Template:Kyoto/Notebook/Aug 21
From 2013.igem.org
(Difference between revisions)
(Created page with "==Aug 21== ===Plusgrow Medium=== <div class="experiment"> <span class="author">Tatsui</span> *material :*8/17 Plusgrow medium 50ml :*7/22 5000x Ampicillin 10µl # Measurin...") |
(→Plusgrow Medium) |
||
| (30 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
*material | *material | ||
:*8/17 Plusgrow medium 50ml | :*8/17 Plusgrow medium 50ml | ||
| - | :*7/22 | + | :*7/22 5000x Ampicillin 10µl |
# Measuring materials and putting them in a 50ml tube | # Measuring materials and putting them in a 50ml tube | ||
</div> | </div> | ||
| Line 61: | Line 61: | ||
===Gel Extraction=== | ===Gel Extraction=== | ||
<div class="experiment"> | <div class="experiment"> | ||
| - | <span class="author">Nakamoto and | + | <span class="author">Nakamoto and Tatsui</span> |
{| class="wikitable" | {| class="wikitable" | ||
!Lane||DNA||Enzyme | !Lane||DNA||Enzyme | ||
| Line 83: | Line 83: | ||
|12 | |12 | ||
|} | |} | ||
| - | [ | + | [[File:Igku Aug21 Gel Extraction(Ptet‐(1))before.jpg]] |
| + | [[File:Igku Aug21 Gel Extraction 2-2.jpg]] | ||
| + | |||
{| class="wikitable" | {| class="wikitable" | ||
!Name||concentration[µg/mL]||260/280||260/230 | !Name||concentration[µg/mL]||260/280||260/230 | ||
| Line 101: | Line 103: | ||
<span class="author">Kojima and Nakamoto</span> | <span class="author">Kojima and Nakamoto</span> | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | !8/20 Ptet-(1) ||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total | + | ! ||8/20 Ptet-(1) ||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total |
|- | |- | ||
| - | |4.5||1||1||3||3||17.5||30 | + | |2 cuts||4.5||1||1||3||3||17.5||30 |
|- | |- | ||
| - | |0.5||0.2||0||1||1||7.3||10 | + | |1 cut||0.5||0.2||0||1||1||7.3||10 |
|- | |- | ||
| - | |0.5||0||0.2||1||1||7.3||10 | + | |1 cut||0.5||0||0.2||1||1||7.3||10 |
|- | |- | ||
| - | |0.5||0||0||1||1||7.5||10 | + | |NC||0.5||0||0||1||1||7.5||10 |
