Team:UC-Santa Cruz/Notebook3
From 2013.igem.org
2013 UCSC IGEM TEAM Biofilm Membrane Notebook.
The primary goal of the Biofilm Membrane team was to produce a high-density monolayer biofilm. A monolayer biofilm structure is critical to the function of the biomachine as the overall goal of the project is to move salt across a membrane, producing potable water. Caulobacter Crescentus (C. crescentus), is a polar body bacteria that forms high density monolayer biofilms and has been well characterized to support its use in biotechnological applications. The biofilm forming capabilities of a strain of C. crescentus (CB15) have been well characerized by a group at Stanford University whose work we have cited. CB15 was obtained from the Standford lab for the purpose of this project.
Week 1: To prepare materials that will be used for future experiment.
8/15
Peptone Yeast Extract (PYE) Protocol
(for preparation of one Liter)
Aim : To make 1 Liter of PYE growth medium for Caulobacter
Materials :
Bacto Peptone 0.2% approx. 2g lot# 1194403
Yeast Extract 0.1% approx. 1g lot# 1307388 vat# 288620
MgSO4 1mM 0.247g EMD#MX0070-1 lot# uk09FZEMS
CaCl2 o.5mM 0.074g lot# A0141702020
1 Liter flask
milliHQ water
autoclave
Procedure :
1) Combine 2g of bacto peptone, 1g of yeast extract, 0.24624g of MgSO4, and 0.07350g of CaCl2 in a sterile 1 Liter flask.
2) Fill flask with millliHQ water to 1 Liter mark on flask.
3) Homogenize mixture by shaking/stiring.
4) Sterilize mixture using the autoclave on fluid cycle for 40 minutes.
5) Let cool, cover, and store in refrigerator.
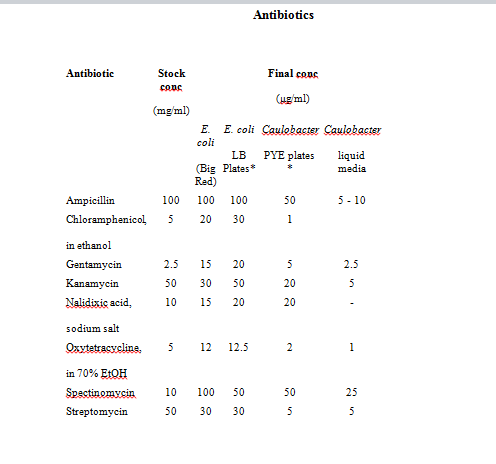
Refer to http://labs.mcdb.lsa.umich.edu/labs/maddock/protocols/media/antibiotics.html </a> for Antibiotics Concentations.
4/9/93
Notes:
Storage: All antibiotics should be stored at 4° C, Tet should be stored at -20° C.
Tet is also light sensitive. Store stock solutions & plates in the dark.
Prep: All antibiotics should be dissolved in sterile distilled water unless otherwise indicated.
Use oxytetracycline for Caulobacter strains.
8/15
Innoculating Ecoli PxYFPC-2
Aim: Picking Ecoli from a plate to get PxYPFC-2 plasmids
Materials:
-Ecoli PxYFPC-2 plate from Stanford
-LB growth medium (made by Aaron)
-Kanamycin ( stock conc. 50mg/ml)
-Inoculating Loop
-Flame
Procedure:
1. Set up a flame
2. Sterilize the loop by heating it over the flame
3. Using the inoculating loop, pick a colony
4. Dip loop into sterile LB broth (100ml)
5. Store in shaking incubator at -37oC for 1 night
8/19 Agar Plates
Aim: To make PYE + antibiotic agar plates
Materials:
-Petri dishes
-PYE (500ml- this is enough to make 15-18 plates)
-Kanamycin/Gentamycin
-Agar (13-15 g per liter) [Lot # 2107450]
-1 Liter Erlenmeyer Flask
Procedure:
