Template:Kyoto/Notebook/Sep 10
From 2013.igem.org
(Difference between revisions)
(→Ligation) |
(→Gel Extraction) |
||
| (One intermediate revision not shown) | |||
| Line 175: | Line 175: | ||
|} | |} | ||
</div> | </div> | ||
| + | [[File:Igku_0910_E3.jpg]] | ||
===Gel Extraction=== | ===Gel Extraction=== | ||
| Line 229: | Line 230: | ||
|} | |} | ||
</div> | </div> | ||
| - | + | [[File:Igku_0910_G3.jpg]] | |
===Liquid Culture=== | ===Liquid Culture=== | ||
Latest revision as of 18:06, 27 September 2013
Contents |
Sep 10
Ligation
| state | Vector | Inserter | Ligation High ver.2 | ||
|---|---|---|---|---|---|
| experiment | 9/6 Pcon-RBS-luxR-DT | 1.9µL | 9/6Plux-RBS-GFP-DT | 22 µL | 3.5µL |
| experiment | 9/6 Plux | 2µL | 9/8 RBS-lysis1-DT | 6.6µL | 3.5µL |
| experiment | 9/6 Plux | 2µL | 9/8 RBS-lysis2-DT | 6.3µL | 3.5µL |
| experiment | 9/8 Ptet-PT181 antisense | 1.6µL | 9/8 spinach-DT | 4.4µL | 3.0µL |
| experiment | 9/8 Pcon-PT181 attenuator | 3.4µL | 9/8 apz 12-1R-PT | 2.1µL | 2.8µL |
| experiment | 9/8 Pcon | 3.4µL | 9/8 apz 12-1R-PT | 2.3µL | 2,9µL |
| experiment | 9/8 DT | 3.0µL | 9/8 Pcon-PT181 attenuator | 7.9µL | 3.5µL |
| experiment | 9/10 pSB1C3 | 1.8µL | 9/8 F3m2 attenuator | 4.6µL | 3.2µL |
| experiment | 9/10 pSB1C3 | 1.8µL | 9/8 F1 attenuator | 3.4µL | 2.6µL |
Miniprep
| DNA | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| spinach-DT | 202.4 | 1.97 | 1.93 |
