Template:Kyoto/Notebook/Aug 19
From 2013.igem.org
(Difference between revisions)
(→Electrophoresis) |
(→Liquid Culture) |
||
| (5 intermediate revisions not shown) | |||
| Line 39: | Line 39: | ||
[[File:Igku Aug19electrophoresis1(colony PCR).png]] | [[File:Igku Aug19electrophoresis1(colony PCR).png]] | ||
<br> | <br> | ||
| + | {| class="wikitable" | ||
| + | !Lane||DNA | ||
| + | |- | ||
| + | |1||100bp ladder | ||
| + | |- | ||
| + | |2||8/18 NC J23100(1) | ||
| + | |- | ||
| + | |3||8/18 J23100-RBS-GFP-DT-(1) | ||
| + | |- | ||
| + | |4||8/18 J23100-RBS-GFP-DT-(2) | ||
| + | |- | ||
| + | |5||8/18 J23100-RBS-luxR-DT-(1) | ||
| + | |- | ||
| + | |6||8/18 J23100-RBS-luxR-DT-(2) | ||
| + | |- | ||
| + | |7||8/18 J23100-RBS-lacZα-DT-(1) | ||
| + | |- | ||
| + | |8||8/18 J23100-RBS-lacZα-DT-(2) | ||
| + | |- | ||
| + | |9||8/18 RBS-lysis3-(1) | ||
| + | |- | ||
| + | |10||8/18 RBS-lysis3-(2) | ||
| + | |- | ||
