Team:British Columbia/Modeling
From 2013.igem.org
| Line 1: | Line 1: | ||
{{:Team:British_Columbia/Templates/MainHeader}} | {{:Team:British_Columbia/Templates/MainHeader}} | ||
| - | + | <html> | |
| + | <style> | ||
| + | .icon { width: 210px; margin 5px;} | ||
| + | #iconbar {padding: 0px 30px;} | ||
| + | #overviewpic{ | ||
| + | float: right; | ||
| + | width: 400px; | ||
| + | margin-top: 20px; | ||
| + | margin-left: 20px; | ||
| + | margin-right: 20px; | ||
| + | } | ||
| + | </style> | ||
| + | |||
| + | <img id="overviewpic" src="https://static.igem.org/mediawiki/2013/b/b2/UBC_Model-PhageControl.png"> | ||
| + | </html> | ||
| + | |||
| + | =Overview= | ||
We realized that strain balance could be modulated in independently immunized cultures using phages to apply dynamic selection pressure. An example of mixed culture fermentation is yogurt, where the quality of the final product requires optimal strain balance. To better understand the dynamics of this situation, we developed both mathematical and numerical models. | We realized that strain balance could be modulated in independently immunized cultures using phages to apply dynamic selection pressure. An example of mixed culture fermentation is yogurt, where the quality of the final product requires optimal strain balance. To better understand the dynamics of this situation, we developed both mathematical and numerical models. | ||
| - | == | + | Our objectives were to: |
| + | #Predict the growth of recombinant E. coli cultures under phage predation. | ||
| + | #Predict caffeine production based on initial starting number of viruses, (ie. multiplicity of infection; MOI). | ||
| + | |||
| + | ==Analytical Model== | ||
===Growth Kinetics under phage predation=== | ===Growth Kinetics under phage predation=== | ||
| Line 18: | Line 38: | ||
We numerically simulated the co-cultures of two flavouring product strains using Gro1. The model considers two strains of bacteria on a two-dimensional surface under phage predation. One of the strains is immune to infection, whereas the other is susceptible. | We numerically simulated the co-cultures of two flavouring product strains using Gro1. The model considers two strains of bacteria on a two-dimensional surface under phage predation. One of the strains is immune to infection, whereas the other is susceptible. | ||
| - | |||
| - | |||
| - | + | =Modeling Details= | |
| + | ===Assumptions=== | ||
| + | #Bacteria is grown in a batch culture and follows the Monod growth equation [1]. | ||
| + | #Bacteria with the CRISPR are nearly 100% immune to the specific phage infection. | ||
| + | #Bacteria without CRISPR system are completely susceptible to phage infection. | ||
| + | #Yield coefficients and phage attachment coefficients are constants. | ||
| + | #Bacterial phages do not decrease significantly in number during the course of an experiment. | ||
| + | #All considered bacterial phages are lytic. | ||
| - | + | == Analytical Model== | |
| - | + | ===Bacteria Growth=== | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
We begin with a populations balance for a given strain $X$, | We begin with a populations balance for a given strain $X$, | ||
\begin{align} | \begin{align} | ||
| Line 80: | Line 93: | ||
\end{align} | \end{align} | ||
| - | + | ===Substrate utilization=== | |
