3 13 15besch.png|150px|thumb|Re-PCR OP, Restriction digest OP with EcoRI; run at 100 V, 0.8 % gel (TAE)]] | ||
| Line 141: | Line 141: | ||
! what !! µl | ! what !! µl | ||
|- | |- | ||
| - | | Fragment FS_22 to FS_13_short ( | + | | Fragment FS_22 to FS_13_short (19-07-2013) || 1 |
|- | |- | ||
| FS_22: (1/10) || 2 | | FS_22: (1/10) || 2 | ||
| Line 182: | Line 182: | ||
* PCR will be repeated with different primer concentrations to estimate whether primer dimes might be the reason for insufficient amplification | * PCR will be repeated with different primer concentrations to estimate whether primer dimes might be the reason for insufficient amplification | ||
| - | ===Restriction digest from FS_22 to FS_13; 2.7 kb; | + | ===Restriction digest from FS_22 to FS_13; 2.7 kb; 25-07-2013 with EcoRI-HF=== |
| Line 190: | Line 190: | ||
! what !! µl | ! what !! µl | ||
|- | |- | ||
| - | | FS_22 to FS_13long( | + | | FS_22 to FS_13long(25-07-2013) || 17 |
|- | |- | ||
| EcoRI || 1 | | EcoRI || 1 | ||
Latest revision as of 18:32, 3 October 2013
Contents |