| + | |11||8/18 R0400(RBS-tetR-DT)-(1) | ||
| + | |- | ||
| + | |12||8/18 R0400(RBS-tetR-DT)-(2) | ||
| + | |- | ||
| + | |13||NC | ||
| + | |- | ||
| + | |14||100bp ladder | ||
| + | |} | ||
| + | |||
| + | [[File:Igku Aug19electrophoresis1(Colony PCR).png]]<br> | ||
| + | {| class="wikitable" | ||
| + | !Name||concentration[µg/mL]||260/280||260/230 | ||
| + | |- | ||
| + | |8/19 lysis1 ① (XbaI & PstI)||87.8||1.06||0.82 | ||
| + | |} | ||
| + | |||
| + | |||
{| class="wikitable" | {| class="wikitable" | ||
!Sample||base pair | !Sample||base pair | ||
| Line 95: | Line 135: | ||
|NaCl||1g | |NaCl||1g | ||
|- | |- | ||
| - | |Agar Pouder|| | + | |Agar Pouder||2g |
|} | |} | ||
</div> | </div> | ||
| Line 211: | Line 251: | ||
|8/18 Ptet -2||Plusgrow medium(+CP) | |8/18 Ptet -2||Plusgrow medium(+CP) | ||
|- | |- | ||
| - | |8/18 RBS-tetR-DT -1||Plusgrow medium(+CP) | + | |8/18 RBS-tetR-DT -1||Plusgrow medium(+CP) |
|- | |- | ||