|} | |} | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | !8/20 Pconst-RBS-luxR-DT-(2) ||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total | + | ! ||8/20 Pconst-RBS-luxR-DT-(2) ||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total |
|- | |- | ||
| - | |2.9||1||1||3||3||19.1||30 | + | |2 cuts||2.9||1||1||3||3||19.1||30 |
|- | |- | ||
| - | |0.3||0.2||0||1||1||7.5||10 | + | |1 cut||0.3||0.2||0||1||1||7.5||10 |
|- | |- | ||
| - | |0.3||0||0.2||1||1||7.5||10 | + | |1 cut||0.3||0||0.2||1||1||7.5||10 |
|- | |- | ||
| - | |0.3||0||0||1||1||7.7||10 | + | |NC||0.3||0||0||1||1||7.7||10 |
|} | |} | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | !8/17 DT||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total | + | ! ||8/17 DT||EcoRI||XbaI||10x BSA||10x buffer M||MilliQ||total |
|- | |- | ||
| - | |5.3||1||1||3||3||16.7||30 | + | |2 cuts||5.3||1||1||3||3||16.7||30 |
|- | |- | ||
| - | |0.5||0.2||0||1||1||7.3||10 | + | |1 cut||0.5||0.2||0||1||1||7.3||10 |
|- | |- | ||
| - | |0.5||0||0.2||1||1||7.3||10 | + | |1 cut||0.5||0||0.2||1||1||7.3||10 |
|- | |- | ||
| - | |0.5||0||0||1||1||7.5||10 | + | |NC||0.5||0||0||1||1||7.5||10 |
|} | |} | ||
| - | * incubate 37°C 1h | + | * incubate at 37°C for 1h |
</div> | </div> | ||
| Line 175: | Line 177: | ||
|} | |} | ||
* The reason why lane 2 and 15 were same is that the well of lane 2 might be broken. | * The reason why lane 2 and 15 were same is that the well of lane 2 might be broken. | ||
| - | [ | + | [[File:Igku Aug21 Electorophoresis Rtet E+X.jpg]]<br> |
<span class="author">Kojima</span> | <span class="author">Kojima</span> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 186: | Line 188: | ||
|3||8/20 DT||EcoRI||XbaI | |3||8/20 DT||EcoRI||XbaI | ||
|} | |} | ||
| - | [ | + | [[File:Igku Aug21 Electrophoresis(J23100‐RBS,DT-E+X)bykojima.jpg ]]<br> |
</div> | </div> | ||
| Line 198: | Line 200: | ||
|1||1kb ladder||-- | |1||1kb ladder||-- | ||
