Template:Kyoto/Notebook/Aug 29
From 2013.igem.org
(Difference between revisions)
(→Gel Extraction) |
(→Gel Extraction) |
||
| Line 195: | Line 195: | ||
[[File:Igku Aug29 Transformation1(N④-1).jpg]]<br> | [[File:Igku Aug29 Transformation1(N④-1).jpg]]<br> | ||
[[File:Igku Aug29 Transformation(N④-2).jpg]]<br> | [[File:Igku Aug29 Transformation(N④-2).jpg]]<br> | ||
| + | |||
| + | {| class="wikitable" | ||
| + | !Lane||Sample||Enzyme1||Enzyme2 | ||
| + | |- | ||
| + | |||100bp ladder||--||-- | ||
| + | |- | ||
| + | |1||8/29 pSB1C3||EcoRI||SpeI | ||
| + | |- | ||
| + | |2||8/29 pSB1C3||EcoRI||SpeI | ||
| + | |- | ||
| + | |3||8/29 pSB1C3||XbaI||PstI | ||
| + | |- | ||
| + | |4||8/29 pSB1C3||XbaI||PstI | ||
| + | |- | ||
| + | |5||8/29 spinach||EcoRI||SpeI | ||
| + | |- | ||
| + | |6||8/29 spinach||EcoRI||SpeI | ||
| + | |- | ||
| + | |7||8/29 tetRaptarmer12-1R(1)||EcoRI||SpeI | ||
| + | |- | ||
| + | |8||8/29 tetRaptarmer12-1R(1)||EcoRI||SpeI | ||
| + | |- | ||
| + | |||100bp ladder||--||-- | ||
| + | |} | ||
[[File:Igku Aug29 Transformation(N⑤).jpg]]<br> | [[File:Igku Aug29 Transformation(N⑤).jpg]]<br> | ||
[[File:Igku Aug29 Transformation()after]]<br> | [[File:Igku Aug29 Transformation()after]]<br> | ||
| + | </div> | ||
Revision as of 18:26, 25 September 2013
Contents |
Aug 29
Colony PCR
| Sample | base pair |
|---|---|
| 8/27 Pcon-pT181 antisense-(1) | 542 |
| 8/26 pT181 attenuator-DT-(1) | 738 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 30s | 30cycles |