25-07-2013
Re-PCR I from FS_22 to FS_13; 2.7 kb; 19-07-2013
3x20µl
- Reaction
| what | µl |
|---|---|
| Fragment FS_22 to FS_13_short (19-07-2013) | 1 |
| FS_22: (1/10) | 4 |
| FS_13_long: (1/10) | 4 |
| Phusion flash Master Mix | 10 |
| dd H2O | 1 |
- Conditions
| Biorad MyCycler* | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 12 | 98 | 1 |
| 65 ↓ 0.5 | 5 | |
| 72 | 1:00 | |
| 18 | 98 | 1 |
| 63 | 5 | |
| 72 | 1:00 | |
| 1 | 72 | 5min |
| 1 | 12 | inf |
Results:
- Amplification of DelOP led to a smear and several unexpected bands
- Re-PCR will be repeated
Re-PCR I from FS_22 to FS_13; 2.7 kb; 19-07-2013
3x20µl
- Reaction
| what | µl |
|---|---|
| Fragment FS_22 to FS_13_short (19-07-2013) | 1 |
| FS_22: (1/10) | 2 |
| FS_13_long: (1/10) | 2 |
| Phusion flash Master Mix | 10 |
| dd H2O | 5 |
- Conditions
| Biorad MyCycler* | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 12 | 98 | 1 |
| 65 ↓ 0.5 | 5 | |
| 72 | 1:00 | |
| 18 | 98 | 1 |
| 63 | 5 | |
| 72 | 1:00 | |
| 1 | 72 | 5min |
| 1 | 12 | inf |
Results:
