Team:Biwako Nagahama/Material & Method
From 2013.igem.org
(→CelC) |
(→CrdS) |
||
| Line 319: | Line 319: | ||
</table> | </table> | ||
</div> | </div> | ||
| - | left | + | <div align="left"> |
| + | <p>Front</p> | ||
<p>Fw0 Primer:GGCCGCTTCTAGATGTATTTCAGTGC</p> | <p>Fw0 Primer:GGCCGCTTCTAGATGTATTTCAGTGC</p> | ||
<p>Rv1 Primer:CGTTTCGAGGGAGAACTCCAGCG</p> | <p>Rv1 Primer:CGTTTCGAGGGAGAACTCCAGCG</p> | ||
| Line 328: | Line 329: | ||
<p>Fw2 Primer:GGCGCTTCAGGCCGATGAGCTG</p> | <p>Fw2 Primer:GGCGCTTCAGGCCGATGAGCTG</p> | ||
<p>Rv0 Primer:GGCGCTACTAGTATTATTATCACCCGAATG</p> | <p>Rv0 Primer:GGCGCTACTAGTATTATTATCACCCGAATG</p> | ||
| + | </div> | ||
<div align="right"> | <div align="right"> | ||
<table border="1"> | <table border="1"> | ||
Revision as of 20:58, 27 September 2013
Material & Method
CelC
Agro Notebook
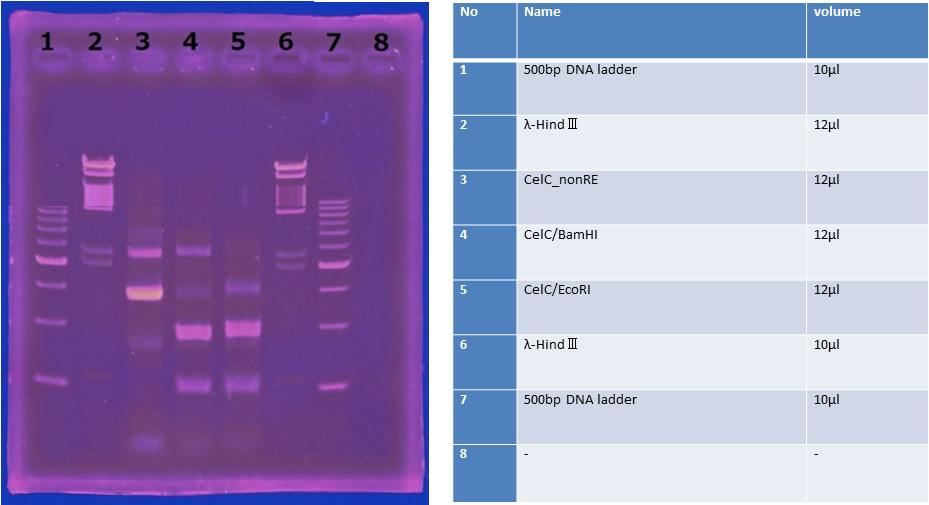
5/31 Cloning of CelC and Restriction Enzyme
By Koki Tsutsumi
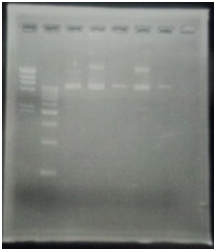
CelC gene had produced clone from Agrobacterium tumefaciens C58, but I confirmed whether it’s true or not. CelC gene has restriction enzyme sites,EcoRI and BamHI.
Inverse PCR
※Restriction Enzyme sol.(line No.5 CelC_EcoRI)which has 1M NaCl dissolved in it, so, the band in lane no.5 appeared as upper-side.
CelC_Fw: TGACGAAAGCACTGATCTGC
CelC_Rv: GAAAAGATCGAAACGGTGG
TA cloning of CelC
16℃ 30min incubate
Ligation of CelC/pMD20 and Transformation in JM109.
Cells were stored on ice for 30min.
After 42℃ 30sec heat shock, cells were stored on ice for 2min.
Then cells were pre-cultured at 37℃ for 1hr, plated to Ampicillin plate.
6/1 Liquid clluture
CelC/pMD20 22 samples at 37°C, for overnight.