The bacteria will need to use a substrate, and the depletion of this substrate is proportional to the growth of the uninfected bacteria and the amount of bacteria. Although the infected bacteria do not multiply when infected, the infected cells may use up substrate to gain the energy needed to replicate the phage. Although it may be the case that the bacteria simply recycle intracellular material, a substrate utilization term ($\gamma$) for the infected cells is utilized (if the bacteria do in fact recycle intracellular material, it follows that this value will be 0). Moreover, when cells lyse, the cell materials can be metabolized by other bacteria and will add to the amount of nutrients available. Thus, the equation for substrate utilization rate is: | The bacteria will need to use a substrate, and the depletion of this substrate is proportional to the growth of the uninfected bacteria and the amount of bacteria. Although the infected bacteria do not multiply when infected, the infected cells may use up substrate to gain the energy needed to replicate the phage. Although it may be the case that the bacteria simply recycle intracellular material, a substrate utilization term ($\gamma$) for the infected cells is utilized (if the bacteria do in fact recycle intracellular material, it follows that this value will be 0). Moreover, when cells lyse, the cell materials can be metabolized by other bacteria and will add to the amount of nutrients available. Thus, the equation for substrate utilization rate is: | ||
| Line 88: | Line 101: | ||
Where $Y_u$ and $Y_i$ are yield constants and $\phi$ is the average amount of substrate released per infected cell during lysis. For clarity, $\delta$ is as described above. | Where $Y_u$ and $Y_i$ are yield constants and $\phi$ is the average amount of substrate released per infected cell during lysis. For clarity, $\delta$ is as described above. | ||
| - | + | ===Model Validation=== | |
Although, only limited amounts of growth curves have been able to be analyzed, the initial quantitative results appear promising and the qualitative results have been found to contain information whether or not the quantitative results align. The qualitative curve were especially good at predicting culture collapse (keep in mind that OD measurements will contain "bacterial bones" which will artificially inflate the concentration of bacteria after lysis). | Although, only limited amounts of growth curves have been able to be analyzed, the initial quantitative results appear promising and the qualitative results have been found to contain information whether or not the quantitative results align. The qualitative curve were especially good at predicting culture collapse (keep in mind that OD measurements will contain "bacterial bones" which will artificially inflate the concentration of bacteria after lysis). | ||
| Line 110: | Line 123: | ||
| - | + | ===Product Formation=== | |
For a given uninfected bacteria $X_u$ that can produce the product $P$, we will first expect the production rate to be first order equation: | For a given uninfected bacteria $X_u$ that can produce the product $P$, we will first expect the production rate to be first order equation: | ||
\begin{align} | \begin{align} | ||
| Line 117: | Line 130: | ||
Where $Y_P$ is the yield coefficient of the product. For infected bacteria we expect the product formation rate to be negligibly small. | Where $Y_P$ is the yield coefficient of the product. For infected bacteria we expect the product formation rate to be negligibly small. | ||
| - | + | ===Extending to Co-culture=== | |
The end goal of our project is to tune product formation by adding phage to a batch reactor at the same time as having one flavour in constant production. Thus, we must model a two culture system. Let our bacterial cultures be called $X$ and $Z$. Let Z be immunized to the phage with our CRISPR system. Our equations become: | The end goal of our project is to tune product formation by adding phage to a batch reactor at the same time as having one flavour in constant production. Thus, we must model a two culture system. Let our bacterial cultures be called $X$ and $Z$. Let Z be immunized to the phage with our CRISPR system. Our equations become: | ||
