From 2013.igem.org
(Difference between revisions)
|
|
| Line 553: |
Line 553: |
| | |9||RBS-GFP-DT||--||-- | | |9||RBS-GFP-DT||--||-- |
| | |} | | |} |
| - | [[File:IgkuxxxxBa 1.jpg]]<br> | + | [[File:Igku Sep26 Electrophoresis(E11) 1.jpg]]<br> |
Revision as of 07:41, 27 September 2013
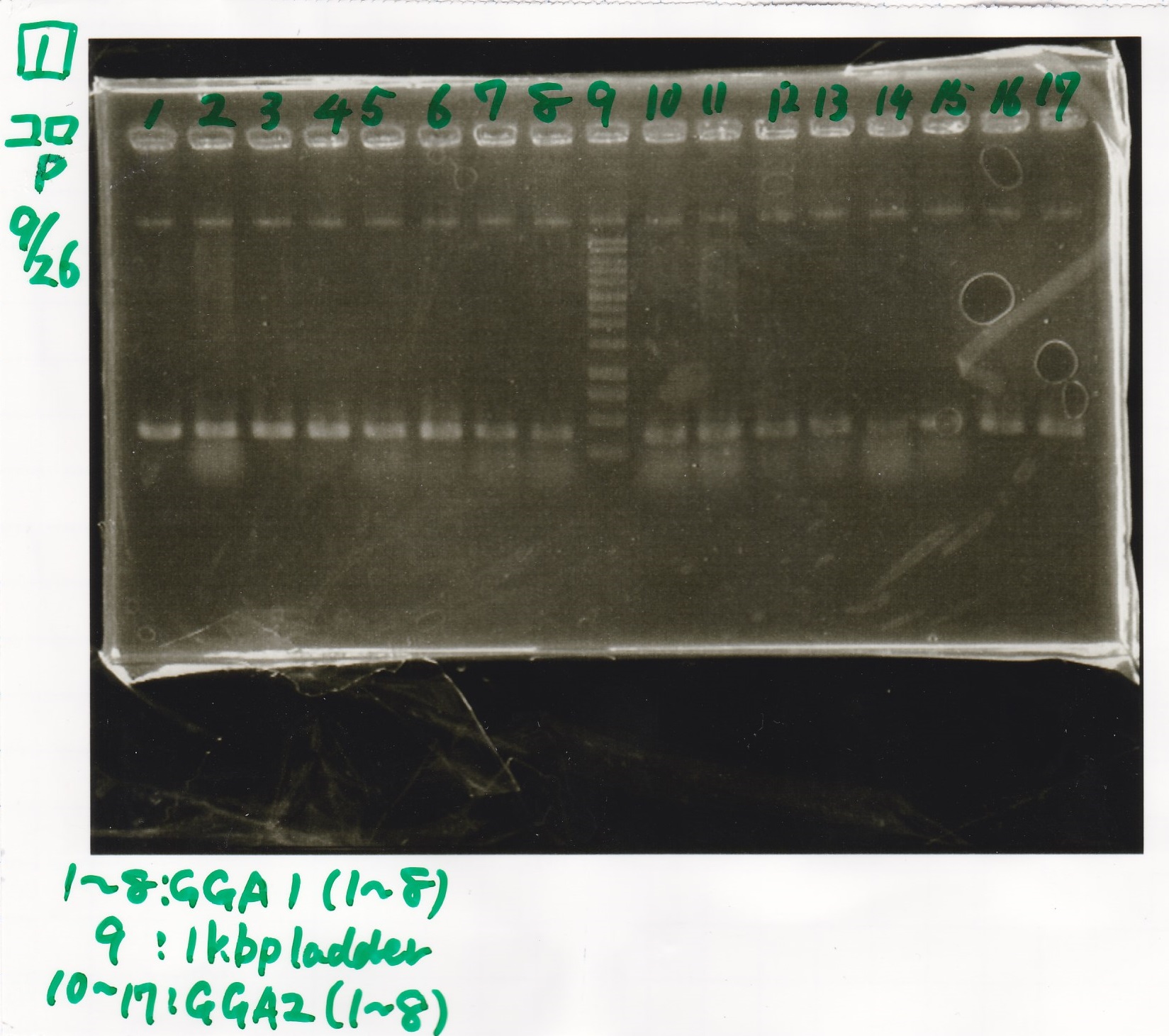
Sep 26
Electrophoresis
No name
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA1-1 | |
|
| 2 | GGA1-2 | |
|
| 3 | GGA1-3 | |
|
| 4 | GGA1-4 | |
|
| 5 | GGA1-5 | |
|
| 6 | GGA1-6 | |
|
| 7 | GGA1-7 | |
|
| 8 | GGA1-8 | |
|
| 9 | 1kbp ladder | |
|
| 10 | GGA2-1 | |
|
| 11 | GGA2-2 | |
|
| 12 | GGA2-3 | |
|
| 13 | GGA2-4 | |
|
| 14 | GGA2-5 | |
|
| 15 | GGA2-6 | |
|
| 16 | GGA2-7 | |
|
| 17 | GGA2-8 | |
|
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA3-1 | |
|
| 2 | GGA3-2 | |
|
| 3 | GGA3-3 | |
|
| 4 | 1kbp ladder | |
|
| 5 | GGA3-4 | |
|
| 6 | GGA3-5 | |
|
| 7 | GGA3-6 | |
|
| 8 | GGA3-7 | |
|
</div>
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA3-8 | |
|
| 2 | GGA4-1 | |
|
| 3 | GGA4-2 | |
|
| 4 | GGA4-3 | |
|
| 5 | 1kbp ladder | |
|
| 6 | GGA4-4 | |
|
| 7 | GGA4-5 | |
|
| 8 | GGA4-6 | |
|

</div>
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA4-7 | |
|
| 2 | GGA4-8 | |
|
| 3 | 1kbp ladder | |
|
| 4 | GGA7-1 | |
|
| 5 | GGA16-1 | |
|
| 6 | NC(RBS-GFP-DT) | |
|
</div>
Ligasion(Golden Gate Assenbly)
No name
| state | Vector | Inserter1 | Inserter2 | Inserter3 | Inserter4 | Inserter5 | (NEB)T4 ligase | (NEB)10*T4 ligase | MilliQ
|