- a smear as well as a band of the expeceted size occured
- bands were cut out and DNA purified using QIAquick Gel Extraction Kit
- DNA will be used for restriction digests to validate amplicon
26-07-2013
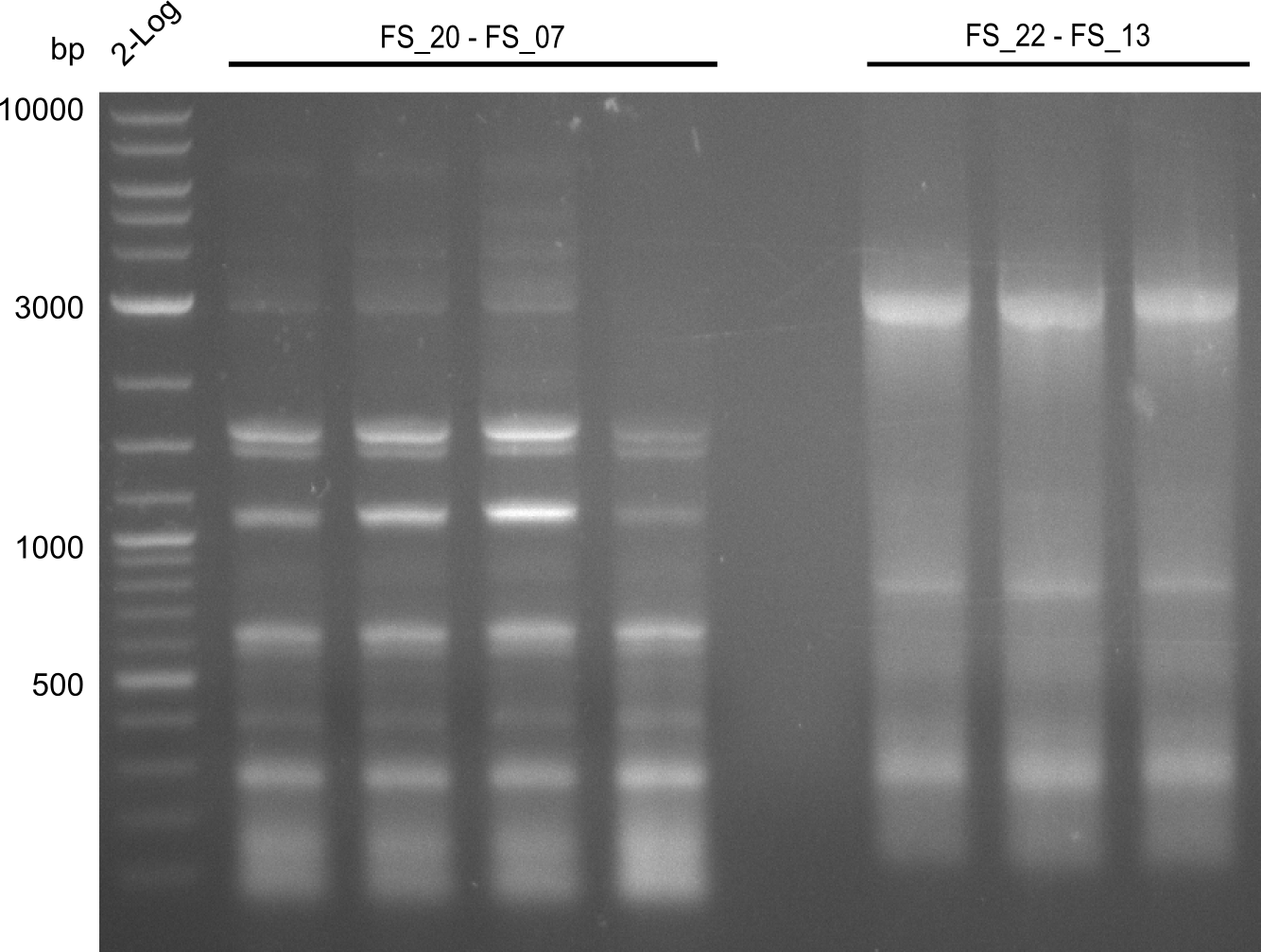
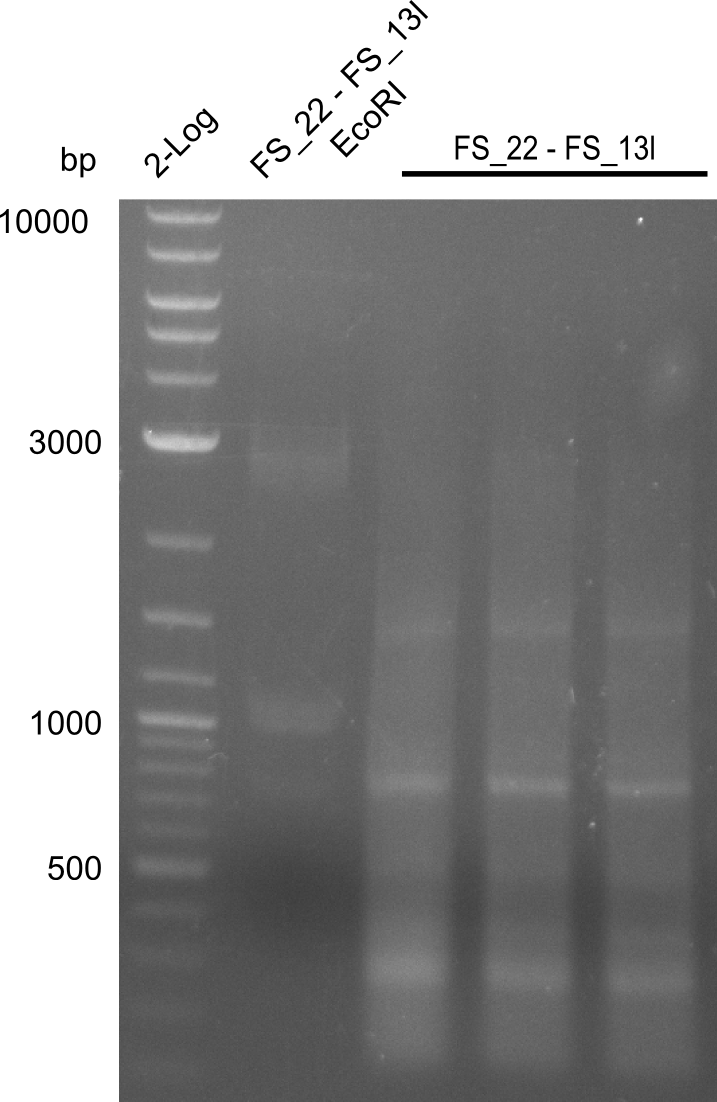
Restriction digest of fragment FS_22 to FS_13s; 2.7 kb; 21-07-2013 with EcoRI-HF
Incubation at 37°C for 1h 45min
| what | µl |
|---|---|
| FS_22 to FS_13short (21-07-2013) | 20 |
| EcorRI-HF | 0.8 |
| CutSmart | 2.5 |
| dd H2O | 2.5 |
| Expected fragment lengths [bp] | 1883, 960 |
Results:
- Restriction digest did not work, the amplified fragment was either not cut or the fragments are not visible due to a very small amount of DNA
- Re-PCR will be repeated to obtain the higher amounts of DNA required for the restriction digest
30-07-2013
Re-PCR from FS_22 to FS_13; 2.7 kb; 19-07-2013
3x20µl
- Reaction
| what | µl |
|---|---|
| Fragment FS_22 to FS_13_short (19-07-2013) | 1 |
| FS_22: (1/10) | 2 |
| FS_13_long: (1/10) | 2 |
| Phusion flash Master Mix | 10 |
| dd H2O | 5 |
- Conditions