6/2 MiniPrep of CelC
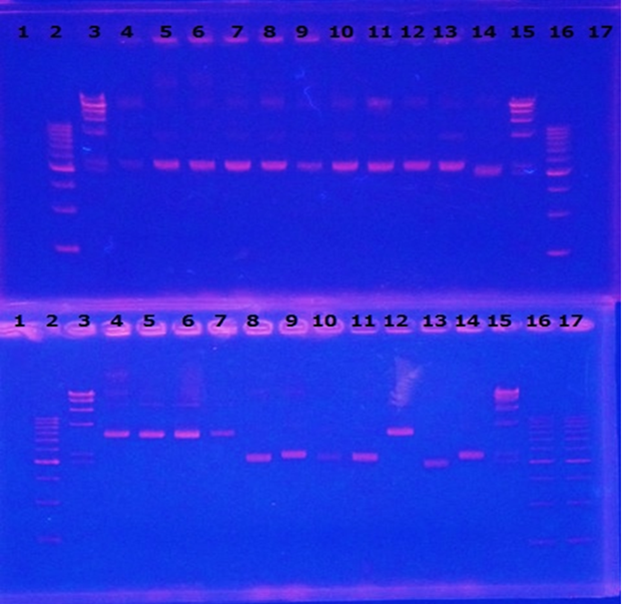
Linear CelC/pMD20 DNA :2736bp. This CelC/pMD20 sample is cccDNA. So,white 6,white 7,white 8,white 9,white 14 probably picked up CelC/pMD20.
6/3 Restriction Enzyme of CelC/pMD20
CelC gene had produced clone from Agrobacterium tumefaciens C58, I confirmed whether it’s true or not. CelC/pMD20 gene has 2 restriction enzyme sites,BamHI
I confirmed the direction of CelC gene.
6/ Sequence of CelC/pMD20
7/18 PointMutation of CelC
CelC gene had Restriction Enzyme Site,EcoRI. We directed the EcoRI site.
Front
Fw1 Primer:
TATATATTCTAGATGAAGAGCGGGATTTCG
Rv2 Primer:
CATTATATCCGAACTCCGGCTG
Rear
Fw2 Primer:
AGCCGGAGTTCGGATATAATGC
Rv1 Primer:
CAGCACGAACTAGTATTATTATCATCGGC
7/19 Gel Purification of CelC gene’s Front Fragment and Rear Fragment
Front Fragment DNA and Rear Fragment DNA that 765bp and 347bp band performed Gel Purification by illustra GFX PCR Purification Kit.
7/21 CelC gene’s Front Fragment and Rear Fragment Overlap PCR
No.6,7,8,9,14 each fragment Overlap PCR completed.
I selected No.8.
7/22 Gel Purification of CelC gene No.8
No.8 DNA that about 1ooo bp band performed Gel Purification by illustra GFX PCR Purification Kit.
8/1 Adapter PCR of CelC gene No.8 (BioBrick Part)
Fw Primer:
GTTTCTTCGAATTCGCGGCCGCTTCTAGATG
Rv Primer:
GTTTCTTCCTGCAGCGGCCGCTACTAGTATTATTATC
8/11 BioBrick of CelC Restrict Enzyme ,EcoRI.
I confirmed BioBrick of CelC non-Restriction Enzyme ,EcoRI.
9/5 Brick Part of CelC and pSB1C3 Restrict Enzyme,EcoRI and PstI.
9/5 Ligation of CelC/pSB1C3 and Transformation in JM109.
9/7 Colony PCR of CelC/pSB1C3
CelC/pSB1C3 DNA(VR-VF2) :1370bp. So,No.5 and No.12 probably picked up CelC/pSB1C3
9/8 Miniprep of CelC/pSB1C3
Linear CelC/pSB1C3 DNA :3126bp. This CelC/pSB1C3 sample is cccDNA. So,No.5 and No.12 probably picked up CelC/pSB1C3.
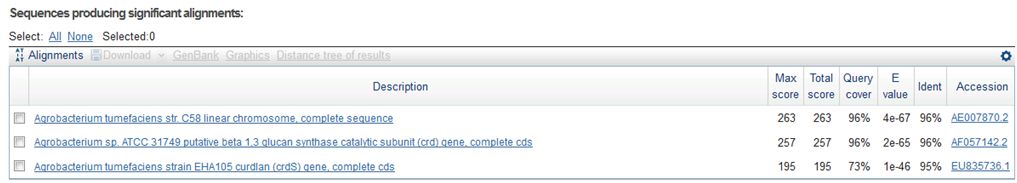
9/15. Sequence of CelC Brick Part sequence.
Result of NCBI BLAST
CrdS
Cloning of CrdS and Restriction Enzyme
By Koki Tsutsumi
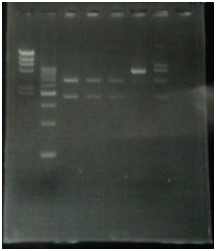
CrdS gene had produced clone from Agrobacterium tumefaciens C58, but I confirmed whether it’s true or not. CrdS gene has restriction enzyme sites,EcoRI and PstI.