| - | |8/18 RBS-tetR-DT -2||Plusgrow medium(+CP) | + | |8/18 RBS-tetR-DT -2||Plusgrow medium(+CP) |
|- | |- | ||
|8/18 J23100-GFP -1||Plusgrow medium(+Amp) | |8/18 J23100-GFP -1||Plusgrow medium(+Amp) | ||
Latest revision as of 03:44, 28 September 2013
Contents |
Aug 19
Colony PCR
| Sample | base pair |
|---|---|
| 8/18 J23100 control -(1) | 1142 |
| 8/18 J23100-RBS-GFP-DT -(1) | 1156 |
| 8/18 J23100-RBS-GFP-DT -(2) | 1156 |
| 8/18 J23100-RBS-luxR-DT -(1) | 1271 |
| 8/18 J23100-RBS-luxR-DT -(2) | 1271 |
| 8/18 J23100-RBS-lacZα-DT -(1) | 670 |
| 8/18 J23100-RBS-lacZα-DT -(2) | 670 |
| 8/18 RBS-lysis3 -(1) | 997 |
| 8/18 RBS-lysis3 -(2) | 997 |
| 8/18 P0440(RBS-tetR-DT) -(1) | 1154 |
| 8/18 P0440(RBS-tetR-DT) -(2) | 1154 |
| negative control | -- |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 1min | 30cycles |
| Lane | DNA |
|---|---|
| 1 | 100bp ladder |
| 2 | 8/18 NC J23100(1) |
| 3 | 8/18 J23100-RBS-GFP-DT-(1) |
| 4 | 8/18 J23100-RBS-GFP-DT-(2) |