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | 8/27 Pcon-pT181antisense-1 |
| 3 | 8/26 pT181attenuator-DT-1 |
| 4 | 100bp ladder |
Electrophoresis
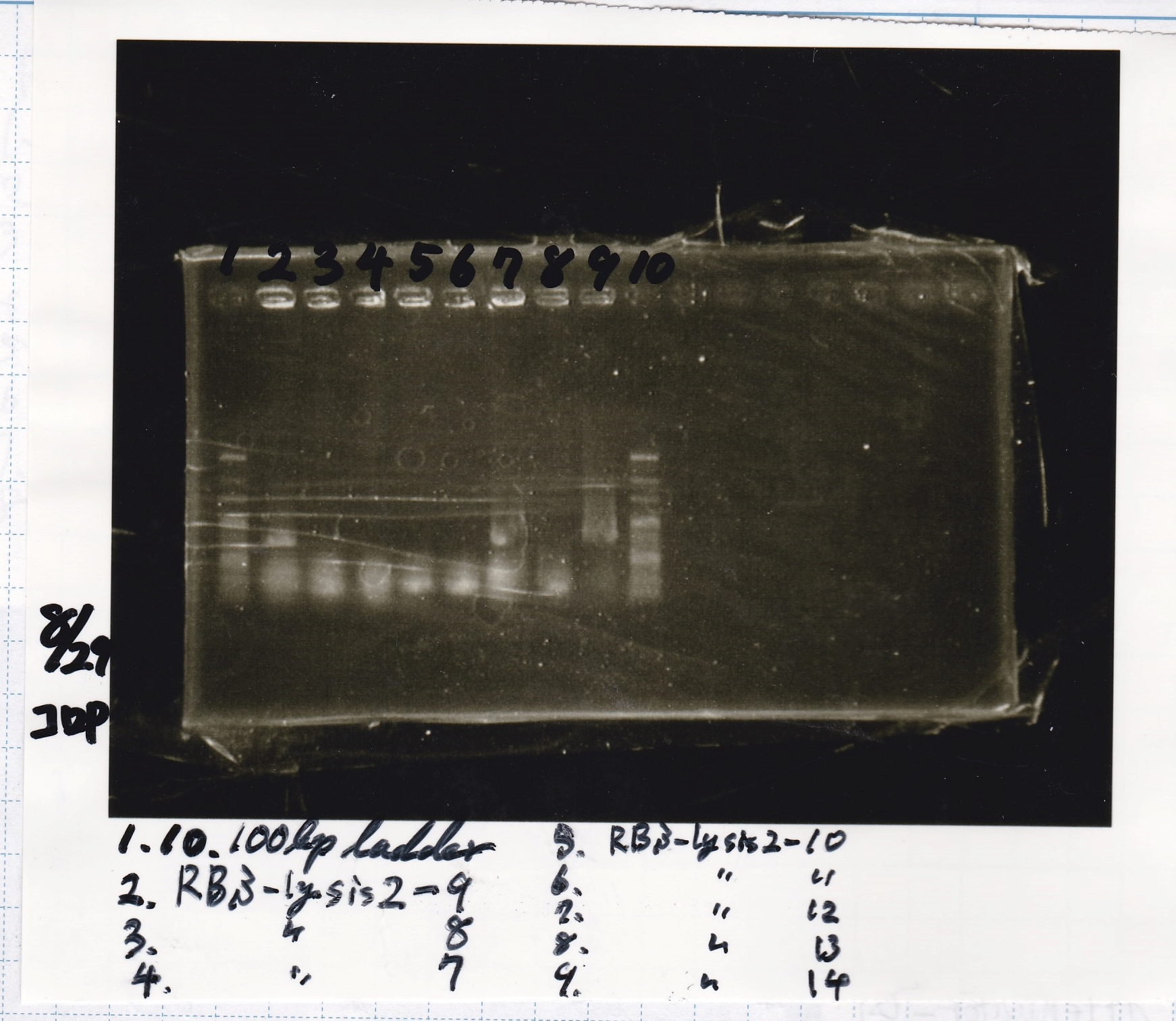
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | 8/21 RBS-lysis2-9(Colony PCR prodution) |
| 3 | 8/21 RBS-lysis2-8(Colony PCR prodution) |
| 4 | 8/21 RBS-lysis2-7(Colony PCR prodution) |
| 5 | 8/21 RBS-lysis2-10(Colony PCR prodution) |
| 6 | 8/21 RBS-lysis2-11(Colony PCR prodution) |
| 7 | 8/21 RBS-lysis2-12(Colony PCR prodution) |
| 8 | 8/21 RBS-lysis2-13(Colony PCR prodution) |
| 9 | 8/21 RBS-lysis2-14(Colony PCR prodution) |
Miniprep
| DNA | concentration [µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| Plac | 153.7 | 2.03 | 2.10 |
| Pλ-luxⅠ-(1) | 76.5 | 2.07 | 2.06 |
| Pλ-luxⅠ-(2) | 86.4 | 2.08 | 2.10 |
| DT | 166.2 | 1.65 | 2.12 |
| J23100 | 190.1 | 1.68 | 2.34 |
Liquid Culture
| Sample | medium |
|---|---|
| 8/21 RBS-Lysis2-9 | Plusgrow medium(+Amp) |
| 8/21 RBS-Lysis2-12 | Plusgrow medium(+Amp) |
| 8/21 RBS-Lysis2-14 | Plusgrow medium(+Amp) |
Restriction Enzyme Digestion
| 8/22 psB1C3-(2) | EcoRI | SpeI | XbaI | PstI | BufferB | BufferD | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 cuts | 8µL | 0.5µL | 0.5µL | 0µL | 0µL | 3µL | 0µL | 0.3µL | 17.7µL | 30µL |
| NC | 1µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0µL | 0.1µL | 7.9µL | 10µL |