1. Add 7.5g of agar and 500mL of PYE to a 1L flask.
2. Homogenize mixture using a stirring bar.
3. Autoclave for 40 mins on fluid cycle.
4. Cool mixture to 50°C
5. add antibiotic to mixture:
6. For antibiotic concentrations refer to:
“A comprehensive set of plasmids for vanillate and xylose-inducible gene expression in Caulobacter Cresentus”
for kanamycin plates: add 250uL kanamycin
for gentamycin plates: add 250uL gentamycin
7. Pour agar into sterilized petri dishes.
8. Let stand for 15 mins to allow agar to solidify.
9. Cover with parafilm and store in refrigerator.
Week 2: To create a standard growth curve for CB15
8/26
Ampicillin dilution
Aim: to make stock ampicillin 50mg/ml
Materials:
-Ampicillin [Lot # 29371]
-BD syringe 0.2micron filtuer
-50ml centrifuge tube
-eppendorf tubes
Procedure:
1. Weight out 0.5 g of Ampicillin
2. Place in 50ml centrifuge tube along w/ 10ml of dH2O.
3. Vortex the solution
4. Using BD syringe filter out 10ml of diluted antibiotic into eppendorf tubes.
5. Fill 20 eppendorf tubes w/ aliquot of 500ul.
8/28
Growth Curve
Aim : to create a standard caulobacter growth curve
Materials:
-PYE+AMP
-50ml centrifuge tubes x6
-CB15
Procedure:
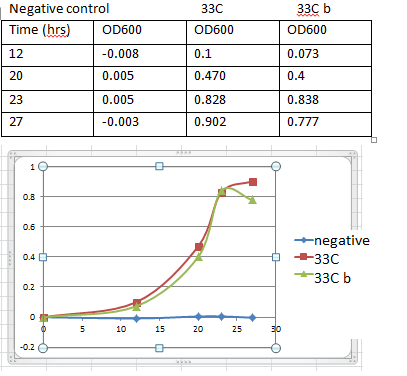
For 33 degrees C
1. Label 1 centrifuge tube “negative” and the other two c tubes. “33 ” and “33 b”
2. Make 80ml of pye + amp.
Final conc of amp = 10ug/ml
Add 80ml of pye into a flask w/ 16ul of ampicillin( stock conc. 50mg/ml)
3. Fill each tube w/ 4ml PYE
4. For negative control tube dip a sterile pipette tip in the tube. Discard tip.
5. For the other 2 tubes pick a colony from CB15 plate w/ a pipette tip. Pick two colonies one for each.
6. Store in incubator at 33C at 300rpm
7. Make a table and later transfer in unto a plot of O.D600 versus time graph.
8. Record an OD reading every couple of hours.
Results
8/26
Inoculation of CB15
Aim: making liquid culture of CB15
Materials:
-CB15(from Stanford)
-Antibiotic
-PYE
Procedure:
1. Add 50ul of Ampicillin (50mg/ml) into 250ml of PYE
Refer to for final concentration of Ampicillin(10ug/ml):
“Dynamics and Control of Biofilms of the Oligotrophic Bacterium Caulobacter Crecentus.”
2. Pipet 20ml of pye + amp into centrifuge tube. Do this twice. Label one “Negative” and the other “CB15”
3. Pick a colony from CB15 plate and eject tip in tube
4. For negative control, drop a blank pipet tip
5. Incubate at 28°C and 180 rpm
Results:
After 1 night PYE medium for negative control turned cloudy. Experiment has been contaminated.
8/29
Making Glycerol Stocks
Aim: for long term storage of CB15
Materials:
-glycerol 60% (filtered)
-cell culture cb15
-eppendorf tubes.