| 5 | 8/18 J23100-RBS-luxR-DT-(1) |
| 6 | 8/18 J23100-RBS-luxR-DT-(2) |
| 7 | 8/18 J23100-RBS-lacZα-DT-(1) |
| 8 | 8/18 J23100-RBS-lacZα-DT-(2) |
| 9 | 8/18 RBS-lysis3-(1) |
| 10 | 8/18 RBS-lysis3-(2) |
| 11 | 8/18 R0400(RBS-tetR-DT)-(1) |
| 12 | 8/18 R0400(RBS-tetR-DT)-(2) |
| 13 | NC |
| 14 | 100bp ladder |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| 8/19 lysis1 ① (XbaI & PstI) | 87.8 | 1.06 | 0.82 |
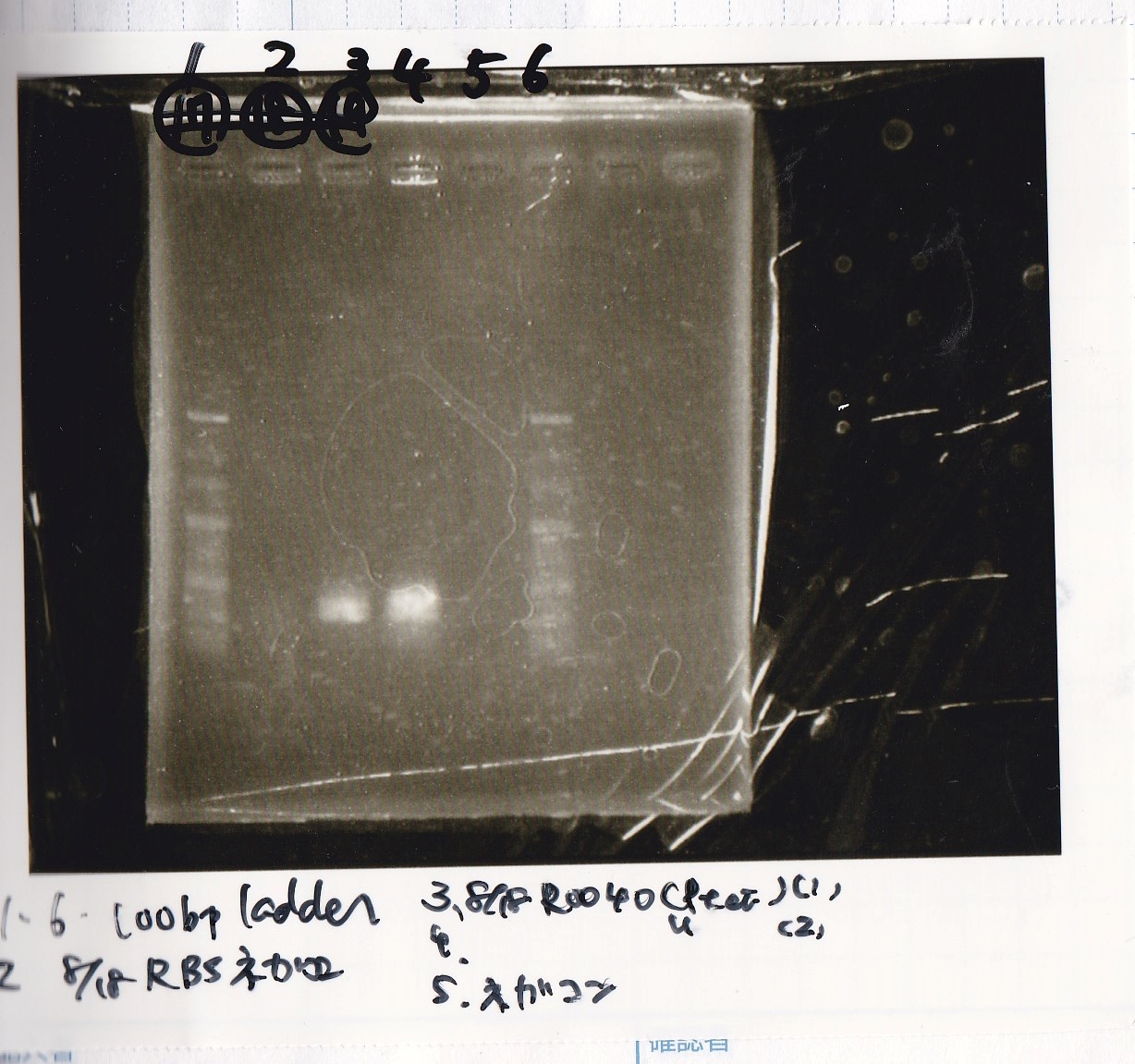
| Sample | base pair |
|---|---|
| 8/18 RBS control -(1) | -- |
| 8/18 R0040(Ptet) -(1) | 686 |
| 8/18 R0040(Ptet) -(2) | 686 |
| negative control | -- |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 30s | 30cycles |
Gel Extraction
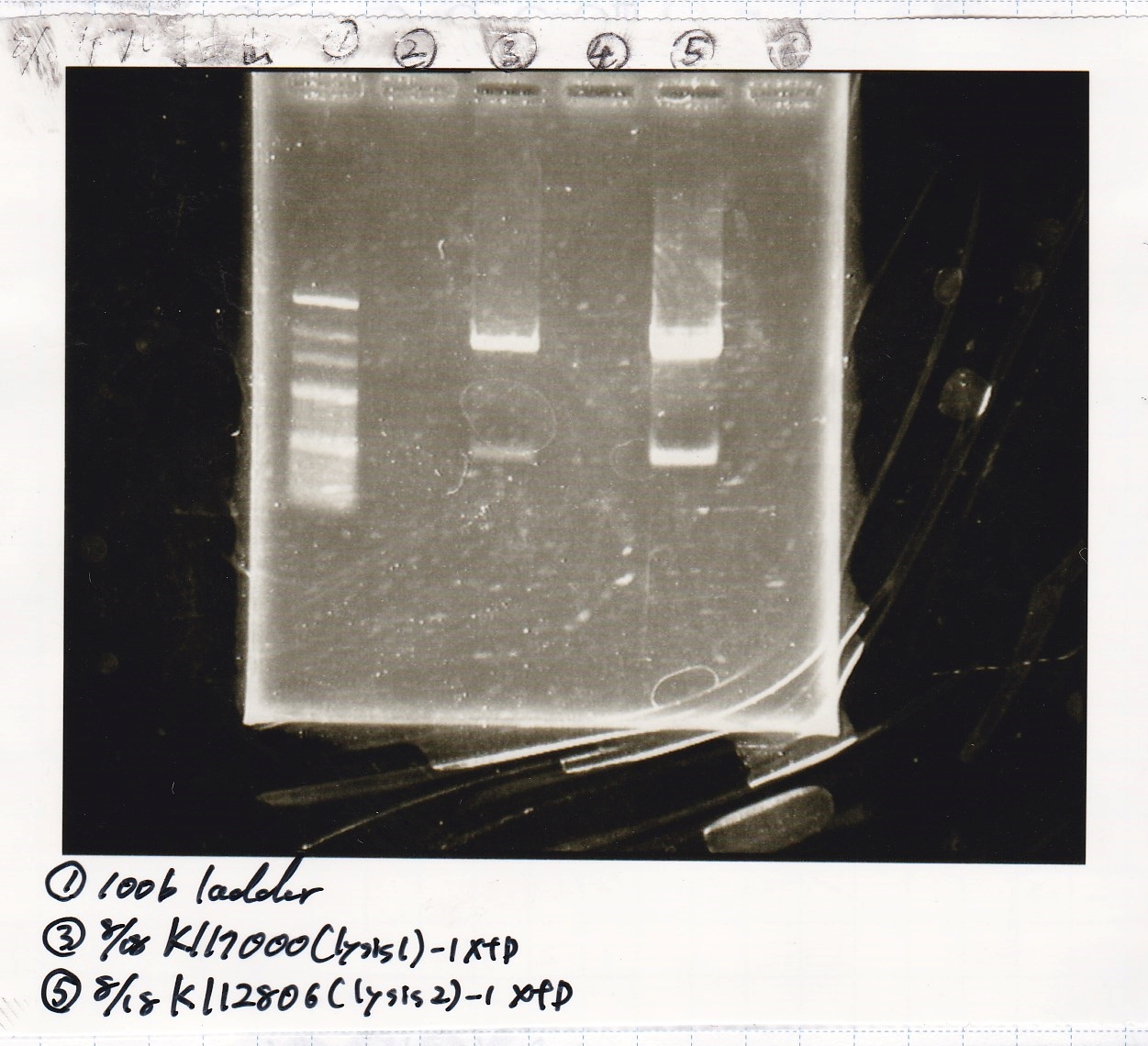
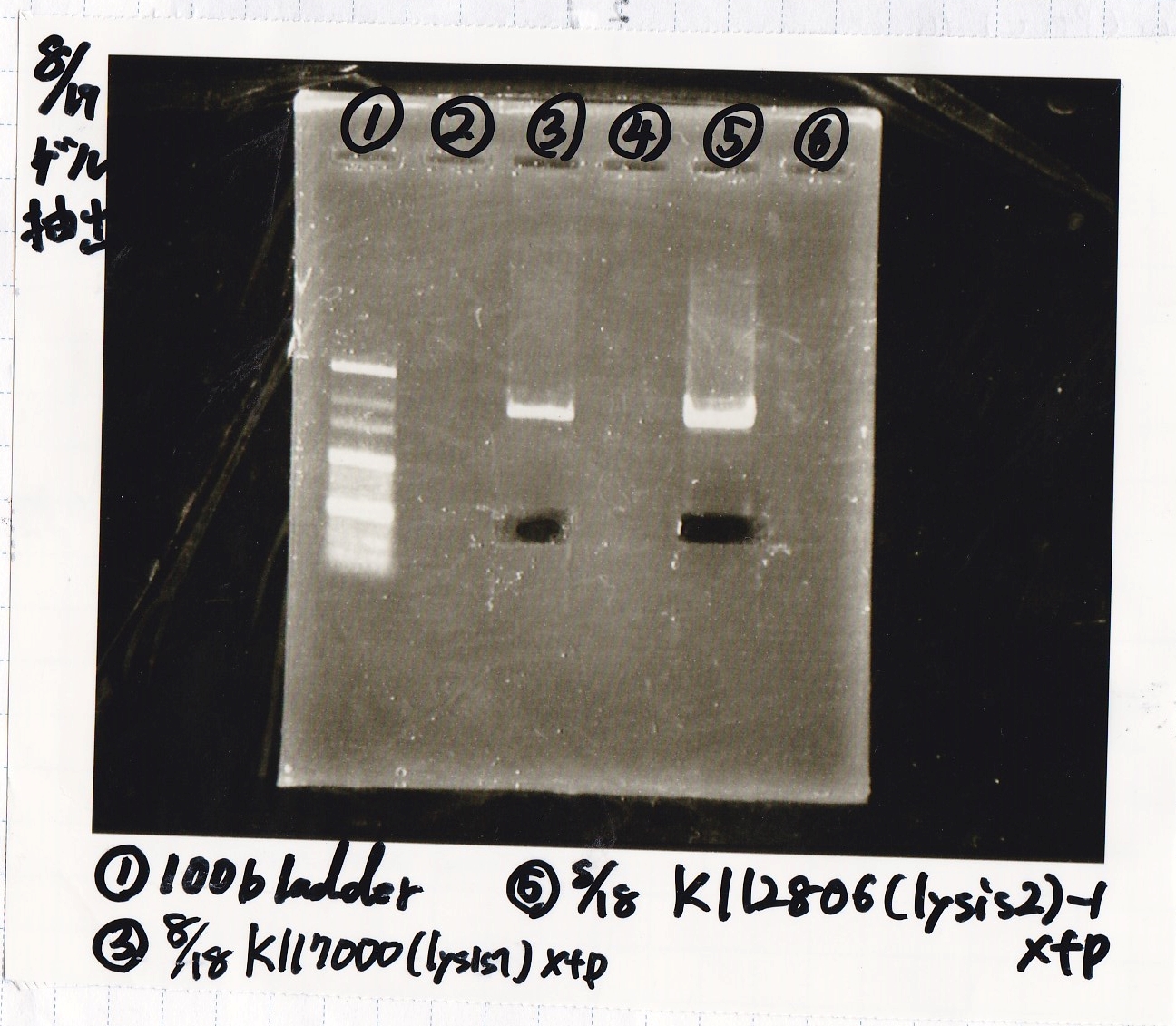
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 100bp ladder | -- |
| 3 | 8/18 K117000(lysis2) -1 | XbaI & PstI |
