Team:Kyoto/projectRNA
From 2013.igem.org
(→Oscillation) |
(→Reporter) |
||
| Line 54: | Line 54: | ||
<div id="repressiontab"> | <div id="repressiontab"> | ||
| - | === | + | ===Repressor=== |
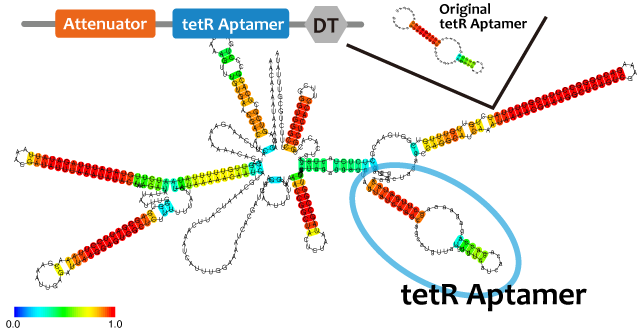
| - | + | We took up TetR aptamer as an example of functional RNA which activates a transcription. TetR aptamer induces tetracycline promoter (Ptet) by binding to tetracycline repressor (TetR), which represses Ptet. When TetR aptamer binds to TetR, induces conformational change of TetR. As a result, TetR cannot come to bind to tetracycline operator (tetO). We ordered MBL=IDT gene synthesis of pT181 attenuator region DNA, antisense DNA and TetR aptamer with prefix and suffix. We transferred these parts to pSB1C3 and constructed device for antisense and attenuator assay (Fig. ). | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | We | + | |
| - | + | ||
| - | + | ||
</div> | </div> | ||
<div id="fusiontab"> | <div id="fusiontab"> | ||
Revision as of 00:06, 28 September 2013
count down
Contents |
RNA Oscillator
Introduction
Motivation
細胞間の相互作用のシミュレーションは、細胞内の条件だけでなく位置関係などさらに複雑な条件を考慮しなければならないため、かなり複雑で手におえない そこで1細胞内でのシミュレーションにおいて、dryとwetの解離の対策を考えてみた。合成生物学のある研究(a fast robustなんとかかんとか)[citation*]では、オシレーションの形成をdryでもwetでも確認している。この実験系では、細胞分裂という、オシレーションの形成に強く影響を与えるであろう要素をゼロに近似できるようなロバストな回路がコンストラクトされている。このことから、dryでwetの系を考慮しきるのが難しいことの1つの解決法として、適切な近似計算によって複雑性を無視できるロバストネスを持った回路を構築することが考えられる。しかしそのような構成要素はそう多くなく、適応する系は限られている。そこで私たちは押しレーションの構築を他のアプローチによって実現し、dryに歩み寄るwetの系を考えてみた。私たちはrnaを構成要素とする押しレーションの構築をゴールとした。
Oscillation
その回路として、ncRNA-mRNAの相互作用を抑制機構に持ち、RNA アプタマーとtetR Proteinを促進機構に用いた以下の様な回路を提案する。アウトプット機構としては、Spinachを想定する。
This circuit oscillates in the following way: First, tet promoter is repressed by tetR at the downstream of constitutive promotor. Then, the oscillator is turned on by IPTG. IPTG activates Plac and tetRaptamer, Spinarch, and Antisense at the downstream of Ptet which are transcribed. Because tetRaptamer activates tet promotor, positive feedback occurs and more and more tetRaptamer, Spinarch, and Antisense are accumulated. Then, this circuit gets fluorescence. After Antisense is accumulated to some extent, tetRaptamer, at the downstream of Atteruator region, is repressed. Then, because new tetRaptamer is not created, the amount of tetRaptamer decreases quickly. So, tet promotor is repressed by tetR protein and the amount of Antisense and Spinarch falls, too. Then, this circuit loses fluorescence. After the amount of Antisense decreases sufficiently, this circuit recovers first condition. Through this cycle, this circuit acts as an oscillator.