Procedure:
1. Prepare a preculuture: Pick a CB15 colony and inoculate it in 5ml of PYE. When culture reaches stationary phase the O.D600 should be around 1.3-1.6
2. Caulobacters are store in 10% glycerol
Refer to: “Caulobacter S-Layer Secretion Kit Manual”
3. Calculate 10% glycerol.
(x amount of 10% glycerol )(60)= (900ul of cells + x)(10)
This comes out to be 180ul of 60% glycerol per 900ul of cb15
4. In an eppendorf tube combine 180ul of 60% glycerol and 900ul of cb15 culture.
5. Shake tube.
6. Label and store tubes at -80C
8/29-8/30
Preparing Electrocompetent CB15
Aim : making electrocompetent CB15
Materials:
-PYE
-eppendorf tubes
-centrifuge tubes(50ml)
-1L falsk
-ice-cold, sterile, distilled water
-ice-cold, sterile 10% glycerol in distilled water.
Procedure : will yield enough electro competent cells for 15 transformation.
1. Diluting 60% stock conc. Glycerol into 10%
Add 80ml of distilled water and 16ml of 60% filtered glycerol from fridge into a flask
2. Autoclave 1000ml of distilled water, 250ml PYE flask (x2), 96ml of 10% glycerol - fluid cycle 40mins
3. Inoculate a CB15 colony into 5ml PYE medium. Grow at 30oC w/ shaking until the O. D is 0.5-1.0 ( should take about 1 night)
4. Record the O.D of the culture above. 0.902
5. Dilution calculation: calculate the amount of culture you would add to 250ml of fresh PYE to achieve an O.D of 0.4-0.6 in 18hrs.
-doubling time of Caulobacter is about 3hrs. = 6 times doubling
26 = 64 -> 0.5/64= 0.008(starting O.D)
(xml)(0.902O.D)=(250ml)(.008O.D) x=2ml
Add 2ml of culture to 250ml PYE
6. Incubate for 18hrs.
7. Split 250ml of culture among five 50ml centrifuge tubes
8. Centrifuge cells at 10000XG for 7mins at 4oC
9. Remove supernatant and resuspend cells by in 50ml of ice-cold sterile, distilled water. Repeat for all tubes. DO NOT VORTEX
10. Centrifuge w/ same conditions
11. Remove supernatant and resuspend the cells in 25ml of ice-cold, sterile, distilled water.
12. Centrifuge.
13. Remove supernatant and resuspend the cells in 1/20 the original volume (50ml in each tube)
1/20 * 50ml = 2.5ml of ice-cold, sterile 10% glycerol.
14. Centrifuge
15. Remove supernatant resuspend cells in 150ul of ice-cold, sterile 10% glycerol.
16. Dispense cells into 50ul aliquot in eppendorf tubes. We ended up w/ 15 tubes total
17. Store at 80oC.
8/30
CB15 Transformation
Aim : Transforming CB15 w/ PxYFPC-2 and PvCFPC-4 plasmids
Materials :
-CB15(from Stanford)
-PxYFPC-2
-PvCFPC-4
-PYE+Kanamycin plates (x3)
-PYE+Gentamycin plates (x3)
-ice-cold sterile dH2O
-electroporation cuvettes (x3)
-sterile beads
Procedure:
1. If frozen, thaw electro competent cells on ice.
2. Place electroporation cuvettes on ice.
3. Dilution of Plasmids:
PxYFPC-2 conc. Is 293.2ng/ml in freezer.
-to dilute add 1ul of stock conc. into 100ul of sterile cold dh20. Making the final conc. 2.932ng/ul -> label this Px.
PvCFPC-4 conc. is 302.3 ng/ul in freezer
-to dilute add 1ul of stock conc. into 100ul of sterile cold dH20. Making the final conc. 3.023ng/ul -> label this Pv.
4. Pipet 100ul of electro competent CB15 cells to a electroporation cuvette.
5. Add 20ng of plasmid DNA so add 7ul of diluted plasmid DNA (PxYFPC-2)
6. Mix around using pipette
7. Electroporate at 2.5kV
8. Record time constant.
9. Take cuvette and immediately add 600ul of PYE. Mix using pipette.
10. Transfer to microcentrifuge tube. Then to a shaking incubator at 33oC / 300rpm/ for 2hrs(we only left it in there for 1hrs)
11. Repeat steps 5-11 for the second plasmid DNA (PvCFPC-4) and negative control. Instead of using plasmid DNA add 7ul of sterile dH20 for negative control.