| experiment | RBS-GFP-DT | 1.0 µL | Pcon-pT181 antisense(E-2A) | 0.6µL | DT(2-1) | 0.5µL | Pcon-pT181 attenuator(1-SA) | 2.1 µL | -- | -- | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | pSB1C3(EcoRI&SpeI) | 1.8 µL | Pcon-pT181 antisense(E-2A) | 0.9 µL | DT(2-1) | 0.5 µL | Pcon-GFP-DT(1-SA) | 1.5 µL | -- | -- | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | RBS-GFP-DT | 1.0 µL | Ptet(E-0) | 0.7µL | Pcon pT181 attenuator(0-1A) | 0.3µL | tet aptamer(1-3) | 0.3 µL | Pcon-tetR-DT(3-2A) | 1.1 µL | Ptet(2-5) | 0.3 µL | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | RBS-GFP-DT | 1.0 µL | Ptet(E-0) | 0.7 µL | Pcon-pT181 attenuator-DT(0-1A) | 1.0 µL | Pcon-tetR-DT(1-2A) | 1.0 µL | Ptet(2-5) | 0.3µL | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | RBS-GFP-DT | 1.0 µL | Pcon-pT181 attenuator-tetR-DT(E-1A) | 1.0 µL | Pcon antisense-spinach-DT(1-2A) | 0.7µL | Pcon-pT 181 attenuator(2-SA) | 2.1 µL | -- | -- | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | RBS-GFP-DT | 1.0 µL | Pcon antisense(E-1A) | 0.5 µL | spinach-DT(1-2) | 1.0 µL | Pcon-GFP-DT(1-2) | 0.8 µL | -- | -- | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | pSB1C3(EcoRI&SpeI) | 1.8 µL | Pcon-pT181 antisense(E-1A) | 0.8µL | spinach-DT(1-2) | 1.0µL | Pcon-GFP-DT(2-SA) | 0.8 µL | -- | -- | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
| experiment | pSB1C3(EcoRI&SpeI) | 1.8 µL | Pcon-tet aptamer-DT(E-1A) | 3.0 µL | Ptet-pT181 antisense(1-3) | 1.5 µL | spinach-DT(3-2) | 1.5 µL | Pcon-GFP-DT(2-5A) | 0.8 µL | -- | -- | 0.5 µL | 0.5 µL | up to 5 µL
|
Liquid Culture
No name
| Sample | medium
|
| Pcon-GFP-DT | Amp
|
| RBS-GFP-DT | CP
|
| Pcon-pT181 attenuator-DT | Amp
|
| Pcon-tet aptamer-DT | Amp
|
| Pcon-spinach-DT | Amp
|
| Pcon-pT181 antisense-spinach-DT | Amp
|
| spinach-DT | CP
|
BSAI Digestion
No name