促進、抑制の各機構と、この回路のアウトプットとしてのSpinachの機能は以下に述べる。
Repressor
We took up non-coding RNA (ncRNA) complementarily binding mRNA as an example of functional RNA which repress transcription. ncRNA in pT181 plasmid (pT181 attenuator) controls the fate of transcriptional elongation in response to an input by complementary antisense RNA. Attenuator region, which lies in 5' untranslated region of a transcript, folds into two different RNA structure. By an interaction with complementary antisense RNA, attenuator region forms Rho-independent terminator and the transcription of the downstream is stopped. Without antisense RNA, attenuator region RNA folds into an alternative structure which allow transcription of the downstream (Novick, 1989). The uniqueness of this mechanism is that it is constructed with only RNA without other small molecules. Synthetic biologists variant of it by means of nucleotide substitution etc. (Takahashi et al, 2013). In this paper, many variants ofpT181 attenuator/antisense is constructed and the attenuation rate of each variants is different. We chose this mechanism in gene repression.
Repressor
We took up TetR aptamer as an example of functional RNA which activates a transcription. TetR aptamer induces tetracycline promoter (Ptet) by binding to tetracycline repressor (TetR), which represses Ptet. When TetR aptamer binds to TetR, induces conformational change of TetR. As a result, TetR cannot come to bind to tetracycline operator (tetO). We ordered MBL=IDT gene synthesis of pT181 attenuator region DNA, antisense DNA and TetR aptamer with prefix and suffix. We transferred these parts to pSB1C3 and constructed device for antisense and attenuator assay (Fig. ).
Fusion
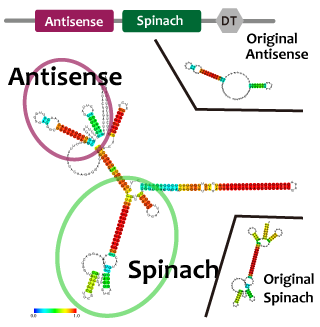
これらを使って遺伝子回路を組み立てるとき、複数のmoduleを同じ機能要素に組みこまなければならないときも十分あり得る。例えば転写抑制の様子をレポートするとき、異なる因子で促進と抑制を行うような系を作るときである。このとき、複数のモジュールを連結したことによる相互作用や立体構造の問題により機能が阻害される可能性がある。タンパク質ではその問題を予測するのは難しいが、RNAであれば配列情報から比較的簡単に二次構造を予測することができ、これらの問題を回避出来る。われわれは、機能を確認したtetR aptamer, Antisense-Attenuator RNA, をそれぞれつなぎあわせ、二次構造を予測し、実際に働いていることを確認した。tetRタンパク質存在下でtetR aptamerとAttenuator antisense RNAを組み合わせたRNAがPtetプロモーター下流のGFPの転写量を増加させるかを確認した。
並びにAttenuator antisense RNAとSpinarchを連結したRNAを発現させ、Attenuator Region下流のGFP遺伝子の発現量が減少していることとSpinarchがDFHBI存在下で蛍光するかどうかを確認した。
Intending to check the process of transcriptional repression system and the system which promotes and represses processes of transcription by using different factors, we have to join some modules into a single RNA strand.
When we combine plural modules, the function of the modules may be inhibited by interactions and steric structures between each other. in the case of RNA,it is easier to predict and avoid the steric problems than that of proteins because we can predict the secondary structure of RNA from its primary structure. We combined tetR aptamer and Antisense-Attenuator RNA, whose functions are confirmed, and predicted secondary structures, as a result it actually worked. We also observed tetR aptamer-Attenuator antisense fusion RNA increased expression level of downstream GFP of tet promoter in the presence of tetR proteins.
-----const-------
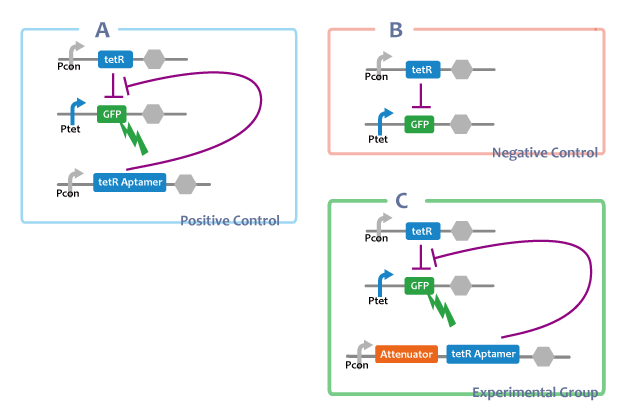
experimental group
a. Pcon-atte-tetRaptamer-DT Ptet-GFP-DT Pcon-tetR-DT
positive control
b. Pcon-tetRaptamer-DT Ptet-GFP-DT Pcon-tetR-DT
-Fusionする前とのtetRaptamerの働きの比較
negative control
c. Ptet-GFP-DT Pcon-tetR-DT
Positive Control
A. Pcon-tetRaptamer Ptet-GFP Pcon-tetR
Through this E.coli, we can confirm that separated tetRaptamer restricts the function of tetR protein.