-Time constants a. pxyfpc-2 5.0
b. pvcfpc-4 5.2
b. negative 4.8
time constant b/w 3.6-4.3 msec are good (depends on the machine)
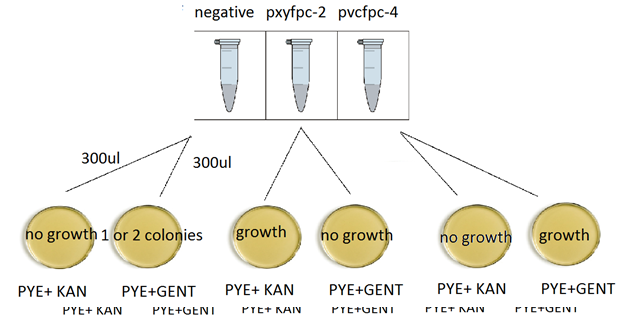
12. Plating:
Plating scheme:
13. For each tube of 600ul aliquot of transformed cells put 300ul in Kan plate and the other 300ul into Gent plate. Repeat this procedure 2 more times for negative control and the other transformation.
14. Incubate plates at 33oC for 3 days.
9/2 (Monday)
After 3 days of incubation, results are as follows
-There are contaminants in the negative control.
-Plates w/ transform cells turned out pretty well. The results are as expected. Since PxYFPC-2 is kanamycin resistant and PvCFPC-4 is gentamycin resistant, transformation w/ PxYFPC-2 has growth only on PYE+KAN plate and PvCFPC-4 has growth on PYE+GENT plate.
Week 3: Production of Biofilms
9/3
Production of Biofilms (Draft)
Aim: To grow high density biofilms.
Materials:
-glass coverslips x6
-filter paper x6
-12 well culture plate
-pye+ amp
-preculture – prepared on 9/2.
Procedure :
1. Check O.D of preculture made on 9/2 by Rolando. 0.D = 1.250
Preculture: a colony was inoculated into a test tube w/ 5ml pye.
2. Autoclave glass coverslips (20min sterilization, 20 min dry)
3. Flame forcep and prepare 100ml PYE + AMP(20ul)
4. Cut filter paper into 2 pieces per sheet ( cut out 8 pieces)
5. Place filter paper/glass in wells.
-modified design w/ 7 wells with filter paper and 5 wells with glass.
6. Fill each wells with 2ml PYE+AMP
7. Inoculate 10 wells w/ 10ul of preculture culture and use 2 wells for negative control (one for glass and one for filter paper)
8. Incubate at 33oC at 350rpm.
Results : Images are obtained from bright field microscope under 40x after 20-24 hrs. of growth. There are no visible cells on filter paper(left) oppose to a semi uniform layer of biofilm forming on the glass surface(right)
9/6
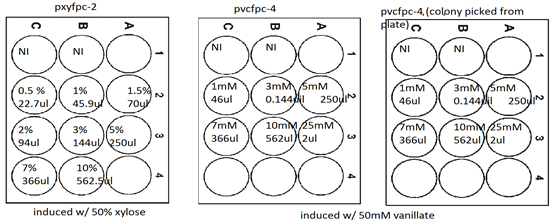
Inducing xylose and vanillate
Aim: To inducing transform cells w/ 0.03% xylose or .5mM vanillate on agar plates.