| Biorad MyCycler* | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 12 | 98 | 1 |
| 65 ↓ 0.5 | 5 | |
| 72 | 1:00 | |
| 18 | 98 | 1 |
| 63 | 5 | |
| 72 | 1:00 | |
| 1 | 72 | 5min |
| 1 | 12 | inf |
Results:
- Amplification of DelOP did not work
- PCR will be repeated with different primer concentrations to estimate whether primer dimes might be the reason for insufficient amplification
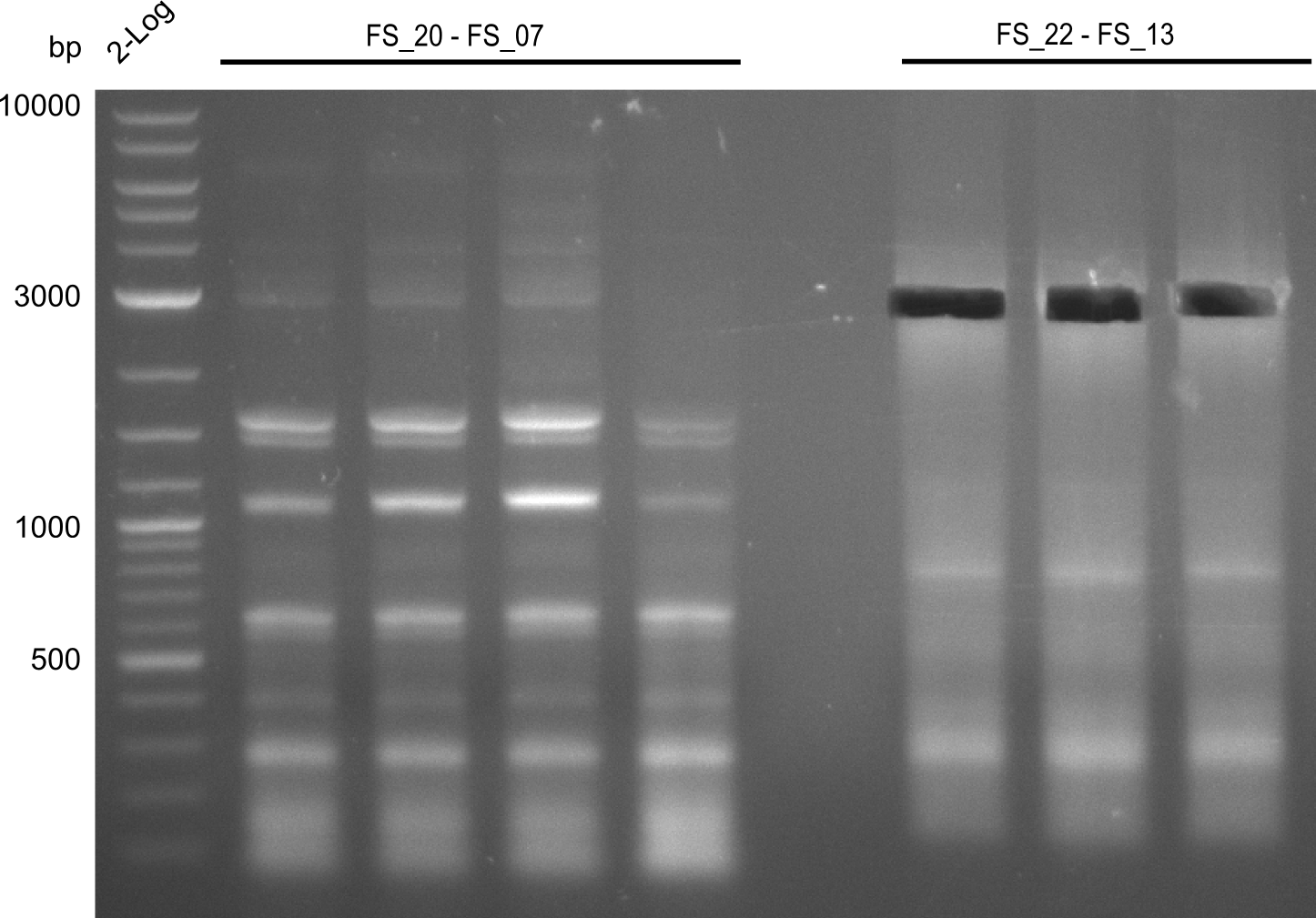
Restriction digest from FS_22 to FS_13; 2.7 kb; 25-07-2013 with EcoRI-HF
Incubation at 37°C for
| what | µl |
|---|---|
| FS_22 to FS_13long(25-07-2013) | 17 |
| EcoRI | 1 |
| Buffer CutSmart | 2 |
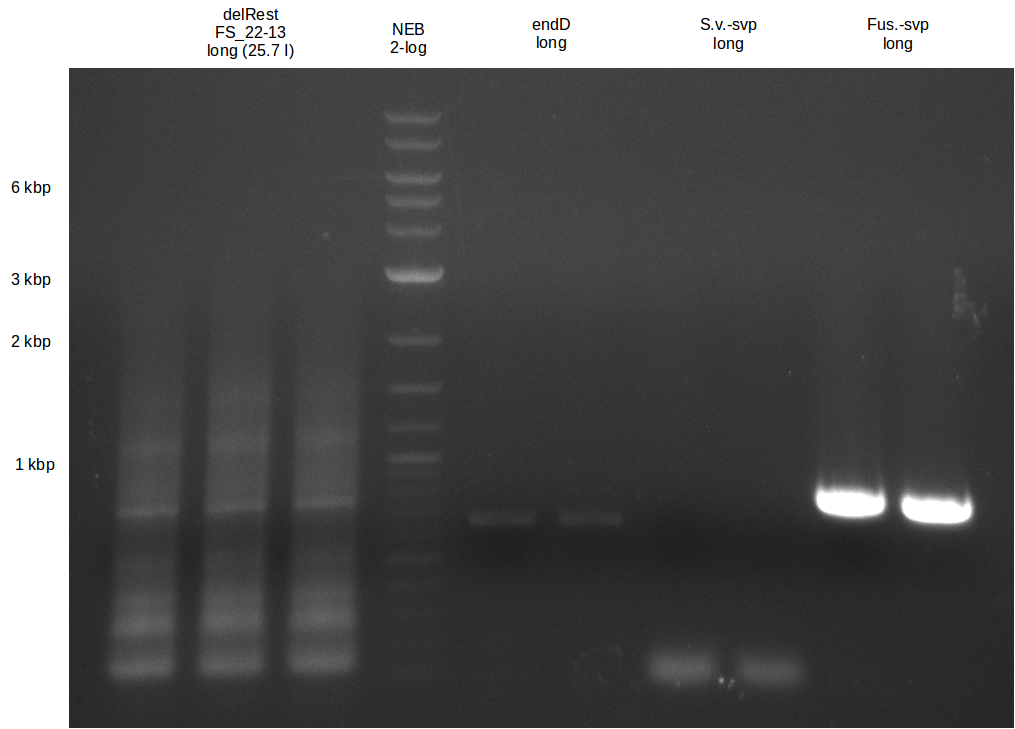
Amplification from FS_22 to FS_13s; 2.7 kb
2x20µl
- Reaction
| what | µl |
|---|---|
| D.acidovorans DSM-39 | 1 |
| FS_22: (1/10) | 4 |
| FS_13_short: (1/10) | 4 |
| Phusion flash Master Mix | 10 |
| dd H2O | 1 |
20µl
- Reaction
| what | µl |
|---|---|
| D.acidovorans DSM-39 | 1 |
| FS_22: (1/10) | 2 |
| FS_13_short: (1/10) | 2 |
| Phusion flash Master Mix | 10 |
| dd H2O | 5 |
- Conditions