| | DNA | MilliQ | BSAI-HF | 10*cut smart | total
|
| Pcon | 7 µL | 10.7 µL | 0.3 µL | 2 µL | 20 µL
|
| RBS-luxR-DT | 10 µL | 7 µL | 0.3 µL | 2 µL | 20 µL
|
Colony PCR
Tatsui
| Sample | base pair
|
| GGA-1 (9~16) | 3177
|
| GGA-2 (9~16) | 3189
|
| GGA-3 (9~21) | 2989
|
| GGA-4 (9~16) | 2939
|
| GGA-6 (1) | 3359
|
| GGA-16(2~4) | 2939
|
| PreDenature | Denature | Annealing | Extension | cycle
|
| 94°C | 94°C | 55°C | 68°C | --
|
| 5min | 30s | 30s | 3min30s | 30cycles
|
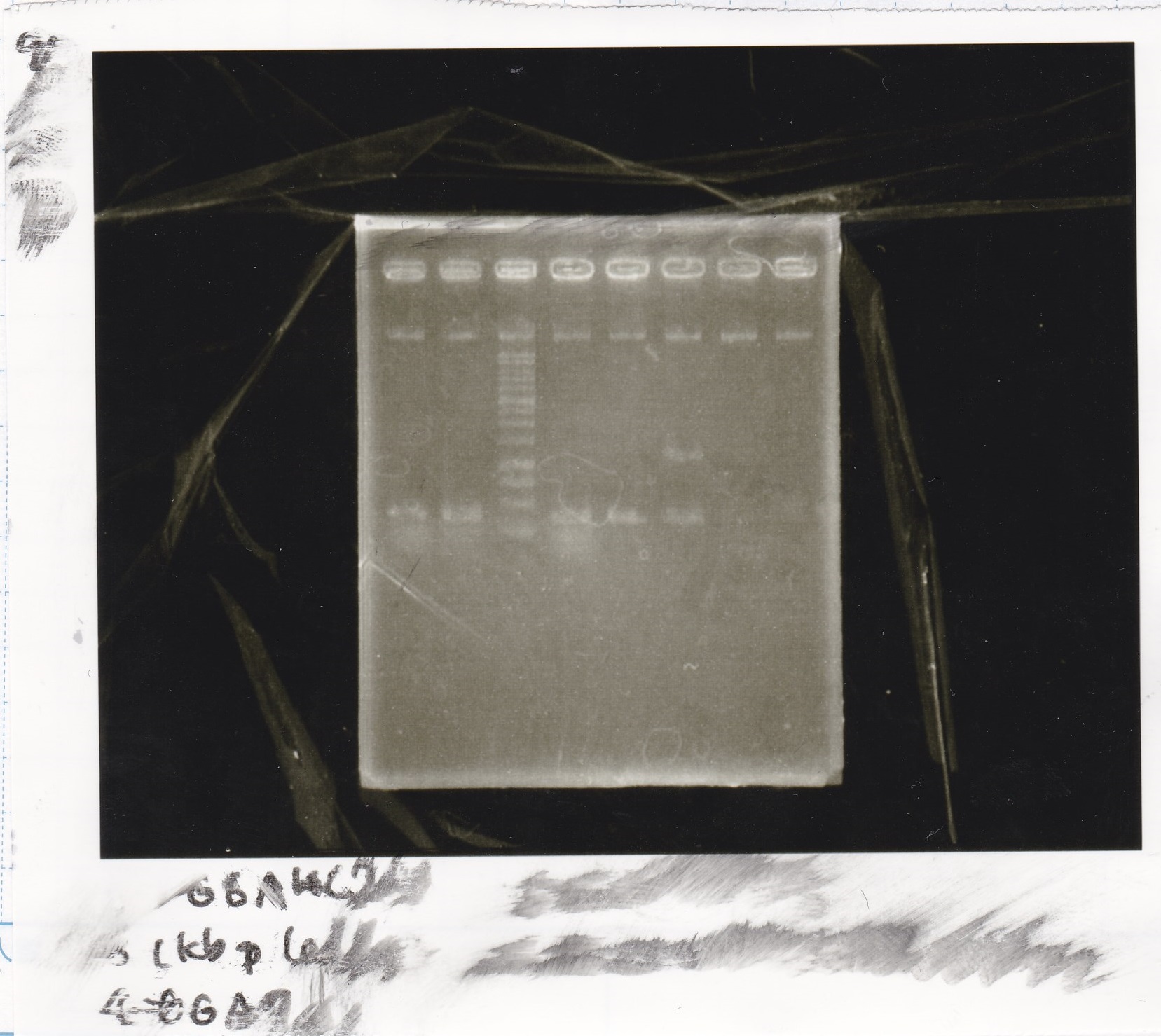
Electrophoresis
No name
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA-1 (9) | -- | --
|
| 2 | GGA-1 (10) | -- | --
|
| 3 | GGA-1 (11) | -- | --
|
| 4 | GGA-1 (12) | -- | --
|
| 5 | GGA-1 (13) | -- | --
|
| 6 | GGA-1 (14) | -- | --
|
| 7 | GGA-1 (15) | -- | --
|
| 8 | GGA-1 (16) | -- | --
|
| 9 | 1kbp ladder | -- | --
|
| 10 | GGA-2 (9) | -- | --
|
| 11 | GGA-2 (10) | -- | --
|
| 12 | GGA-2 (11) | -- | --
|
| 13 | GGA-2 (12) | -- | --
|
| 14 | GGA-2 (13) | -- | --
|
| 15 | GGA-2 (14) | -- | --
|
| 16 | GGA-2 (15) | -- | --
|
| 17 | GGA-2 (16) | -- | --
|

| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA-4 (9) | -- | --
|
| 2 | GGA-4 (10) | -- | --
|
| 3 | GGA-4 (11) | -- | --
|
| 4 | GGA-4 (12) | -- | --
|
| 5 | GGA-4 (13) | -- | --
|
| 6 | GGA-4 (14) | -- | --
|
| 7 | GGA-4 (15) | -- | --
|
| 8 | GGA-4 (16) | -- | --
|
| 9 | 1kbp ladder | -- | --
|
| 10 | GGA-6 (1) | -- | --
|
| 11 | GGA-16 (2) | -- | --
|
| 12 | GGA-16 (3) | -- | --
|
| 13 | GGA-16 (4) | -- | --
|

</div>
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA-3 (9) | -- | --
|
| 2 | GGA-3 (10) | -- | --
|
| 3 | GGA-3 (11) | -- | --
|
| 4 | GGA-3 (12) | -- | --
|