| 5 | 8/18 K117000(lysis2) -1 | XbaI & PstI |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| 8/18 lysis2-1(XbaI & PstI) | 3.1 | 3.64 | 0.19 |
| 8/18 lysis2-1(XbaI & PstI) | 3.4 | 1.89 | 0.13 |
LB Medium Plate
| volume | 200ml |
|---|---|
| Bacto T2ypton | 2g |
| Bacto yeast extract | 1g |
| NaCl | 1g |
| Agar Pouder | 2g |
Restriction Enzyme Digestion
| lysis2-1 | XbaI | PstI | 10xbuffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 10µL | 1µL | 1µL | 3µL | 3µL | 12µL | 30µL |
| 1 cut | 2µL | 0.2µL | -- | 1µL | 1µL | 5.8µL | 10µL |
| 1 cut | 2µL | -- | 0.2µL | 1µL | 1µL | 5.8µL | 10µL |
| NC | 2µL | -- | -- | 1µL | 1µ | 6µL | 10µL |
- at 37°C, for 1h
| 8/19 lysis1-1 | XbaI | PstI | 10xbuffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 2µL | 0.5µL | 0.8µL | 2µL | 2µL | 13µL | 20µL |
| 1 cut | 0.5µL | 0.2µL | -- | 1µL | 1µL | 7.3µL | 10µL |
| 1 cut | 0.5µL | -- | 0.2µL | 1µL | 1µL | 7.3µL | 10µL |
| NC | 0.5µL | -- | -- | 1µL | 1µL | 7.5µL | 10µL |