|- | |- | ||
| - | |2||8/21 J23100-RBS-luxR-DT 25µL|| | + | |2||8/21 J23100-RBS-luxR-DT 25µL||EcoRI+XbaI |
|- | |- | ||
| - | |3||8/21 J23100-RBS-luxR-DT 25µL|| | + | |3||8/21 J23100-RBS-luxR-DT 25µL||EcoRI+XbaI |
|- | |- | ||
|4||--||-- | |4||--||-- | ||
|- | |- | ||
| - | |5||8/21 Ptet 25µL|| | + | |5||8/21 Ptet 25µL||EcoRI+XbaI |
|- | |- | ||
| - | |6||8/21 Ptet 25µL|| | + | |6||8/21 Ptet 25µL||EcoRI+XbaI |
|} | |} | ||
| - | [[File: | + | [[File:Igku Aug21 Gel Extraction(J23100,Ptet①)before.jpg]]<br> |
| - | [[File: | + | [[File:Igku Aug21 Gel Extraction(J23100,Ptet)after.jpg]]<br> |
</div> | </div> | ||
<!-- ここまでをコピーしてね --> | <!-- ここまでをコピーしてね --> | ||
===Electrophoresis=== | ===Electrophoresis=== | ||
| - | |||
<div class="experiment"> | <div class="experiment"> | ||
<span class="author">Honda</span> | <span class="author">Honda</span> | ||
| Line 230: | Line 231: | ||
|5||8/21 DT||--||-- | |5||8/21 DT||--||-- | ||
|} | |} | ||
| - | [[File: | + | [[File:Igku_Aug21electrophoresis.jpg]]<br> |
</div> | </div> | ||
| - | |||
===Ligation=== | ===Ligation=== | ||
| - | |||
<div class="experiment"> | <div class="experiment"> | ||
<span class="author">No name</span> | <span class="author">No name</span> | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | ! | + | !state||colspan="2"|Vector||colspan="2"|Inserter||Ligation High ver.2||total |
|- | |- | ||
| - | |8/21 Pcn-RBS-luxR-DT(+Amp)||2.3|| | + | |experiment||8/21 Pcn-RBS-luxR-DT(+Amp)||2.3||8/21 Pcn-RBS-GFP-DT||1.7||2||6 |
|- | |- | ||
| - | |8/21 Pcn-RBS- | + | |NC||8/21 Pcn-RBS-luxR-DT(+Amp)||2.3||MilliQ||1.7||2||6 |
|- | |- | ||
| - | | | + | |experiment||8/18 Plux(+CP)||1.1||8/19 RBS-GFP-DT||5||3.1||9.2 |
|- | |- | ||
| - | |MilliQ|| | + | |NC||8/18 Plux(+CP)||1.1||MilliQ||5||3.1||9.2 |
|- | |- | ||
| - | | | + | |experiment||8/18 RBS(+Amp)||2.2||8/19 lysis1||6.0||4.1||12.3 |
| - | | | + | |
| - | + | ||
| - | + | ||
| - | + | ||
|- | |- | ||