| 5 | 1kbp ladder | -- | --
|
| 6 | GGA-3 (13) | -- | --
|
| 7 | GGA-3 (15) | -- | --
|
| 8 | GGA-3 (14) | -- | --
|
</div>
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | GGA-3 (17) | -- | --
|
| 2 | GGA-3 (18) | -- | --
|
| 3 | GGA-3 (19) | -- | --
|
| 4 | 1kbp ladder | -- | --
|
| 5 | GGA-3 (20) | -- | --
|
| 6 | GGA-3 (21) | -- | --
|
| 7 | GGA-3 (16) | -- | --
|

</div>
Electrophoresis
No name
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | RBS-GFP-DT(RNA) | -- | --
|
| 2 | 1kbp ladder | -- | --
|
| 3 | Pcon-RBS-GFP-DT(RNA) | -- | --
|

Liquid Culture
Hirano
| Sample | medium
|
| Pcon-GFP-DT | --
|
| RBS-GFP-DT | --
|
| Pcon-tet aptamer-DT | --
|
| Pcon-spinach-DT | --
|
| Pcon-pT181 antisense-spinach-DT | --
|
| spinach-DT | --
|
Colony PCR
Hirano
| Sample | base pair
|
| 9/25 Pcon-pH181 attenuator+RBS-GFP-DT-1 | --
|
| 9/25 Pcon-pH181 attenuator+RBS-GFP-DT-2 | --
|
| 9/25 Pcon-pH181 attenuator+RBS-GFP-DT-3 | --
|
| 9/25 Pcon-pH181 attenuator+RBS-GFP-DT-4 | --
|
| 9/24 Ptet-RBS-GFP-DT-1 | --
|
| PreDenature | Denature | Annealing | Extension | cycle
|
| 94°C | 94°C | 55°C | 68°C | --
|
| 5min | 30s | 30s | 1min40s | 30cycles
|
Ligasion(Golden Gate Assenbly)
Hirano
| state | Vector | Inserter1 | Inserter2 | Inserter3 | Inserter4 | Inserter5
|
| experiment | GGA2 | Pcon-attenuator(E-1A) | Pcon-tetR-DT(1-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI) |
|
| experiment | GGA3 | Pcon-tetap-DT(E-1A) | Pcon-tetR-DT(1-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI) |
|
| experiment | GGA4 | Pcon-spi-DT(E-1A) | Pcon-tetR-DT(1-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI) |
|