| Plux-RBS-GFP-DT | 164.2 | 2.80 | 2.06 |
| Pcon-DT attenuator | 205.4 | 1.81 | 2.39 |
| RSBJC3-2 | 240.8 | 1.73 | 1.43 |
| Plaz-1 | 240.8 | 1.73 | 1.43 |
Restriction Enzyme Digestion
| 9/10 Plux-RBS-GFP-DT | EcoRI | SpeI | Buffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 12.1µL | 1µL | 1µL | 3µL | 3µL | 9.9µL | 30µL |
| NC | 0.6µL | 0µL | 0µL | 1µL | 1µL | 7.4µL | 10µL |
| 9/10 Spinach-DT | XbaI | PstI | Buffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 9.9µL | 1µL | 1µL | 3µL | 3µL | 12.1µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 1µL | 1µL | 7.5µL | 10µL |
| 9/6 apt12-1R -DT | XbaI | PstI | Buffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 8.0µL | 1µL | 1µL | 3µL | 3µL | 14.0µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL |
| 9/10 Pcon-pt81 attenuator | EcoRI | SpeI | Buffer | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2cuts | 9.8µL | 1µL | 1µL | 3µL | 3µL | 12.2µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 1µL | 1µL | 7.5µL | 10µL |
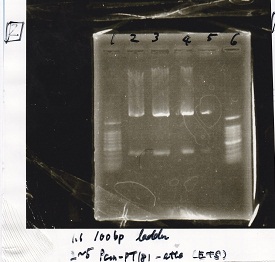
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | Plux-RBS-GEP-DT | EcoRI | SpeI |
| 3 | Plux-RBS-GEP-DT | -- | -- |
| 4 | Spinach-DT | XbaI | PstI |
| 5 | Spinach-DT | -- | -- |
| 6 | aptamer 121R-DT | XbaI | PstI |
| 7 | aptamer 121R-DT | -- | -- |
| 8 | Pcon-pt181 attenuator | EcoRI | SpeI |
| 9 | Pcon-pt181 attenuator | -- | -- |
| 10 | 100bp ladder | -- | -- |