- at 37°C, for 1h
Ligation
| state | Vector | Inserter | Ligation High ver.2 | ||
|---|---|---|---|---|---|
| experiment | RBS | 0.3 | lysis2 | 7.4 | 4 |
| NC | RBS | 0.3 | MilliQ | 7.4 | 4 |
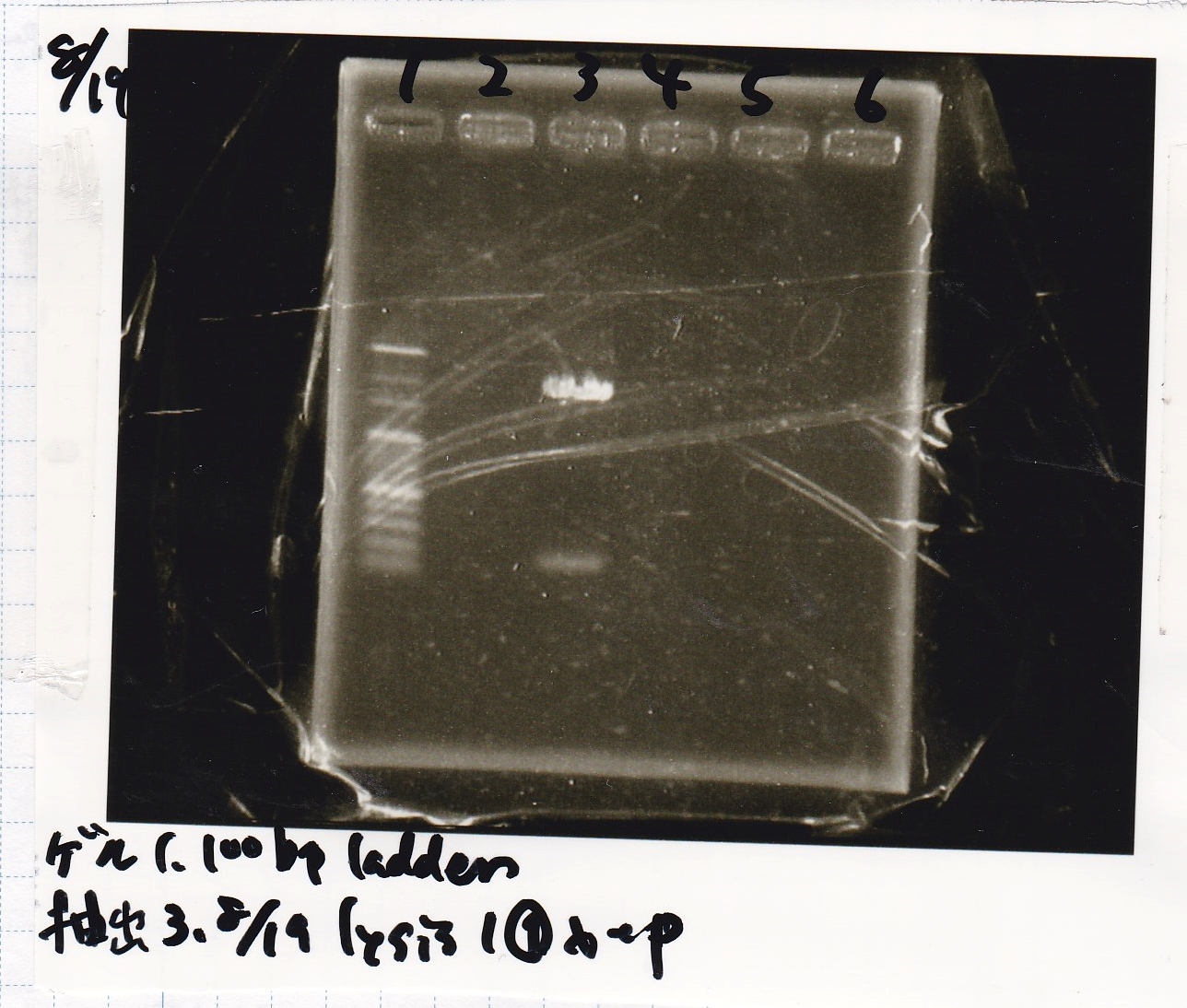
Electrophoresis
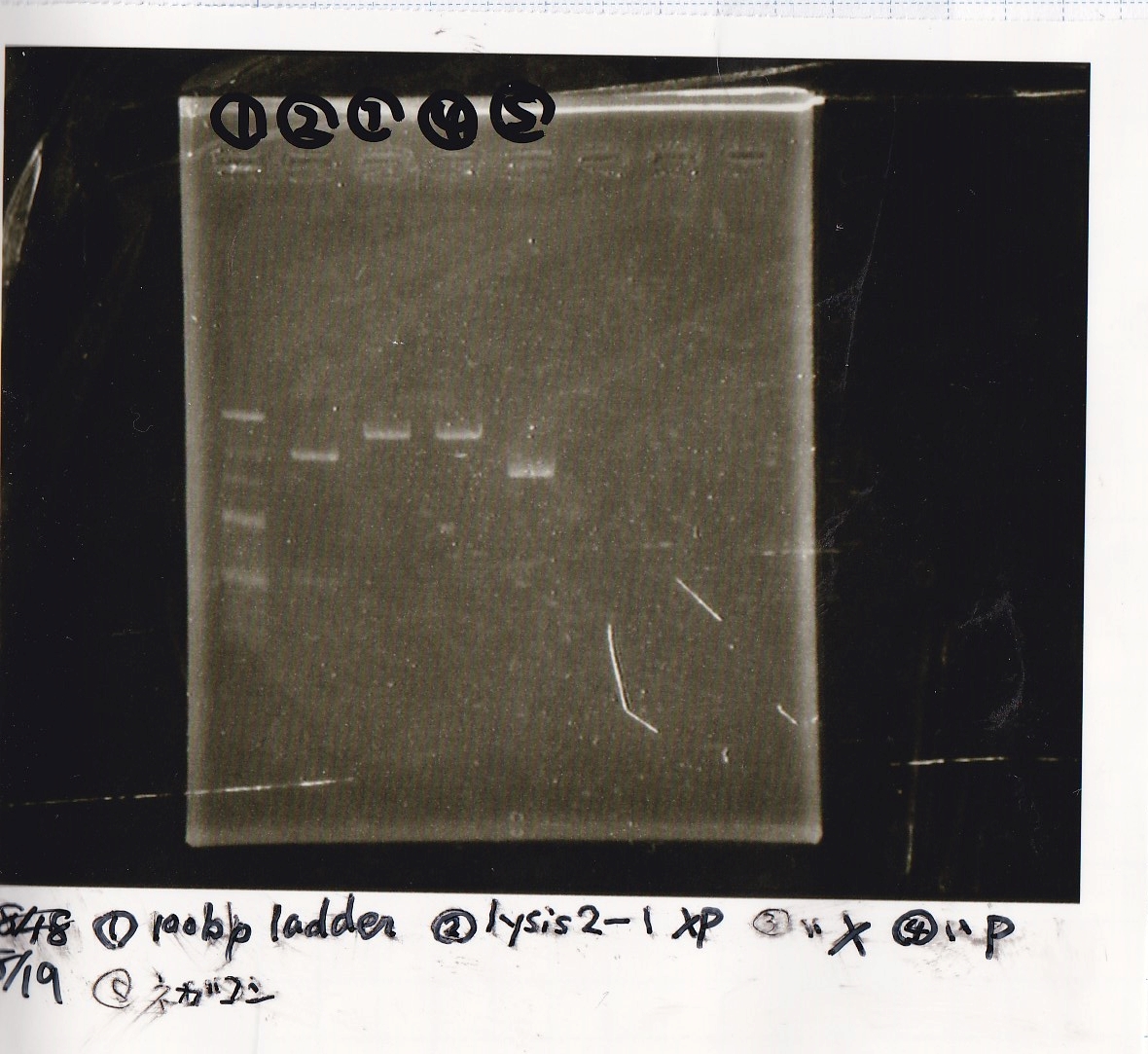
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | 8/19 lysis1 | XbaI | PstI |
| 3 | 8/19 lysis1 | XbaI | -- |
| 4 | 8/19 lysis1 | -- | PstI |
| 5 | 8/19 lysis1 | -- | -- |
| 6 | 8/19 lysis1(Gel Extraction Product) | XbaI | PstI |
Transformation
| Name | Sample | Competent Cells | Plate |
|---|---|---|---|
| 8/18 Plux+RBS-GFP-DT | 2µL | 20µL | CP |
| 8/18 Plux(NC) | 2µL | 20µL | CP |
| 8/19 RBS+lysis2 | 2µL | 20µL | Amp |
| 8/15 Pbad-araC | 2µL | 20µL | Kan |
- Incubate at 37°C
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 100bp ladder | -- |
| 2 | 8/19 lysis1 ① | XbaI & PstI |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| 8/19 lysis1 ① (XbaI & PstI) | 87.8 | 1.06 | 0.82 |