\begin{align} | \begin{align} | ||
| Line 149: | Line 162: | ||
| - | + | ==Numerical Model== | |
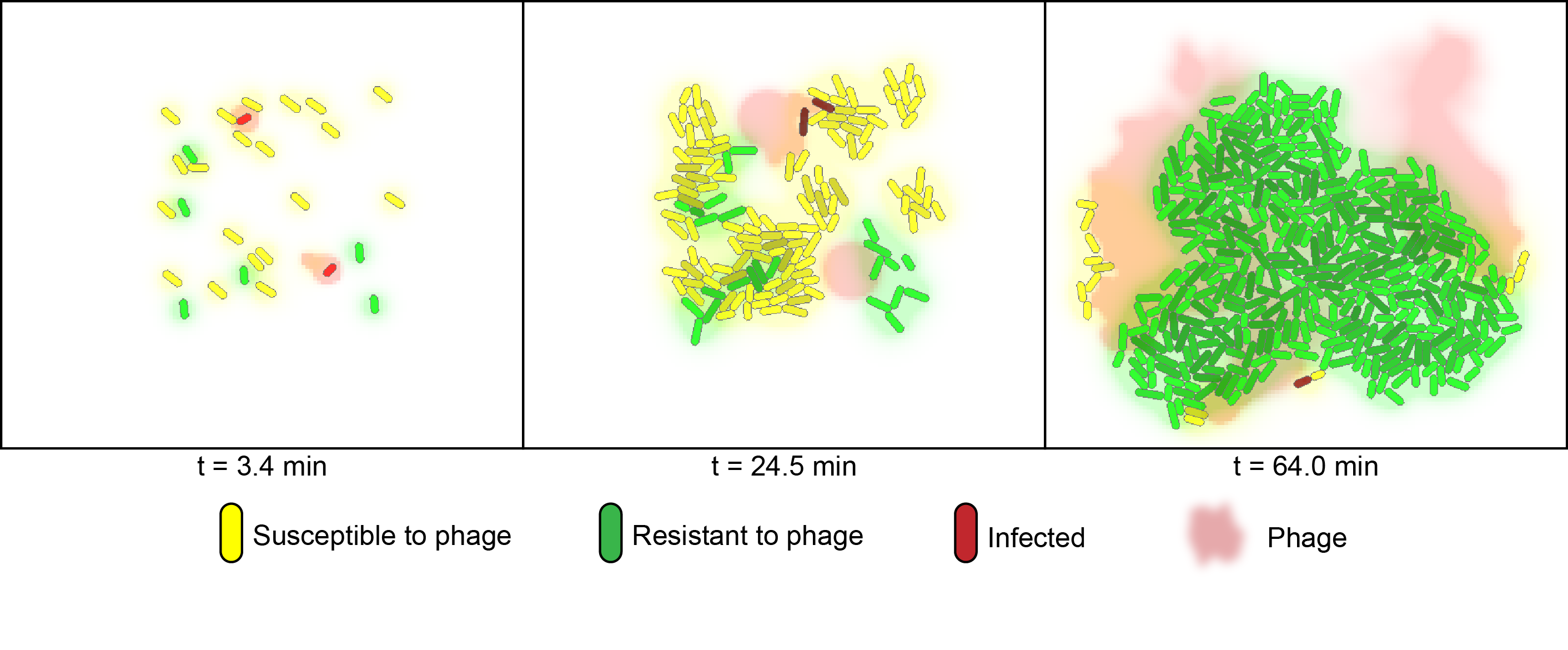
For the computational model of co-cultures we utilized a software developed by the Klavins Lab at the University of Washington [5]. The model considers two strains of bacteria on a two dimensional plane under attack of one virus. One strain has the CRISPR element and is immune to infection whereas the other is susceptible. In the simulation, susceptible bacteria entering regions with high phage concentrations are very likely to become infected and upon lysis increase the phage concentration of that region. | For the computational model of co-cultures we utilized a software developed by the Klavins Lab at the University of Washington [5]. The model considers two strains of bacteria on a two dimensional plane under attack of one virus. One strain has the CRISPR element and is immune to infection whereas the other is susceptible. In the simulation, susceptible bacteria entering regions with high phage concentrations are very likely to become infected and upon lysis increase the phage concentration of that region. | ||
| Line 169: | Line 182: | ||
| - | + | == References== | |
[1] Shuler, M.L. & Kargi, F. (2002). Bioprocess Engineering: Basic Concepts (2nd ed.). Prentice Hall, Upper Saddle River, NJ. | [1] Shuler, M.L. & Kargi, F. (2002). Bioprocess Engineering: Basic Concepts (2nd ed.). Prentice Hall, Upper Saddle River, NJ. | ||
Revision as of 02:56, 25 October 2013
iGEM Home



Overview
We realized that strain balance could be modulated in independently immunized cultures using phages to apply dynamic selection pressure. An example of mixed culture fermentation is yogurt, where the quality of the final product requires optimal strain balance. To better understand the dynamics of this situation, we developed both mathematical and numerical models.
Our objectives were to:
- Predict the growth of recombinant E. coli cultures under phage predation.
- Predict caffeine production based on initial starting number of viruses, (ie. multiplicity of infection; MOI).
Analytical Model
Growth Kinetics under phage predation
Monod growth kinetics were used to describe bacterial growth and the Poisson distribution was used to determine the infected populations based on the phage:bacteria ratio. We trained the model on a set of experimental growth curves and validated it against another set.
Compound production from a mixed culture
Our basic growth kinetic model was then extended to bacterial co-cultures. Yield terms were added to account for the production of two different flavours. Using this approach, we demonstrate that by using different virus inoculums, it is possible to achieve different final amounts of compounds from a mixed culture.
Numerical Model
We numerically simulated the co-cultures of two flavouring product strains using Gro1. The model considers two strains of bacteria on a two-dimensional surface under phage predation. One of the strains is immune to infection, whereas the other is susceptible.
Modeling Details
Assumptions
- Bacteria is grown in a batch culture and follows the Monod growth equation [1].
- Bacteria with the CRISPR are nearly 100% immune to the specific phage infection.
- Bacteria without CRISPR system are completely susceptible to phage infection.
- Yield coefficients and phage attachment coefficients are constants.
- Bacterial phages do not decrease significantly in number during the course of an experiment.
- All considered bacterial phages are lytic.