| experiment | GGA5 | Pcon-antisense(E-1A) | DT(1-3) | Pcon-tetR-DT(3-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI)
|
| experiment | GGA6 | Pcon-attenuator(E-1A) | tetap-DT(1-3) | Pcon-tetR-DT(3-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI)
|
| experiment | GGA7 | Pcon-tetR-DT(E-2A) | Ptet(2-S) | RBS-GFP-DT(EcoRI&XbaI) | |
|
| PreDenature | Denature | Annealing | Extension | cycle
|
| 37°C | 16°C | 50°C | 80°C | --
|
| 3min | 4min | 5min | 5min | 25cycles
|
Liquid Culture
Colony PCR
Plating
| Sample | Use plate
|
| 9/26 GGA2 | LB(CP)
|
| 9/26 GGA3 | LB(CP)
|
| 9/26 GGA4 | LB(CP)
|
| 9/26 GGA5 | LB(CP)
|
| 9/26 GGA6 | LB(CP)
|
| 9/26 GGA7 | LB(CP)
|
| 9/26 GGA11 | LB(CP)
|
| 9/26 GGA312 | LB(CP)
|
| 9/26 GGA13 | LB(CP)
|
| 9/26 GGA14 | LB(CP)
|
| 9/26 GGA15 | LB(CP)
|
| 9/26 GGA20 | LB(CP)
|
| 9/26 GGA23 | LB(CP)
|
| 9/26 GGA24 | LB(CP)
|
| 9/26 Ptet+RBS-GFP-DT | LB(CP)
|
</div>
incubate 37°C
Restriction Enzyme Digestion
Kojima
| 9/24 pSB1C3 | EcoRI | SpeI | buffer | MilliQ | total
|
| 2cuts | 11µL | 1µL | 1µL | 3µL | 14µL | 30µL
|
| NC | 0.5µL | 0µL | 0µL | 1µL | 8.5µL | 10µL
|
| 9/24 Pcon-pT181attenuator-aptamer12-1R-DT | EcoRI | SpeI | buffer | MilliQ | total
|
| 2cuts | 8.7µL | 1µL | 1µL | 3µL | 16.3µL | 30µL
|
| NC | 0.4µL | 0µL | 0µL | 1µL | 8.6µL | 10µL
|
| 9/24 pT181attenuator | EcoRI | SpeI | buffer | MilliQ | total
|
| 2cuts | 3.1µL | 1µL | 1µL | 3µL | 21.9µL | 30µL
|
| NC | 0.2µL | 0µL | 0µL | 1µL | 8.8µL | 10µL
|
| 8/17 RBS-GFP-DT | EcoRI | BSA | buffer | MilliQ | total
|
| 1cut | 16.4µL | 1µL | 3µL | 3µL | 6.6µL | 30µL
|
| NC | 0.4µL | 0µL | 1µL | 1µL | 7.6µL | 10µL
|
| 9/17 pSB4K5 | EcoRI | PstI | BSA | buffer | MilliQ | total
|
| 2cuts | 8.5µL | 1µL | 1µL | 3µL | 3µL | 13.5µL | 30µL