Colony PCR
| Sample | base pair |
|---|---|
| 9/9 Pcon-pT181 antisense | -- |
| 9/8 J223100(entA) | -- |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 1min10sec | 30 |
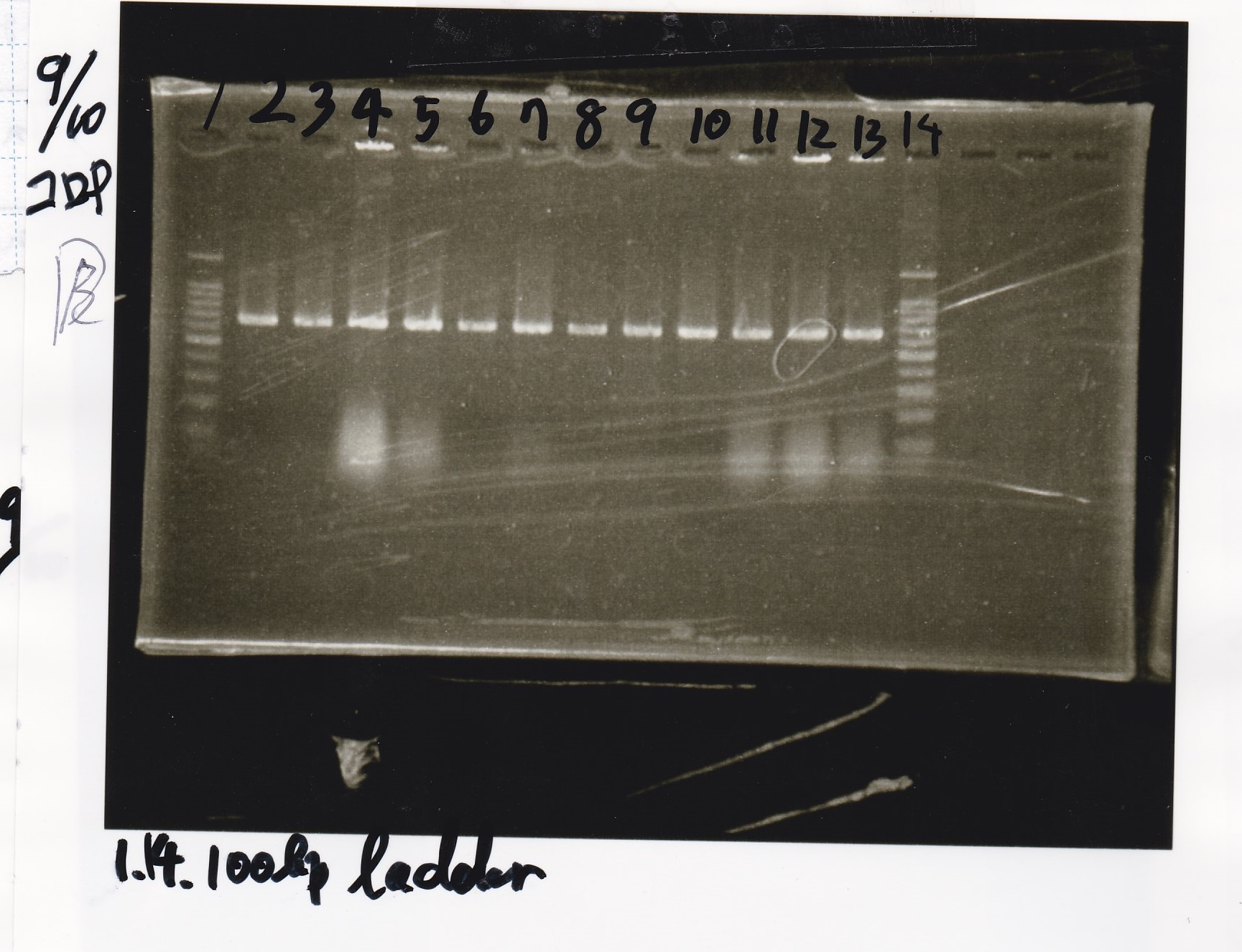
Electrophoresis
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | Pcon Pt181 antisense1+spinach-DT |
| 3 | Pcon Pt181 antisense2+spinach-DT |
| 4 | Pcon Pt181 antisense3+spinach-DT |
| 5 | Pcon Pt181 antisense4+spinach-DT |
| 6 | Pcon Pt181 antisense5+spinach-DT |
| 7 | Pcon Pt181 antisense6+spinach-DT |
| 8 | Pcon Pt181 antisense7+spinach-DT |
| 9 | Pcon Pt181 antisense8+spinach-DT |
| 10 | Pcon Pt181 antisense9+spinach-DT |
| 11 | Pcon Pt181 antisense10+spinach-DT |
| 12 | Pcon Pt181 antisense11+spinach-DT |
| 13 | Pcon Pt181 antisense12+spinach-DT |
| 14 | 100bp ladder |
| Lane | Sample |
|---|---|
| 1 | 1kbp ladder |
| 2 | Pcon entA1 |
| 3 | Pcon entA2 |
| 4 | Pcon entA3 |
| 5 | 1kbp ladder |
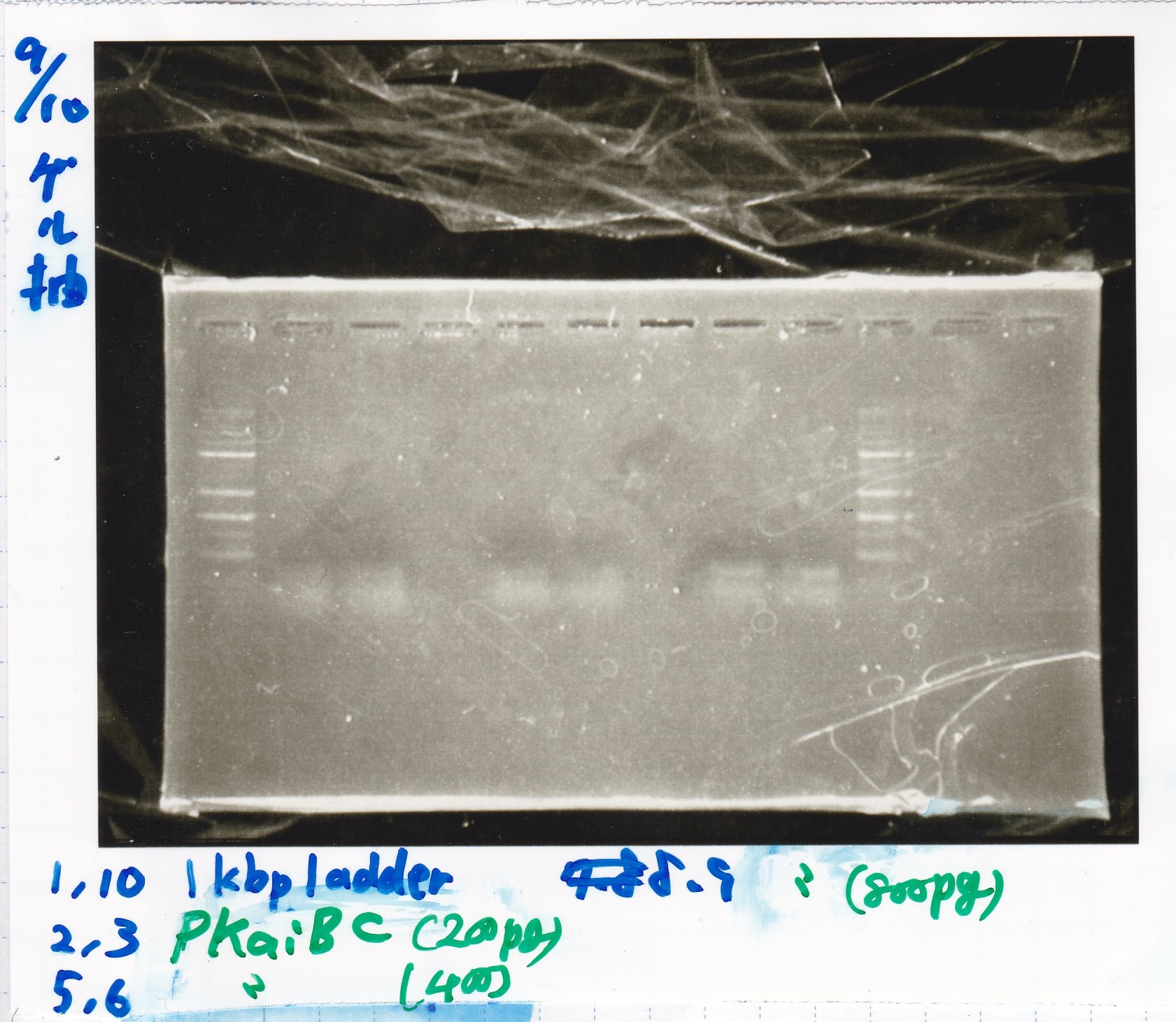
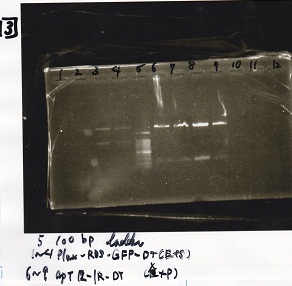
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 2 | KaiA(200pg) | -- |
| 3 | KaiA(200pg) | -- |
| 4 | -- | -- |
| 5 | KaiA(400pg) | -- |
| 6 | KaiA(400pg) | -- |
| 7 | -- | -- |
| 8 | KaiA(800pg | -- |
| 9 | KaiA(800pg) | -- |
| 10 | 1kbp ladder | -- |
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 2 | PKaiBC(200pg) | -- |
| 3 | PKaiBC(200pg) | -- |
| 4 | -- | -- |
| 5 | PKaiBC(400pg) | -- |