| - | |8/18 | + | |experiment||8/18 RBS(+Amp)||2.2||8/19 lysis2||3.6||2.9||8.7 |
|- | |- | ||
| - | |8/ | + | |NC||8/18 RBS(+Amp)||2.2||MilliQ||3.6||2.9||8.7 |
|- | |- | ||
| - | | | + | |experiment||8/21 Ptet||1.5||8/20 RBS-tetR-DT (2)||2.1||1.8||5.4 |
|- | |- | ||
| - | | | + | |NC||8/21 Ptet||1.5||MilliQ||2.1||1.8||5.4 |
| - | | | + | |
| - | | | + | |
|} | |} | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
</div> | </div> | ||
| - | |||
===Transformation=== | ===Transformation=== | ||
| Line 330: | Line 296: | ||
===Gel Extraction=== | ===Gel Extraction=== | ||
| - | |||
<div class="experiment"> | <div class="experiment"> | ||
<span class="author">Okazaki</span> | <span class="author">Okazaki</span> | ||
| Line 336: | Line 301: | ||
!Lane||DNA||Enzyme | !Lane||DNA||Enzyme | ||
|- | |- | ||
| - | |1||1kb ladder|| | + | |1||1kb ladder||-- |
|- | |- | ||
|2||--||-- | |2||--||-- | ||
|- | |- | ||
| - | |3||8/21 DT | + | |3||rowspan="2"|8/21 DT||rowspan="2"|EcoRI & XbaI |
|- | |- | ||
| - | |4 | + | |4 |
|} | |} | ||
| - | |||
| - | |||
</div> | </div> | ||
| - | |||
===Miniprep=== | ===Miniprep=== | ||
Latest revision as of 03:45, 28 September 2013
Contents |
Aug 21
Plusgrow Medium
- material
- 8/17 Plusgrow medium 50ml
- 7/22 5000x Ampicillin 10µl
- Measuring materials and putting them in a 50ml tube
Liquid culture
| Sample | medium |
|---|---|
| 8/20 tRNA-spinach -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 tRNA-spinach -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_P(1076) -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_P(1076) -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_1R(113) -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_1R(113) -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_1M(105) -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 TetR-aptamer 12_1M(105) -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 PT181 attenuator -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 