Negative Control
B. Ptet-GFP Pcon-tetR
This E.coli shows that tetR protein represses the expression of genes at the downstream of tet promotor.
Experimental Group
C. Pcon-atte-tetRaptamer Ptet-GFP Pcon-tetR
------const-------
We used centroid fold (URL) and mfold (URL) to predict the secondary structure of RNA(a). As the picture shown below, the structure of tetR aptamer is not affected by attenuator stem loop. This suggests that the efficiency of tetR aptamer is not affected by the existence of attenuator stem loop.
Centroid fold, mfoldのfig-tetR aptamer only----tetR aptamer-antisense
tetRが常時発現されている状態では、AttenuatorとtetRaptamerを連結したRNAを転写する大腸菌(figC)のGFP発現量は、tetRaptamerを転写しない大腸菌(figB)よりも多く、ほかのRNAと連結していないtetRaptamerを転写する大腸菌(figA)と比較して{ほぼ同等 or 小さい}であることから、AttenuatorとtetRaptamerを連結すると、tetRaptamerは{全く干渉せずに機能する or 効果は下がるが機能する}ことがわかる。
If tetR is expressed, E.coli in which the united RNA(figC) was introduced expresses more GFP than E.coli which didn’t have the tetRaptamer sequence(figB). By comparing E.coli expressing independent tetRaptamer(figA) and E.coli expressing the united RNA(figC), it is convinced that tetRaptamer next door to Attenuator { works as well as independent one / works more weakly than a independent one. However, it certainly works.}
Experiment
(・Construction) ・転写確認 ・構造予測
Result
(・コンスト泳動結果)
RT-PCR
We performed RT-PCR to confirm transcription of tetR aptamer, antisense-spinach, spinach, and GFP(GFP generator). File:ElectrophoresisRT
Structure Prediction
Conclusion
We confirmed transcription of tetR aptamer, antisense-spinach, spinach, and GFP by using RT-PCR method.
We predicted second structure of fusion RNA: atenuator-tetRaptamer and antisence-spinach with centroid fold. It seems to be expected structure and to function as expected.
We got ready for construction oscilator circuit in wet lab.
future work
・定数探し ・シュミレーションする ・Attenuator-tetR アプタマーのコンストラクション ・機能確認 ・実際にwetでオシレーションができることをみる ・wetとdryを比較する
文章のたまり場
旧conclusion
In this project we confirmed the function of activator (tetR / tetR aptamer) and repressor (Attenuator region / Antisense). Moreover, we predicted the secondary structure of linked RNA (Antisense RNA-tetR aptamer) to check the influence of linkage to the structure, and finally we confirmed that actually the function of tetR aptamer do not lost. Our outcome of this project will directly connects to the progress of synthetic biology, especially constructing gene circuits. These two types of functional RNA will play important role when we regulate gene expression in peculiar gene cycle. For example, we may regulate a gene circuit which contains rapid gene transcriptional cascades by these RNA modules.
Future Work
To show this possibility, we designed a gene circuit which uses these RNA module. This circuit produces transcriptional oscillation. Oscillation circuits are important and essential gene circuits in many organisms and always be in the center of synthetic biology, therefore it is suitable for the cutting edge of new type of gene regulation.
When it comes to oscillation, we have to have a module which acts as reporter to show the changing amount of post-transcriptional RNA. Usually, protein reporters such as GFP are used for this purpose. However, in this circuit protein reporters may not be able to be used, because of the length of the period of the oscillation. Because RNA’s degradation is so fast and RNA do not need to be translated or folded like protein, the period of oscillation should be too short. According to XX who constructed similar gene circuit using RNA modules, this kinds of circuit produces 10 minutes cycle reaction. This means protein degradation is too slow (takes XX hours even with the degradation tag) [要出典] to image this RNA oscillation.