| 6 | PKaiBC(400pg) | -- |
| 7 | -- | -- |
| 8 | PKaiBC(800pg | -- |
| 9 | PKaiBC(800pg) | -- |
| 10 | 1kbp ladder | -- |
Liquid Culture
| Sample | medium |
|---|---|
| 9/4 J23100①(JM109) | LB(Amp) |
| 9/8 J23100① (entA) | LB(Amp) |
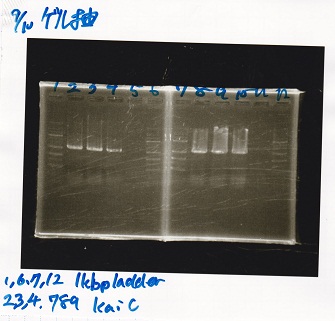
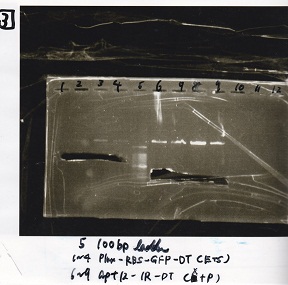
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 1kbp ladder | -- |
| 2 | KaiC | -- |
| 3 | KaiC | -- |
| 4 | KaiC | -- |
| 5 | -- | -- |
| 6 | 1kbp ladder | -- |
| 7 | 1kbp ladder | -- |
| 8 | KaiC | -- |
| 9 | KaiC | -- |
| 10 | KaiC | -- |
| 11 | -- | -- |
| 12 | 1kbp ladder | -- |
Liquid Culture
| Sample | medium |
|---|---|
| 9/10 Pcon-pt181 antisense-Spinach-DT | Plusgrow |
Colony PCR
| Sample | base pair |
|---|---|
| 9/9 Phad larac-1 RBS-lux1-DT | 2331 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 2min | 30 |
| Sample | base pair |
|---|---|
| 9/9 Pcon-lacZα (psB4KS) | 712 |
| 9/9 Pcon-Spinach-DT(psB4KS) | 605 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 42s | 30 |
| Sample | base pair |
|---|---|
| 9/9 Plac+RBS-lysis3}-DT | 1273 |
| 9/9 Pcon+RBS-lux1-DT | 1079 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 1min12s | 30 |
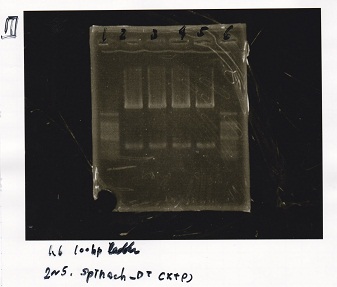
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | 9/9 Pcou-RBS-lacZα-DT(p3B4KS9)-1 | -- | -- |
| 3 | 9/9 Pcou-RBS-lacZα-DT(p3B4KS9)-2 | -- | -- |
| 4 | 9/9 Pcou-Spinach-DT(pB4KS)-1 | -- | -- |