| 2 cuts | 8µL | 0µL | 0µL | 0.5µL | 0.5µL | 0µL | 3µL | 0.3µL | 17.7µL | 30µL |
| NC | 1µL | 0µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0.1µL | 7.9µL | 10µL |
| 8/21 tRMA-spinach(1) | XbaI | SpeI | BufferB | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 4µL | 0.5µL | 0.5µL | 3µL | 0.3µL | 21.7µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 1µL | 0.1µL | 8.4µL | 10µL |
| 8/21 tetR aptamer12-1R(1) | EcoRI | SpeI | BufferB | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 3µL | 0.5µL | 0.5µL | 3µL | 0.3µL | 22.7µL | 30µL |
| NC | 0.4µL | 0µL | 0µL | 1µL | 0.1µL | 8.5µL | 10µL |
| 8/21 PT181 attenuator-(2) | EcoRI | SpeI | XbaI | PstI | BufferB | BufferD | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 cuts | 4µL | 0.5µL | 0.5µL | 0µL | 0µL | 3µL | 0µL | 0.3µL | 21.7µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0µL | 0.1µL | 8.4µL | 10µL |
| 2 cuts | 4µL | 0µL | 0µL | 0.5µL | 0.5µL | 0µL | 3µL | 0.3µL | 21.7µL | 30µL |
| NC | 0.5µL | 0µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0.1µL | 8.4µL | 10µL |
| 8/21 PT181 antisense-(2) | EcoRI | SpeI | XbaI | PstI | BufferB | BufferD | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 cuts | 10µL | 0.5µL | 0.5µL | 0µL | 0µL | 3µL | 0µL | 0.3µL | 15.7µL | 30µL |
| NC | 1µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0µL | 0.1µL | 7.9µL | 10µL |
| 2 cuts | 10µL | 0µL | 0µL | 0.5µL | 0.5µL | 0µL | 3µL | 0.3µL | 15.7µL | 30µL |
| NC | 1µL | 0µL | 0µL | 0µL | 0µL | 0µL | 1µL | 0.1µL | 7.9µL | 10µL |