Analytical Model
Bacteria Growth
We begin with a populations balance for a given strain $X$, \begin{align} \frac{dX}{dt} = \frac{dX_i}{dt} + \frac{dX_u}{dt} \end{align} The infected bacteria is represented by $X_i$, uninfected bacteria by $X_u$ and $X = X_i + X_u$. We use the Monod equation to model the substrate limited bacterial growth. \begin{align} \mu_{X} = \mu_{max, X} \frac{S}{K_{S, X} + S} \end{align}
Where $\mu_{X}$ is the bacterial growth rate, $\mu_{max, X}$ is the maximum bacterial growth rate, $S$ is the growth limiting substrate and $K_s$ is the amount of substrate remaining when the growth rate is half of maximum. Due to environmental factors, some bacteria will die; the death rate ($k_d$) is assumed to be directly proportional to the bacteria population. A lag phase is modelled by multiplying a dampening term to the growth rate, namely $(1 - e^{-\alpha t})$, where $\alpha$ is defined as a proportionality constant. The slowing of growth from exponential to stationary is modelled with yet another dampening term to be multiplied to the growth: $exp\big[{-\big(\frac{X}{X_c}\big)^m\big]}$. This dampening term is related to the quorom sensing in bacteria, once the bacteria reach some characteristic concentration ($X_c$) the bacteria begin to slow down growth. Thus the uninfected cell growth is described by: \begin{align} \frac{dX_u}{dt} = \big( \mu_{X, u}e^{-\big(\frac{X}{X_c}\big)^m} - k_{d_{X,u}} \big) X_u\big(1 - e^{-\alpha t}\big) \end{align} For simplicity, in this model, $m$, $X_c$, and $\alpha$ are all to be empirically determined and assumed constant. In reality, each constant is dependent on many variables, for example: $\alpha(temperature, pressure, [substrate], environmental \ conditions, bacterial \ strain, more)$. Similarly for infected cells: \begin{align} \frac{dX_i}{dt} = \big( \mu_{X, i}e^{-\big(\frac{X}{X_c}\big)^m} - k_{d_{X,i}} \big) X_i\big(1 - e^{-\alpha t}\big) - \delta \end{align}
However, a major difference is that the decay rate for the infected cells is dependent on the latency time of the virus and thus we introduce $\delta$. The function $\delta$ is defined as follows, \begin{align} \delta = &N(\tau)X_i , \ \ during \ lysis\\ & 0, \ \ \ \ \ \ \ \ \ \ \ \ \ otherwise \end{align} Here $N(\tau)$ is a normal distribution over some time $\tau$, in other words, it is expected that the infected population will lyse according to a normal distribution over the domain $(-\tau, \tau)$.
Viral Growth
It is now necessary to consider the phage population $V$ as it directly affects the amount of bacteria that are infected at any given time. To describe the phage population it is necessary to consider how many phages are released per cell lysis, this number is referred to as the burst size $(\beta)$. \begin{align} V = V_0 + X_i \beta ^{\frac{t}{LT}} \end{align} Where $V_0$ is the initial phage populations and $LT$ is the latency time (they time between infection and lysis). However, since lab techniques are tricky to measure the exact phage at various time points as well as the exact amount of infected bacteria, it is desired to simplify the model. It is reasonable to use a stepwise function that describes the amount of free phage present. Also, since the burst size is large, on the order of $100$ [2], we see that the loss of current phage due to infection of cells is small (roughly 1%), thus to simplify computations we consider this to be negligible. With these deductions we arrive at the following: \begin{align} V = V_0 + E(X_i)\beta^{\big[\frac{t}{LT}\big]} \end{align} Where $E(X_i)$ is the expected infected population and $\big[\frac{t}{LT}\big]$ is the largest integer equal or less than $\frac{t}{LT}$. To find the expected infected population, the Poisson distribution is used based on the MOI. However, there is an efficiency of viral attachment $\epsilon \in (0, 1)$ (in fact the efficiency has been found to be a function of certain proteins) [3, 4]. \begin{align} MOI = \frac{V}{X} \end{align}
\begin{align} E(X_i) = \epsilon X(1 - e^{-MOI}) \end{align}
Substrate utilization
The bacteria will need to use a substrate, and the depletion of this substrate is proportional to the growth of the uninfected bacteria and the amount of bacteria. Although the infected bacteria do not multiply when infected, the infected cells may use up substrate to gain the energy needed to replicate the phage. Although it may be the case that the bacteria simply recycle intracellular material, a substrate utilization term ($\gamma$) for the infected cells is utilized (if the bacteria do in fact recycle intracellular material, it follows that this value will be 0). Moreover, when cells lyse, the cell materials can be metabolized by other bacteria and will add to the amount of nutrients available. Thus, the equation for substrate utilization rate is:
\begin{align} \frac{dS}{dt} = - \big[ \frac{\mu_u}{Y_u}X_u + \frac{\gamma}{Y_i}X_i \big] + \phi \delta \end{align} Where $Y_u$ and $Y_i$ are yield constants and $\phi$ is the average amount of substrate released per infected cell during lysis. For clarity, $\delta$ is as described above.