To solve this problem, we will suggest a new RNA module, which called spinach. This is a kind of aptamer, which is designed by Jeremy S. Paige, Karen Y. Wu, and Samie R. Jaffrey.20 They imitated the structure of GFP in this project. The designing of Spinach is changing the structure of an aptamer which specifically combines with DFHBI, which has similar structure to fluorophore of GFP. Denatured GFP doesn’t have fluorescence. Only if GFP is folded correctly, the fluorophore of GFP, which is in inner area, emits fluorescence. Therefore, we can confirm whether there is Spinarch in a sample by adding DFHBI. If the sample contains Spinarch, the sample will emit fluorescent. Vice versa. Spinach may degrade first enough for the oscillation, therefore we propose this for reporter of this oscillation.
The circuit of oscillation which uses spinach and the two RNA module is like below. This describes mechanism of producing oscillation.
この回路がオシレーションを形成する仕組みは、以下のようになっている。初期条件として、Constitutive Promoterにより合成されたTetRにより、Ptetはrepressされている。 オシレーションの開始はPtet下流のPlacがIPTGにより誘導されることである。これによってRNA-Actが合成開始され、その中のtetR aptamer配列がPtetをactivateする。 ActivateされたPtetはさらにRNA-Actを合成し、ここでポジティブ・フィードバックがかかることでRNA-Act, RNA-Repともにその量を増やす。すると、RNA-Repの配列内のSpinachにより緑色蛍光が確認される。 RNA-Repの量が十分に増えると、そのAttenuator antisenseの部位がRNA-ActのAttenuator locusに結合し、RNA-Actの転写量を減少させる。 するとTetR-AptamerによるActivationが小さくなることで、RNA-Act, RNA-Repの量が減少する。すると、Spinachによる蛍光は減衰する。 RNA-Repの量が十分に減少すると、Attenuator antisenseによる転写抑制が解かれ、再びRNA-Actの転写量が増えることとなる。これが繰り返されることで、オシレーションを作り上げている。この回路からは、RNAならではの分解・生成が速い性質によって、10分周期程度の短いSpinach蛍光のオシレーションを生むことが出来ると予測できる。
This circuit oscillates in the following way: First, tet promoter is repressed by tetR at the downstream of constitutive promotor. Then, the oscillator is turned on by IPTG. IPTG activates Plac and tetRaptamer, Spinarch, and Antisense at the downstream of Ptet which are transcribed. Because tetRaptamer activates tet promotor, positive feedback occurs and more and more tetRaptamer, Spinarch, and Antisense are accumulated. Then, this circuit gets fluorescence. After Antisense is accumulated to some extent, tetRaptamer, at the downstream of Atteruator region, is repressed. Then, because new tetRaptamer is not created, the amount of tetRaptamer decreases quickly. So, tet promotor is repressed by tetR protein and the amount of Antisense and Spinarch falls, too. Then, this circuit loses fluorescence. After the amount of Antisense decreases sufficiently, this circuit recovers first condition. Through this cycle, this circuit acts as an oscillator. Since RNA is generated and resolved quickly, this circuit should oscillate as quick as about in 10 minutes' cycle.
☝☝たぶん時制がめちゃくちゃですごめんなさい(´._.`)
Achievement
我々は、このプロジェクトで以下のことを達成した。
①
②
③
④
⑤
⑥
Parts List
<groupparts>iGEM013 Kyoto</groupparts>
Reference
[x][http://www.nature.com/nature/journal/v456/n7221/abs/nature07389.html Jesse Stricker et al.(2008)"A fast, robust and tunable synthetic gene oscillator" Nature 456, 516-519]
[7(仮)][http://www.ncbi.nlm.nih.gov/pubmed/19246008 Anke Hunsicker et al.(2009)"An RNA aptamer that induces transcription"Chem Biol,16(2),173-180]
[20(仮)][http://www.sciencemag.org/content/333/6042/642.abstract Jeremy S. Paige et al.(2011)"RNA Mimics of Green Fluorescent Protein"Science Vol. 333 no. 6042 pp. 642-646]
[5(仮)][http://www.ncbi.nlm.nih.gov/pubmed/23761434 Melissa K. Takahashi and Julius B. Lucks.(2013)"A modular strategy for engineering orthogonal chimeric RNA transcription regulators"Nucleic Acids Research 41(15),7577-88]
[http://www.ncrna.org/ Functional RNA Project provided by Computational Biology Research Center (CBRC)]
 "
"