PT181 attenuator -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion1 attenuator -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion1 attenuator -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion3m2 attenuator -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion3m2 attenuator -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 PT181 antisense -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 PT181 antisense -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion1 antisense -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion1 antisense -(2) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion6 antisense -(1) | 8/21 Plusgrow medium(+Amp) |
| 8/20 Fusion6 antisense -(2) | 8/21 Plusgrow medium(+Amp) |
- incubate 37°C 10hour
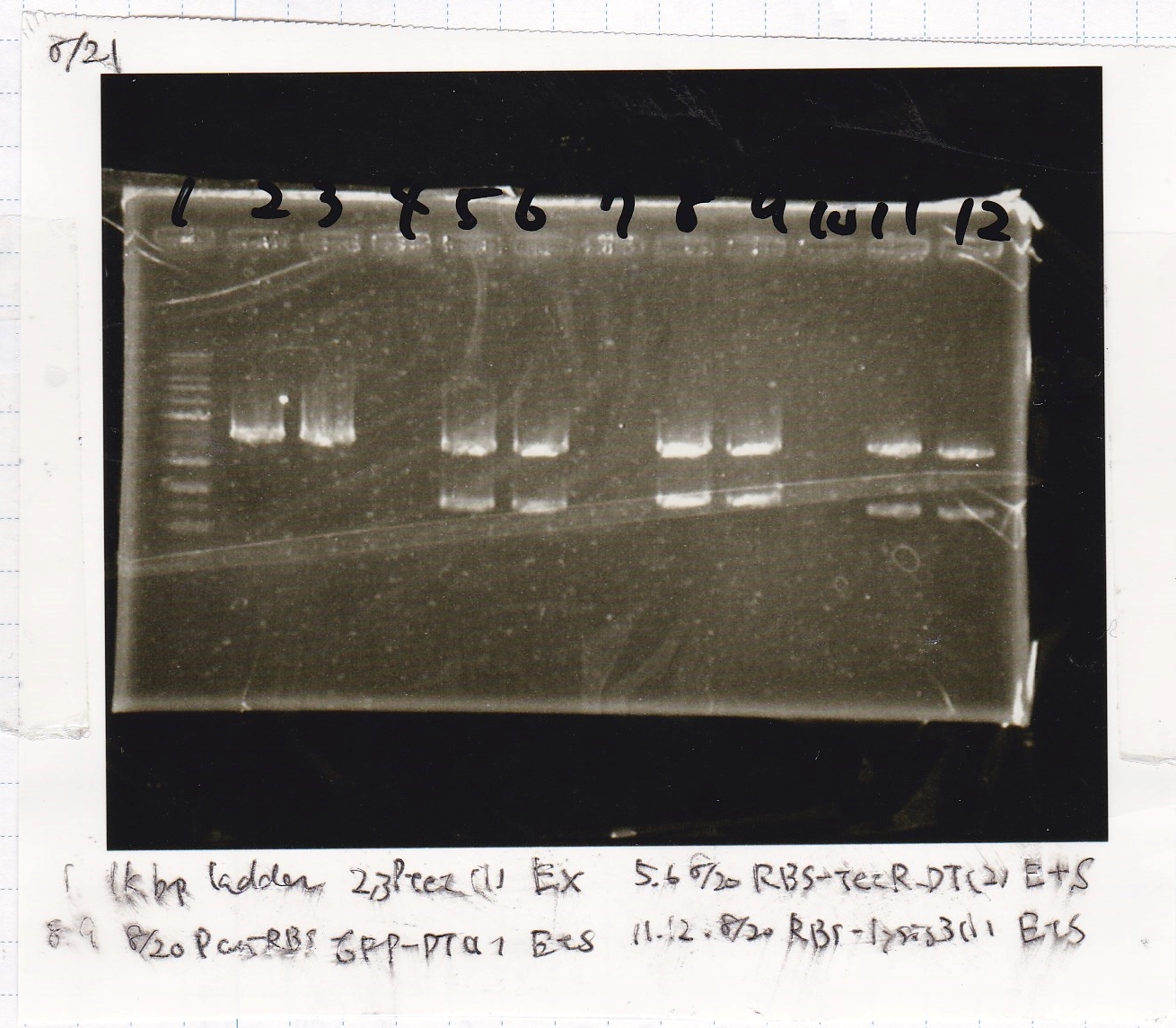
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 2 | 8/20 Ptet -(1) | EcoRI & XbaI |
| 3 | ||
| 5 | 8/20 RBS-tetR-DT -(2) | EcoRI & SpeI |
| 6 | ||
| 8 | 8/20 Pconst-RBS-GFP-DT -(1) | EcoRI & SpeI |
| 9 | ||
| 11 | 8/20 RBS-lysis3 -(1) | EcoRI & SpeI |
| 12 |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| Ptet (EcoRI & XbaI) | 21 | 1.55 | 0.78 |
| RBS-tetR-DT (EcoRI & SpeI) | 12 | 1.45 | 0.62 |
| Pconst-RBS-GFP-DT (EcoRI & SpeI) | 11 | 1.36 | 0.60 |
| RBS-lysis3 (EcoRI & SpeI) | 10 | 1.26 | 0.50 |
Ristriction Enzyme Digestion
| 8/20 Ptet-(1) | EcoRI | XbaI | 10x BSA | 10x buffer M | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 4.5 | 1 | 1 | 3 | 3 | 17.5 | 30 |
| 1 cut | 0.5 | 0.2 | 0 | 1 | 1 | 7.3 | 10 |
| 1 cut | 0.5 | 0 | 0.2 | 1 | 1 | 7.3 | 10 |
| NC | 0.5 | 0 | 0 | 1 | 1 | 7.5 | 10 |
| 8/20 Pconst-RBS-luxR-DT-(2) | EcoRI | XbaI | 10x BSA | 10x buffer M | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 2.9 | 1 | 1 | 3 | 3 | 19.1 | 30 |
| 1 cut | 0.3 | 0.2 | 0 | 1 | 1 | 7.5 | 10 |
| 1 cut | 0.3 | 0 | 0.2 | 1 | 1 | 7.5 | 10 |
| NC | 0.3 | 0 | 0 | 1 | 1 | 7.7 | 10 |