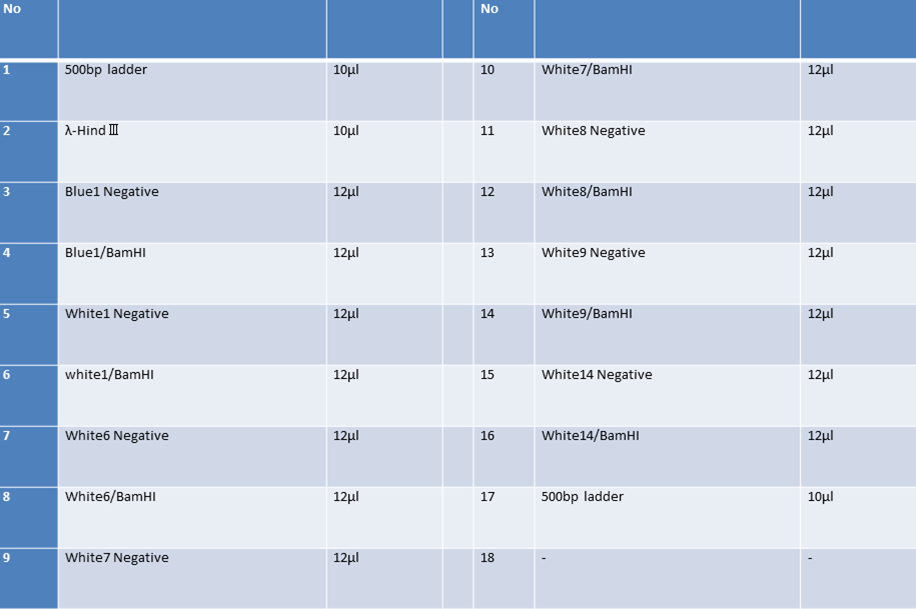
| No | Name | volume |
| 1 | 500bp DNA ladder | 10μL |
| 2 | λ-HindⅢ | 10μL |
| 3 | CrdS | 12μL |
| 4 | - | - |
| 5 | - | - |
| 6 | - | - |
| 7 | - | - |
| 8 | - | - |
| No | Name | volume |
| 1 | 500bp DNA ladder | 10μL |
| 2 | λ-HindⅢ | 10μL |
| 3 | CrdS_nonRE | 12μL |
| 4 | CrdS/EcoRⅠ | 12μL |
| 5 | CrdS/PstⅠ | 12μL |
| 6 | λ-HindⅢ | 10μL |
| 7 | 500bp DNA ladder | 10μL |
| 8 | - | - |
Inverse PCR
| Prime STAR MAX | 25μL |
| 10pmol/μL Primer F | 1μL |
| 10pmol/μL Primer R | 1μL |
| PCR反応液(2ng) | 1μL |
| dH2O | 22μL |
| Total | 22μL |
CrdS_Fw: AGTACGATCCGCTATTTTCCCG
CrdS_Rv: CAGACCAAGATTTCGCGAACTC
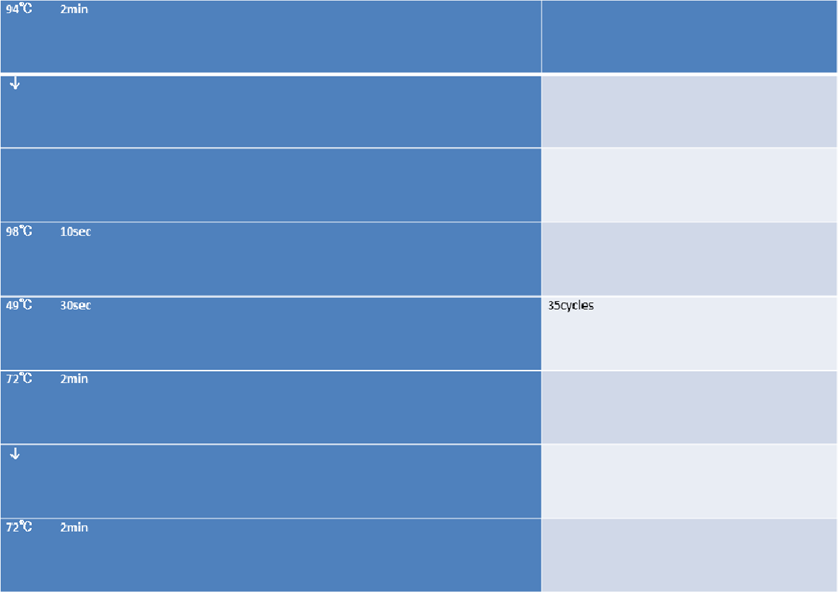
94℃ 2min
↓
98℃ 10sec
48℃ 30sec
72℃ 2min
↓
72℃ 2min
TA cloning of CrdS
| PCR product | 1μL |
| pMD20 | 2μL |
| Ligation kit ver.2 | 3μL |
16℃ 30min incubate
Ligation of CrdS/pMD20 and Transformation in JM109
| Competent cell | 100μL |
| DNA | 6μL |
↓Cells were stored on ice for 30min.