Liquid Culture
| Sample | medium |
|---|---|
| 8/18 Ptet -1 | Plusgrow medium(+CP) |
| 8/18 Ptet -2 | Plusgrow medium(+CP) |
| 8/18 RBS-tetR-DT -1 | Plusgrow medium(+CP) |
| 8/18 RBS-tetR-DT -2 | Plusgrow medium(+CP) |
| 8/18 J23100-GFP -1 | Plusgrow medium(+Amp) |
| 8/18 J23100-luxR -1 | Plusgrow medium(+Amp) |
| 8/18 J23100-luxR -2 | Plusgrow medium(+Amp) |
| 8/18 J23100-lacZα -1 | Plusgrow medium(+Amp) |
| 8/18 RBS-lysis3 -1 | Plusgrow medium(+Amp) |
| 8/18 RBS-lysis3 -2 | Plusgrow medium(+Amp) |
Master Plate
| Number | Use LB plate(+Amp) |
|---|---|
| 1 | 8/18 J23100-GFP -1 |
| 2 | 8/18 J23100-luxR -1 |
| 3 | 8/18 J23100-luxR -2 |
| 4 | 8/18 J23100-lacZα -1 |
| 5 | 8/18 RBS-lysis3 -1 |
| 6 | 8/18 RBS-lysis3 -2 |
| Number | Use LB plate(+CP) |
|---|---|
| 1 | 8/18 Ptet -1 |
| 1 | 8/18 Ptet -2 |
| 3 | 8/18 RBS-tetR-DT -1 |
| 4 | 8/18 RBS-tetR-DT -2 |
 "
"