| 8/17 DT | EcoRI | XbaI | 10x BSA | 10x buffer M | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 5.3 | 1 | 1 | 3 | 3 | 16.7 | 30 |
| 1 cut | 0.5 | 0.2 | 0 | 1 | 1 | 7.3 | 10 |
| 1 cut | 0.5 | 0 | 0.2 | 1 | 1 | 7.3 | 10 |
| NC | 0.5 | 0 | 0 | 1 | 1 | 7.5 | 10 |
- incubate at 37°C for 1h
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 1kbp ladder | -- | -- |
| 2 | 8/20 Ptet -(1) | EcoRI | XbaI |
| 3 | 8/20 Ptet -(1) | EcoRI | -- |
| 4 | 8/20 Ptet -(1) | -- | XbaI |
| 5 | 8/20 Ptet -(1) | -- | -- |
| 6 | 8/20 J23100-RBS-luxR-DT -(2) | EcoRI | XbaI |
| 7 | 8/20 J23100-RBS-luxR-DT -(2) | EcoRI | -- |
| 8 | 8/20 J23100-RBS-luxR-DT -(2) | -- | XbaI |
| 9 | 8/20 J23100-RBS-luxR-DT -(2) | -- | -- |
| 10 | 8/20 DT | EcoRI | XbaI |
| 11 | 8/20 DT | EcoRI | -- |
| 12 | 8/20 DT | -- | XbaI |
| 13 | 8/20 DT | -- | -- |
| 14 | 1kbp ladder | -- | -- |
| 15 | 8/20 Ptet -(1) | EcoRI | XbaI |
- The reason why lane 2 and 15 were same is that the well of lane 2 might be broken.
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 1kbp ladder | -- | -- |
| 2 | 8/20 J23100-RBS-luxR-DT -(2) | EcoRI | XbaI |
| 3 | 8/20 DT | EcoRI | XbaI |
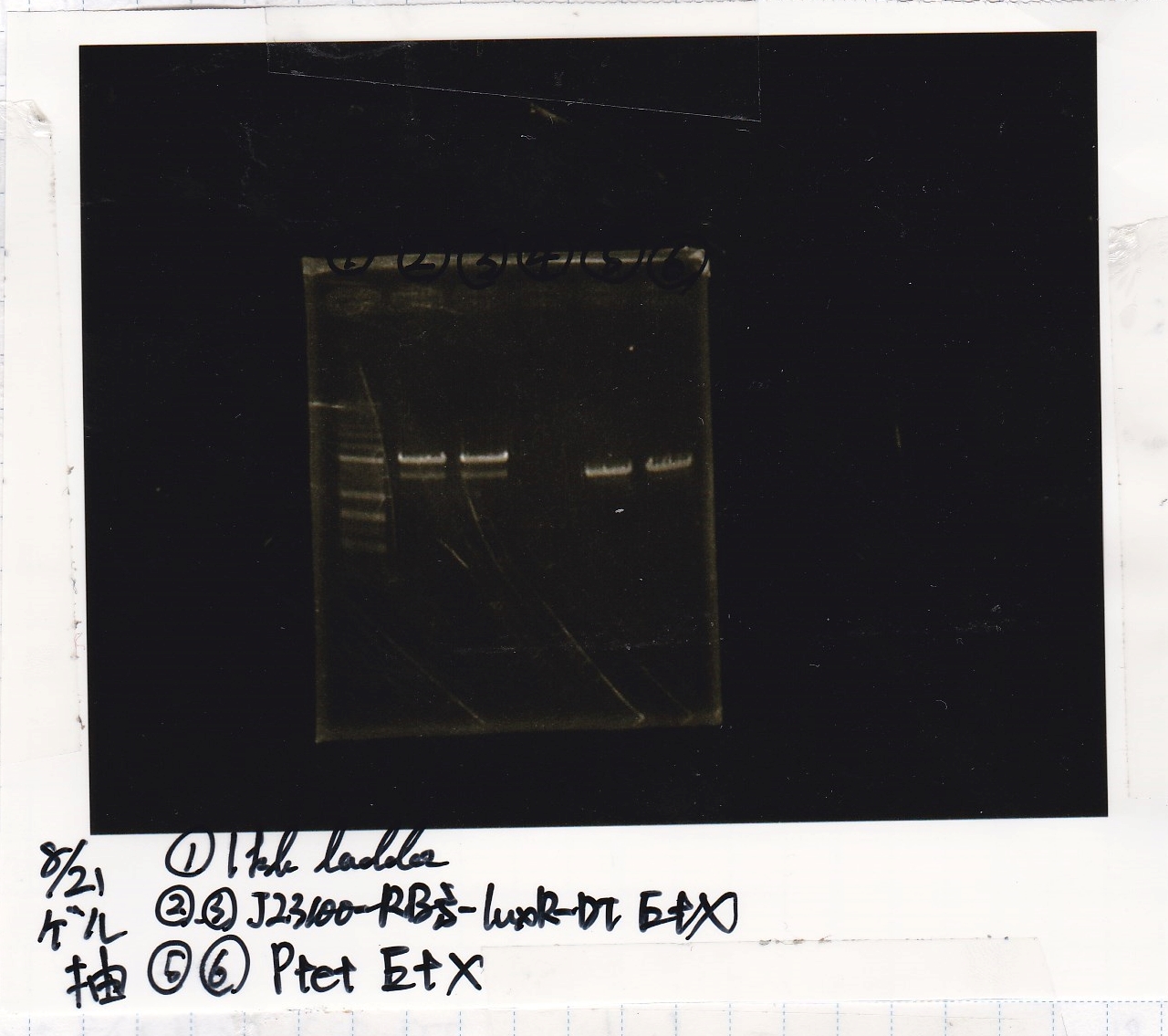
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kb ladder | -- |
| 2 | 8/21 J23100-RBS-luxR-DT 25µL | EcoRI+XbaI |
| 3 | 8/21 J23100-RBS-luxR-DT 25µL | EcoRI+XbaI |
| 4 | -- | -- |
| 5 | 8/21 Ptet 25µL | EcoRI+XbaI |
| 6 | 8/21 Ptet 25µL | EcoRI+XbaI |
Electrophoresis
Ligation
| state | Vector | Inserter | Ligation High ver.2 | total | ||
|---|---|---|---|---|---|---|
| experiment | 8/21 Pcn-RBS-luxR-DT(+Amp) | 2.3 | 8/21 Pcn-RBS-GFP-DT | 1.7 | 2 | 6 |
| NC | 8/21 Pcn-RBS-luxR-DT(+Amp) | 2.3 | MilliQ | 1.7 | 2 | 6 |
| experiment | 8/18 Plux(+CP) | 1.1 | 8/19 RBS-GFP-DT | 5 | 3.1 | 9.2 |
| NC | 8/18 Plux(+CP) | 1.1 | MilliQ | 5 | 3.1 | 9.2 |
| experiment | 8/18 RBS(+Amp) | 2.2 | 8/19 lysis1 | 6.0 | 4.1 | 12.3 |
| experiment | 8/18 RBS(+Amp) | 2.2 | 8/19 lysis2 | 3.6 | 2.9 | 8.7 |