| Biorad MyCycler* | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 12 | 98 | 1 |
| 65 ↓ 0.5 | 5 | |
| 72 | 1:00 | |
| 18 | 98 | 1 |
| 63 | 5 | |
| 72 | 1:00 | |
| 1 | 72 | 5min |
| 1 | 12 | inf |
Results:
- Amplification of DelOP resulted in a small band atthe desired lenght, but also a smear and several unexpected bands
- bands were cut out and DNA purified using QIAquick Gel Extraction Kit
- amplicon will be used for restriction digest to validate the construct
Amplification from FS_22 to FS_13(s/l); 2.7 kb
- Reaction
| what | µl |
|---|---|
| D.acidovorans DSM-39 | 1 |
| FS_22: (1/10) | 2 |
| FS_13_short/FS_13_long: (1/10) | 2 |
| Phusion flash Master Mix | 10 |
| DMSO | 1/- |
| dd H2O | 4/5 |
- Conditions
| Biorad C1000 Touch | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 30 | 98 | 1 |
| 72 | 1:00 | |
| 1 | 72 | 5 min |
| 1 | 12 | inf |
- Conditions
| Biorad MyCycler* | ||
|---|---|---|
| Cycles | temperature [°C] | Time [s] |
| 1 | 98 | 10 |
| 30 | 98 | 1 |
| 70 | 5 | |
| 72 | 1:00 | |
| 1 | 72 | 5 min |
| 1 | 12 | inf |
 "
"