| 5 | 9/9 Pcou-Spinach-DT(pB4KS)-2 | -- | -- |
| 6 | 100bp ladder | -- | -- |
Transformation
| Name | Plate |
|---|---|
| F1 attenuator +p3B1C3 | CP |
| F3n2 attenuator +p3B1C3 | CP |
| F1 antisense +p3B1C3 | CP |
| F6 antisense+p3B1C3 | CP |
| apt12-P +p3B1C3 | CP |
| apt12-1M +p3B1C3 | CP |
| Pcon-luxR+Plux-GFP | Amp |
| Plux+RBS-lysis1-DT | CP |
| Plux+RBS-lysis2-DT | CP |
| Ptet-pT181antisense+Spinach-DT | CP |
| Pcon-pT181 attenuator+apt121R-DT | Amp |
| Pcon-1 apt12-1R-DT | Amp |
| DT+Pcon-pT181 attenuator | CP |
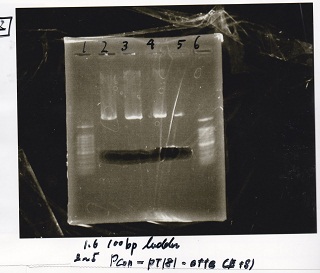
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | Spinach-DT | XbaI | PstI |
| 3 | Spinach-DT | XbaI | PstI |
| 4 | Spinach-DT | XbaI | PstI |
| 5 | Spinach-DT | XbaI | PstI |
| 6 | 100bp ladder | -- | -- |
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | Pcon-pT181 attenuator | EcoRI | SpeI |
| 3 | Pcon-pT181 attenuator | EcoRI | SpeI |
| 4 | Pcon-pT181 attenuator | EcoRI | Spei |
| 5 | Pcon-pT181 attenuator | EcoRI | SpeI |
| 6 | 100bp ladder | -- | -- |
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | Plux-RBS-GFP-DT | EcoRI | SpeI |
| 2 | Plux-RBS-GFP-DT | EcoRI | SpeI |
| 3 | Plux-RBS-GFP-DT | EcoRI | SpeI |
| 4 | Plux-RBS-GFP-DT | EcoRI | SpeI |
| 5 | 100bp ladder | -- | -- |
| 6 | apt12-1R-DT | XbaI | PstI |
| 7 | apt12-1R-DT | XbaI | PstI |
| 8 | apt12-1R-DT | XbaI | PstI |
| 9 | apt12-1R-DT | XbaI | PstI |
 "
"