| NC | 8/18 RBS(+Amp) | 2.2 | MilliQ | 3.6 | 2.9 | 8.7 |
| experiment | 8/21 Ptet | 1.5 | 8/20 RBS-tetR-DT (2) | 2.1 | 1.8 | 5.4 |
| NC | 8/21 Ptet | 1.5 | MilliQ | 2.1 | 1.8 | 5.4 |
Transformation
| Name | Sample | Competent Cells | Total | Plate |
|---|---|---|---|---|
| 8/21 Pcon-RBS-GFP-DT+Pcon-RBS-GFP-DT | 1µL | 10µL | 11µL | Amp |
| 8/21 Pcon-RBS-GFP-DT NC | 1µL | 10µL | 11µL | Amp |
| 8/18 RBS+8/19 lysis1 | 1µL | 10µL | 11µL | Amp |
| 8/18 RBS+8/19 lysis2 | 1µL | 10µL | 11µL | Amp |
| 8/18 RBS NC | 1µL | 10µL | 11µL | Amp |
| 8/18 Plux+8/18 RBS-GFP-DT | 1µL | 10µL | 11µL | CP |
| 8/18 Plux+8/18 RBS-GFP-DT | 1µL | 10µL | 11µL | CP |
| 8/21 Ptet(pm)+8/20 RBS-tetR-DT(2) | 1µL | 10µL | 11µL | CP |
| 8/21 Ptet(pm)+8/20 RBS-tetR-DT(2) NC | 1µL | 10µL | 11µL | CP |
| 8/21 pSB1C3 | 1µL | 10µL | 11µL | CP |
LB Medium Plate
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kb ladder | -- |
| 2 | -- | -- |
| 3 | 8/21 DT | EcoRI & XbaI |
| 4 |
Miniprep
| DNA | concentration [µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| tRNA-spinach-(1) | 234 | 1.64 | 2.14 |
| tRNA-spinach-(2) | 440 | 1.60 | 1.91 |
| tetR-aptamer 12-p(1076)-(1) | 150 | 1.61 | 2.03 |
| tetR-aptamer 12-p(1076)-(2) | 374 | 1.45 | 1.66 |
| tetR-aptamer 12-1R(113)-(1) | 304 | 1.68 | 2.21 |
| tetR-aptamer 12-1R(113)-(2) | 382 | 1.47 | 1.66 |
| tetR-aptamer 12-1M(105)-(1) | 374 | 1.68 | 2.20 |
| tetR-aptamer 12-1M(105)-(2) | 286 | 1.31 | 1.43 |
| PT181 attenuator-(1) | 330 | 1.66 | 2.11 |
| PT181 attenuator-(2) | 270 | 1.68 | 2.18 |
| Fusion1 attenuator-(1) | 306 | 1.67 | 2.20 |
| Fusion1 attenuator-(2) | 282 | 1.60 | 1.94 |
| Fusion3m2 attenuator-(1) | 332 | 1.68 | 2.25 |
| Fusion3m2 attenuator-(2) | 326 | 1.67 | 2.10 |
| PT181 antisense-(1) | 178 | 1.64 | 1.68 |
| PT181 antisense-(2) | 116 | 1.63 | 1.95 |
| Fusion1 antisense-(1) | 300 | 1.67 | 2.21 |
| Fusion1 antisense-(2) | 164 | 1.42 | 1.46 |
| Fusion6 antisense-(1) | 336 | 1.65 | 2.18 |
| Fusion6 antisense-(2) | 192 | 1.65 | 2.01 |
 "
"