Model Validation
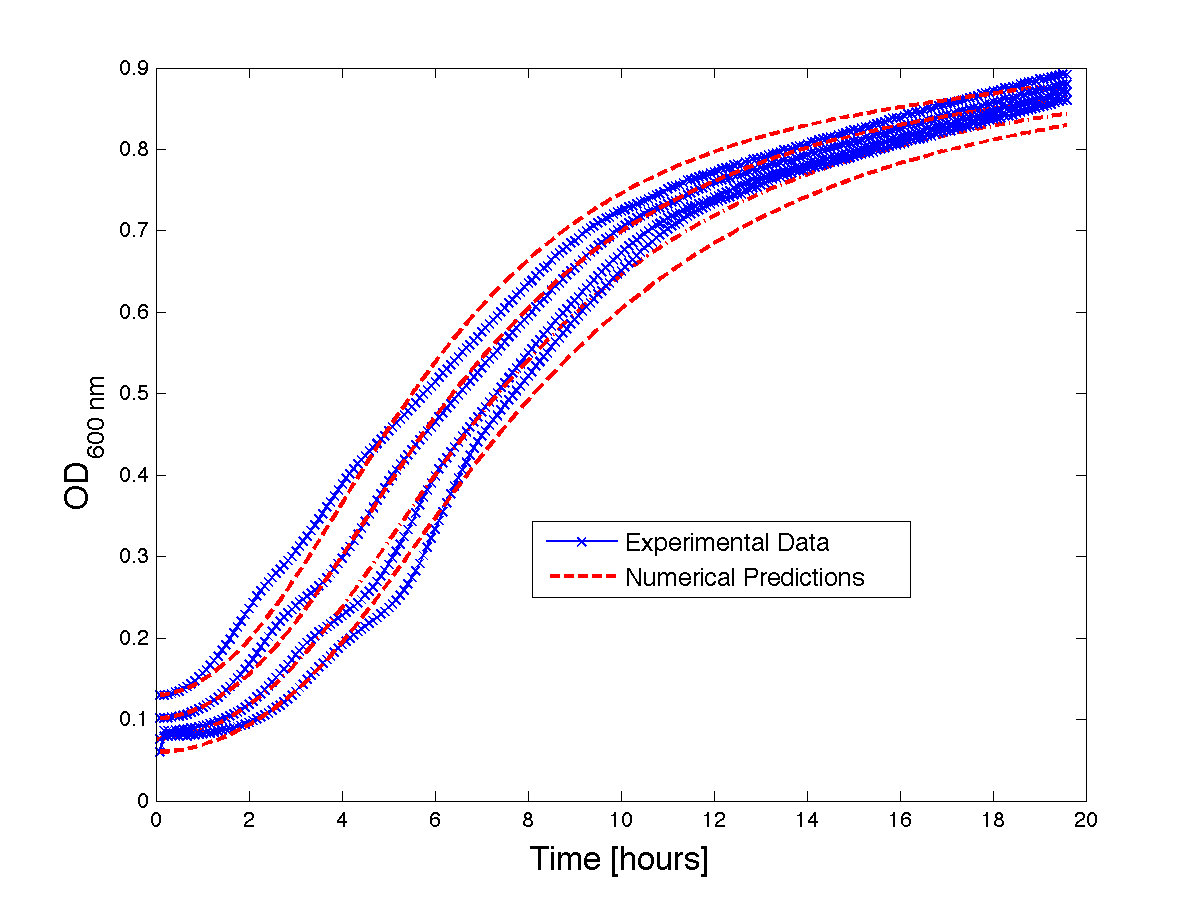
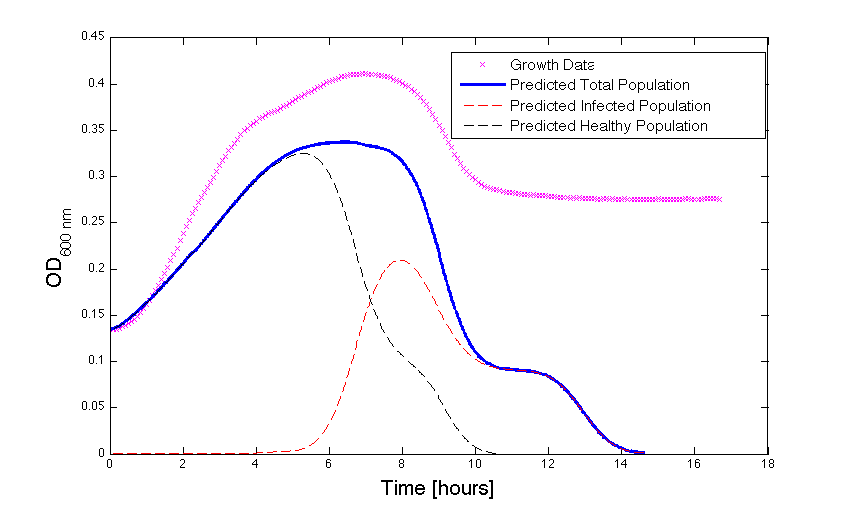
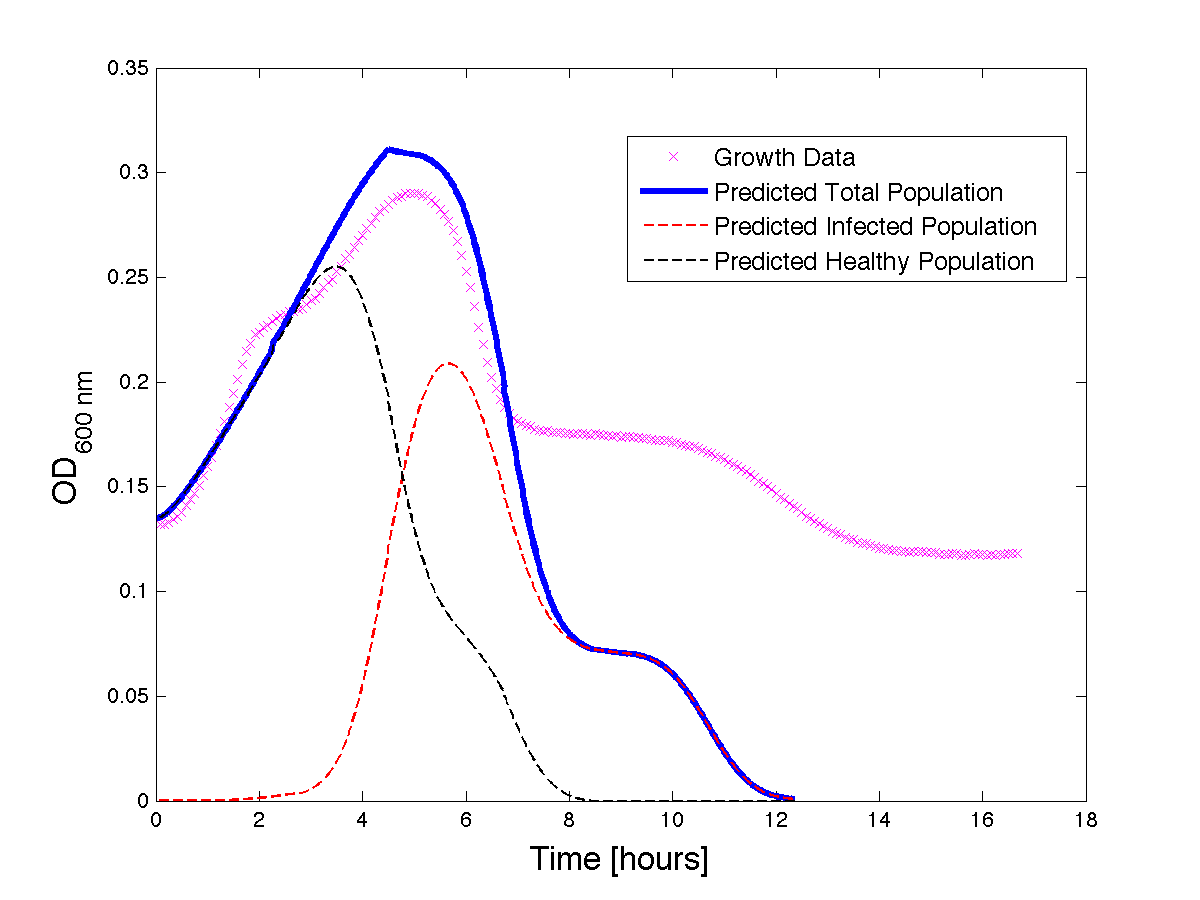
Although, only limited amounts of growth curves have been able to be analyzed, the initial quantitative results appear promising and the qualitative results have been found to contain information whether or not the quantitative results align. The qualitative curve were especially good at predicting culture collapse (keep in mind that OD measurements will contain "bacterial bones" which will artificially inflate the concentration of bacteria after lysis).