- incubated 37°C 1hour
Transformation
| Name | Sample | Competent Cells | Total | Plate |
|---|---|---|---|---|
| 8/27 pT181 attenuator-DT | 2µL | 20µL | 22µL | CP |
| 8/27 RBS-lysis3-DT | 2µL | 20µL | 22µL | CP |
| 8/28 RBS-lysis3-DT | 2µL | 20µL | 22µL | CP |
| 8/28 Pcon-RBS-GFP-DT-Pcon-RBS-LuxR-DT | 2µL | 20µL | 22µL | Amp |
| 8/28 RBS-lysis1-DT | 2µL | 20µL | 22µL | CP |
| 8/28 Plux-RBS-GFP-DT | 2µL | 20µL | 22µL | CP |
| 8/28 Spinach-DT | 2µL | 20µL | 22µL | CP |
Gel Extraction
| Lane | DNA | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 100bp ladder | -- | -- | |
| 9 | 8/29 pT181attenuator(2) | EcoRI | SpeI |
| 10 | 8/29 pT181attenuator(2) | EcoRI | SpeI |
| 11 | 8/29 pT181attenuator(2) | XbaI | PstI |
| 12 | 8/29 pT181attenuator(2) | XbaI | PstI |
| 13 | pT181antisense(2) | EcoRI | SpeI |
| 14 | pT181antisense(2) | EcoRI | SpeI |
| 15 | pT181antisense(2) | XbaI | PstI |
| 16 | pT181antisense(2) | XbaI | PstI |
| 100bp ladder | -- | -- |
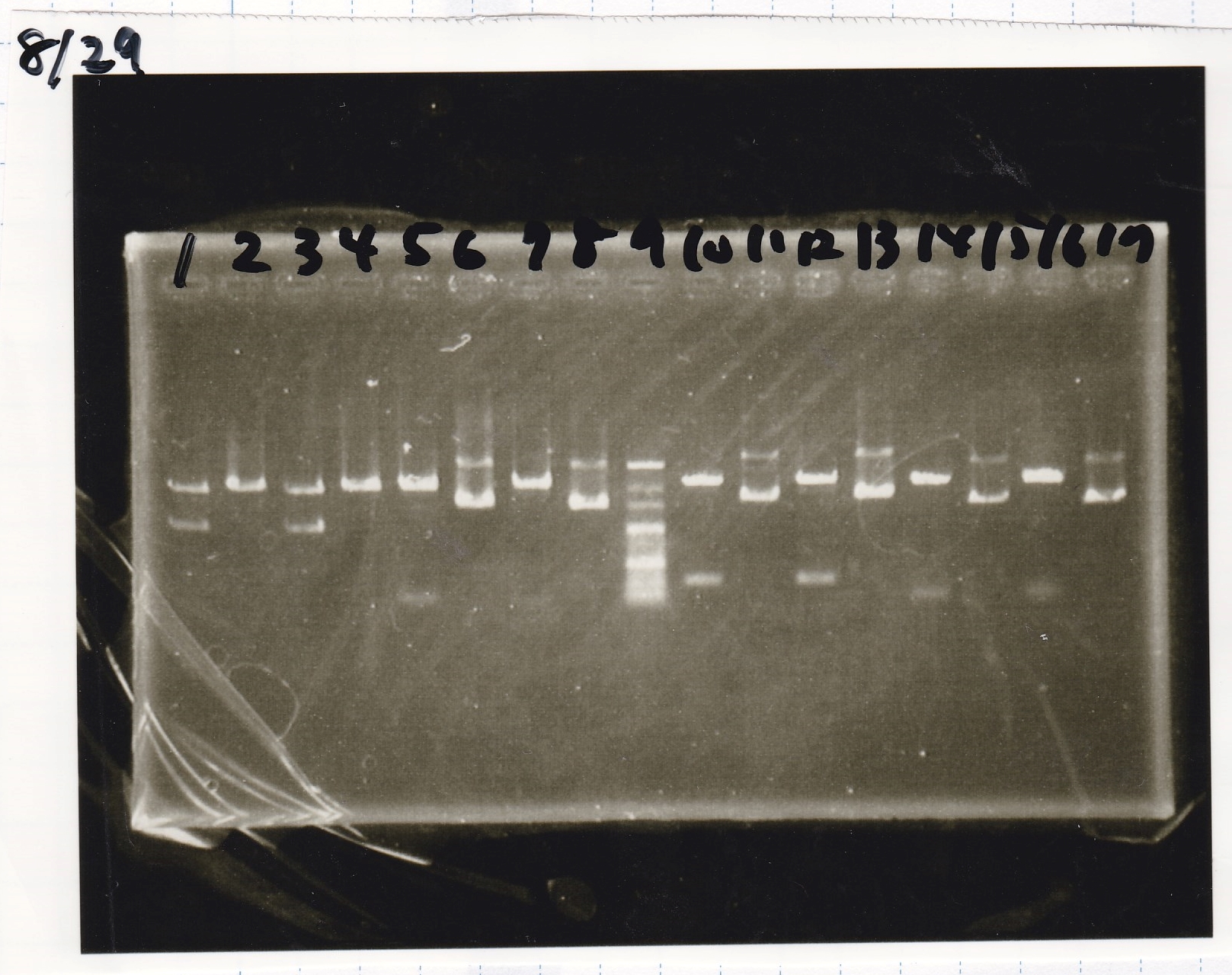
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 100bp ladder | -- | -- | |
| 1 | 8/29 pSB1C3 | EcoRI | SpeI |
| 2 | 8/29 pSB1C3 | EcoRI | SpeI |
| 3 | 8/29 pSB1C3 | XbaI | PstI |
| 4 | 8/29 pSB1C3 | XbaI | PstI |
| 5 | 8/29 spinach | EcoRI | SpeI |
| 6 | 8/29 spinach | EcoRI | SpeI |
| 7 | 8/29 tetRaptarmer12-1R(1) | EcoRI | SpeI |
| 8 | 8/29 tetRaptarmer12-1R(1) | EcoRI | SpeI |
| 100bp ladder | -- | -- |
 "
"