|
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL
|
| 9/14 pSB4K5 | EcoRI | PstI | BSA | buffer | MilliQ | total
|
| 2cuts | 7.3µL | 1µL | 1µL | 3µL | 3µL | 14.7µL | 30µL
|
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL
|
| 9/22 Pcon-pT181attenuator-DT | EcoRI | SpeI | buffer | MilliQ | total
|
| 2cuts | 5.4µL | 1µL | 1µL | 3µL | 19.6µL | 30µL
|
| NC | 0.3µL | 0µL | 0µL | 1µL | 8.7µL | 10µL
|
| 8/7 RBS-GFP-DT 1 | XbaI | PstI | BSA | buffer | MilliQ | total
|
| 2cuts | 8.1µL | 1µL | 1µL | 3µL | 3µL | 13.9µL | 30µL
|
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL
|
| 9/26 RBS-GFP-DT 1 | XbaI | BSA | buffer | MilliQ | total
|
| 1cut | 8.1µL | 1µL | 3µL | 3µL | 14.9µL | 30µL
|
| 9/16 Pλ-luxI | EcoRI | PstI | BSA | buffer | MilliQ | total
|
| 2cuts | 14.7µL | 1µL | 1µL | 3µL | 3µL | 7.3µL | 30µL
|
| NC | 0.7µL | 0µL | 0µL | 1µL | 1µL | 7.3µL | 10µL
|
| 9/21 Ptet | EcoRI | SpeI | buffer | MilliQ | total
|
| 2cuts | 14.2µL | 1µL | 1µL | 3µL | 10.8µL | 30µL
|
| NC | 0.7µL | 0µL | 0µL | 1µL | 8.3µL | 10µL
|
| 9/24 Pcon-pT181attenuator-aptamer12-1R-DT | XbaI | PstI | BSA | buffer | MilliQ | total
|
| 2cuts | 8.7µL | 1µL | 1µL | 3µL | 3µL | 13.3µL | 30µL
|
| NC | 0.4µL | 0µL | 0µL | 1µL | 1µL | 7.6µL | 10µL
|
Electrophoresis
Kojima
| Lane | Sample | Enzyme1 | Enzyme2
|
| 1 | pSB1C3 | EcoRI | SpeI
|
| 2 | pSB1C3 | -- | --
|
| 3 | Pcon-pT181attenuator-aptamer12-1R-DT | EcoRI | SpeI
|
| 4 | Pcon-pT181attenuator-aptamer12-1R-DT | -- | --
|
| 5 | 1kb ladder | -- | --
|
| 6 | pT181attenuator | EcoRI | SpeI
|
| 7 | pT181attenuator | -- | --
|
| 8 | RBS-GFP-DT | EcoRI | --
|
| 9 | RBS-GFP-DT | -- | --
|

 "
"