Figure 1 - Bacteria Growth curves from lag phase to approaching stationary phase. The model was optimized on another set of data and was tested for validity on this set.
Figure 2 - Bacteria Growth under phage predation. In models where the quantitative values differ, there is still qualitative information that can be extracted.
Figure 3 - A better fit for bacteria Growth under phage predation. Also included, the predicted infected and uninfected populations.
Product Formation
For a given uninfected bacteria $X_u$ that can produce the product $P$, we will first expect the production rate to be first order equation: \begin{align} \frac{d[P]}{dt} = \frac{\mu_{X_u}}{Y_P}X_u \end{align} Where $Y_P$ is the yield coefficient of the product. For infected bacteria we expect the product formation rate to be negligibly small.
Extending to Co-culture
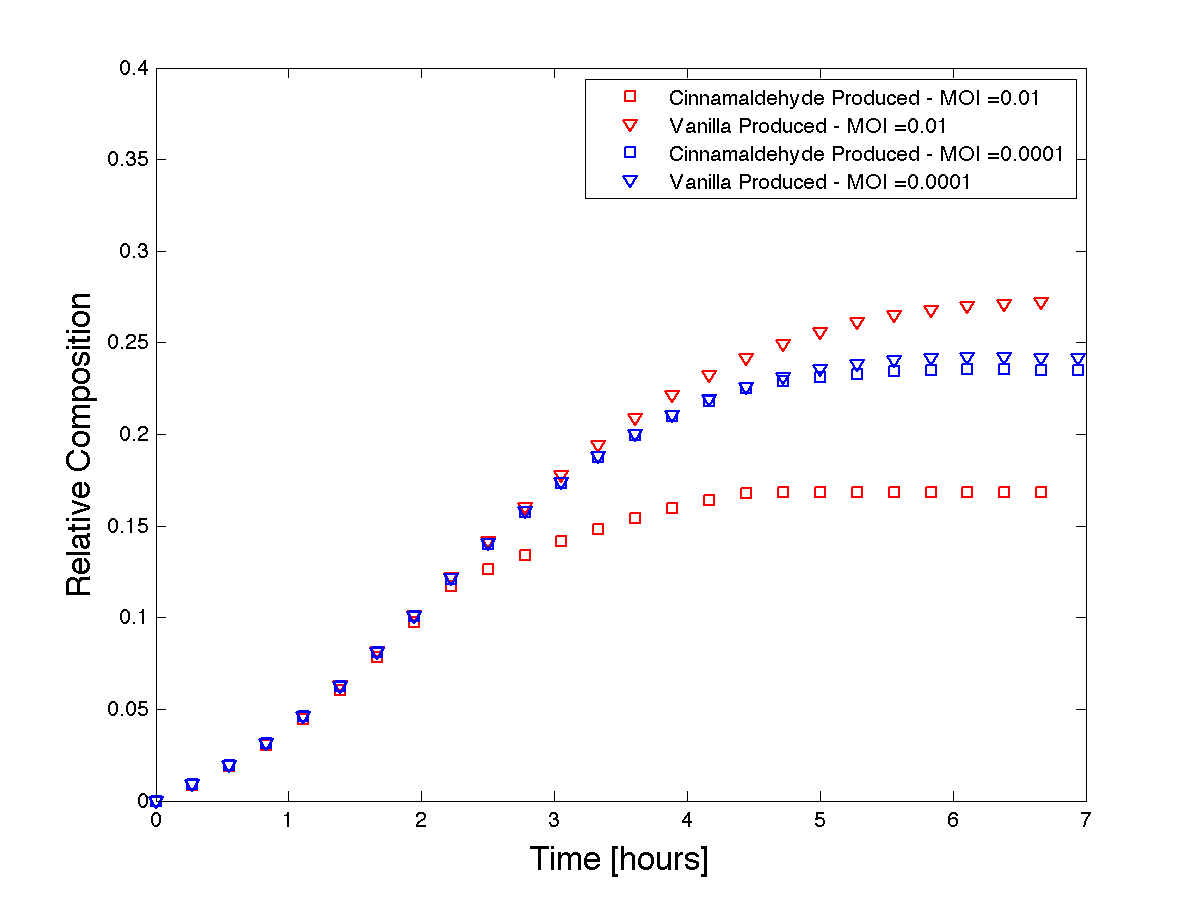
The end goal of our project is to tune product formation by adding phage to a batch reactor at the same time as having one flavour in constant production. Thus, we must model a two culture system. Let our bacterial cultures be called $X$ and $Z$. Let Z be immunized to the phage with our CRISPR system. Our equations become: \begin{align} &\frac{dX}{dt} = \frac{dX_i}{dt} + \frac{dX_u}{dt} \\ &\frac{dZ}{dt} = \frac{dZ_i}{dt} + \frac{dZ_u}{dt} \\ &\frac{dX_u}{dt} = \big( \mu_{X, u}e^{-\big(\frac{X}{X_c}\big)^m} - k_{d_{X,u}} \big) X_u\big(1 - e^{-\alpha t}\big) \\ &\frac{dZ_u}{dt} = \big( \mu_{Z, u}e^{-\big(\frac{Z}{Z_c}\big)^m} - k_{d_{Z,u}} \big) Z_u\big(1 - e^{-\alpha t}\big) \\ &\frac{dX_i}{dt} = \big( \mu_{X, i}e^{-\big(\frac{X}{X_c}\big)^m} - k_{d_{X,i}} \big) X_i\big(1 - e^{-\alpha t}\big) - \delta\\ &\frac{dZ_i}{dt} = 0\\ &\frac{dS}{dt} = - \big[ \frac{\mu_{X_u}}{Y_u}X_u + \frac{\gamma_X}{Y_i}X_i + \frac{\mu_Z}{Y_Z}Z\big] + \phi \delta \\ &\frac{dP_X}{dt} = \frac{\mu_X}{Y_{P_X}}X_u\\ &\frac{dP_Z}{dt} = \frac{\mu_Z}{Y_{P_Z}}Z_u \end{align} Subject to conditions: \begin{align} &X(0) = X_0\\ &Z(0) = Z_0\\ &Z_i(t) = 0\\ &S(0) = S_0\\ &S(t) = S_t\\ &P_X(0) = 0\\ &P_Z(0) = 0\\ &X = X_i + X_u\\ &X_i = E(X_i) = \epsilon X(1 - e^{-MOI})\\ &OD(t) = OD_Z(t) + OD_X(t) \end{align}
Figure 4 - Theoretical product formations based on co-culture dynamics. The thicker lines depict a higher MOI than the thinner lines.
Numerical Model
For the computational model of co-cultures we utilized a software developed by the Klavins Lab at the University of Washington [5]. The model considers two strains of bacteria on a two dimensional plane under attack of one virus. One strain has the CRISPR element and is immune to infection whereas the other is susceptible. In the simulation, susceptible bacteria entering regions with high phage concentrations are very likely to become infected and upon lysis increase the phage concentration of that region.
References
[1] Shuler, M.L. & Kargi, F. (2002). Bioprocess Engineering: Basic Concepts (2nd ed.). Prentice Hall, Upper Saddle River, NJ.
[2] Simon, E. H., Tessman, I. (1963). Thymidine-requiring Mutants of Phage T4. PNAS, 50, 526–532.
[3] Storms, Z. J., Arsenault, E., Sauvageau, D., & Cooper, D. G. (2010). Bacteriophage adsorption efficiency and its effect on amplification. Bioprocess and Biosystems Engineering, 33(7), 823–31.
[4] Storms, Z. J., Smith, L., Sauvageau, D., & Cooper, D. G. (2012). Modeling bacteriophage attachment using adsorption efficiency. Biochemical Engineering Journal, 64, 22–29.
[5] Klavins Lab, https://depts.washington.edu/soslab/gro/index.html
 "
"