↓After 42℃ 30sec heat shock, cells were stored on ice for 2min.
↓Then cells were pre-cultured at 37℃ for 1hr, plated to Ampicillin plate.
Liquid culluture
CrdS/pMD20 21 samples at 37℃,for overnight
MiniPrep of CrdS/pMD20
Linear CrdS/pMD20 DNA :4995bp. This CrdS/pMD20 sample is cccDNA. So, probably picked up CrdS/pMD20.
| No | Name | volume |
| 1 | 500bp DNA ladder | 10μL |
| 2 | λ-HindⅢ | 10μL |
| 3 | CrdS No. | 12μL |
| 4 | CrdS No. | 12μL |
| 5 | CrdS No. | 12μL |
| 6 | CrdS No. | 12μL |
| 7 | CrdS No. | 12μL |
| 8 | CrdS No. | 12μL |
Restriction Enzyme of CrdS/pMD20
CrdS gene had produced clone from Agrobacterium tumefaciens C58, I confirmed whether it’s true or not. CrdS/pMD20 gene has 2 restriction enzyme sites,EcoRI.
| No | Name | volume |
| 1 | 500bp DNA ladder | 10μL |
| 2 | λ-HindⅢ | 10μL |
| 3 | CrdS No. | 12μL |
| 4 | CrdS No. | 12μL |
| 5 | CrdS No. | 12μL |
| 6 | CrdS No. | 12μL |
| 7 | CrdS No. | 12μL |
| 8 | CrdS No. | 12μL |
I comfirmed the direction of CrdS gene.
Sequence of CrdS/pMD20
PointMutation of CrdS
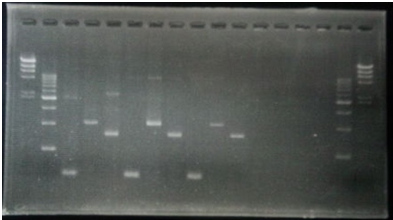
CrdS gene had Restriction Enzyme Site,EcoRI. We removed the EcoRI site.
| No | Name | volume |
| 1 | 500bp DNA ladder | 10μL |
| 2 | λ-HindⅢ | 10μL |
| 3 | CrdS Negative Front Fragment | 12μL |
| 4 | CrdS Negative Rear Fragment | 12μL |
| 5 | CrdS No.6 Front Fragment | 12μL |
| 6 | CrdS No.6 Middle Fragment | 12μL |
| 7 | CrdS No.6 Rear Fragment | 12μL |
| 8 | CrdS No.15 Front Fragment | 12μL |
| 9 | CrdS No.15 Middle Fragment | 12μL |
| 10 | CrdS No.15 Rear Fragment | 12μL |
| 11 | - | - |
| 12 | - | - |
| 13 | - | - |
| 14 | - | - |
| 15 | λ-HindⅢ | 10μL |
| 16 | 500bp DNA ladder | 10μL |
Front
Fw0 Primer:GGCCGCTTCTAGATGTATTTCAGTGC
Rv1 Primer:CGTTTCGAGGGAGAACTCCAGCG
Middle
Fw1Primer:CGCTGGAGTTCTCCCTCGAAACG
Rv2Primer:CAGCTCATCGGCCTGAAGCGCC
Rear
Fw2 Primer:GGCGCTTCAGGCCGATGAGCTG
Rv0 Primer:GGCGCTACTAGTATTATTATCACCCGAATG
| 5xPS Buffer | 10μL |
| dNTP Mixture | 4μL |
| 10pmol/µl Primer Fw1 or Fw2 | 1μL |
| 10pmol/µl Primer Rv2 or Rv1 | 1μL |
| Templete | 1μL |
| Prime STAR HS | 0.5μL |
| dH2O | 32.5μL |
| Total | 50μL |
 "
"