Materials:
-pye+gent agar plate(x2)
-pye+kan agar plate(x2)
-1% xylose
-50mM vanillate
Procedure:
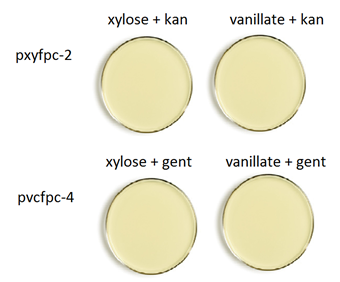
Plating scheme:
1. Calculate the amount of 1% xylose to add on agar plates
- Xylose = 0.03%
15ml(0.03g/100ml) = x (.25g/25ml)
x = .45ml = 450ul
squirt 450ul of 1% xylose one pye + kan and one pye + gent plate. Wait 15mins until they dry
2. Calculate the amount of 50mM vanillate to add on agar plates (pye +gent)
- Vanillate = 0.5mM
50mM x = (15 + x )(0.5mM) x= 151.5ul
squirt 151.1ul of 50mM vanillate one pye+ gent and one + kan plate. Wait until they dry
3. Pick a colony from CB15 + PxYFPC-2 transform plate and streak on a kan plate with xylose. Repeat for the other kan plate with vanillate.
4. Repeat step 3 for PvCFPC-4 on gent plates.
5. Incubate for 2 or 3 days at 33oC
6. Look at plates under UV lamp
Results: There is no difference in fluorescent b/w induced and non-induced plates.
9/5
Protocol for 50mM vanillate
Aim: to make 50mM vanillate from vanillic acid.
Materials:
-0.21g vanillic acid [lot b185020]
-naoh
-centrifuge tube
-.2 micron filter
-syringe
Procedure:
Calculation
X = (0.050)(0.025)(168.5) = 0.210 g vanillic acid
1. Add 0.210g of vanillic acid acid into a centrifuge tube along w/ 25ml dh20.
2. Vortex solution. Until it homogenizes
3. Titrate w/ NaOH until pH reaches 7.4
9/9
Inducing xylose or vanillate into liquid cultures.
Aim: Inducing xylose or vanillate into liquid cultures.
Materials:
-PYE + Kan
-PYE+ Gent
-50mM vanillate
-50% xylose
-glass test tubes (x4)
-50ml centrifuge tubes (x4)
Procedure:
Calculations:
2mM vanillate in 25ml PYE from 50mM stock concentration vanillate
X(50mM)= ( 10+x)(2) x= 412ul
1% xylose in 10ml PYE from 50% stock concentration xylose.
X(50mM)= ( 10+x) x= 204ul
1. Add 5ml of PYE+Kan to a test tube, then inoculate it w/ pxyfpc-2 by picking a colony from transformed plate. Make a negative
2. Repeat step 1 for pvcfpc-4. Use pye + gent instead of pye + kan
3. Incubate at 33oC 300rpm overnight.
4. Prepare 4 tubes, two w/ 10ml pye+kan and the rest w/ pye + gent.
5. For the two 10ml pye+kan inoculate each tube w/ 2.0ml of overnight preculture(pxyfpc-2). Do the same for pvcfpc-4 (2ml of preculture(pvcfpc-4) into pye+gent)
6. Wait a couple of hours until O.D reaches 0.4-0.6.
O.D 0.537 when induce
7. Inoculate one pye+kan tube w/ 416ul of 50% xylose. The second one is negative control (Noninduced)
8. Inoculate one pye +gent tube w/ 204ul of 50mM vanillate. Second one is noninduced
9. Incubate at 33oC 300rpm overnight.
10. Check if cells fluoresce under w/ U.V lamp.
Results: Induction w/ 2mM vanillate ( 2 tubes next to each other on the right) doesn’t seem to fluoresce under U.V light. On the contrary tube(far left) with 1% has a different color compare to its’ noninduced negative control

 Left:YFP Middle:Dapi Right:DIC
Left:YFP Middle:Dapi Right:DIC
Images are taken using a confocal microscope at 100x
9/10
1X PBS (phosphate buffer saline) 1L
Aim: To make PBS for DAPI staining
Materials:
-8g of NaCl 137mM NaCl
-0.2g of KCl [lot 123796] 2.7mM KCl
-1.44g of Na2HPO4 [lot 116747] 10mM Na2HPO4
-0.24g KH2PO4 2mM KH2PO4
-HCl
Procedure:
1. Recipe above is for 1L. We’re only making 500ml so divide everything by 2.
2. Add 4g NaCl, 0.1g KCl, 0.72g Na2HPO4, and 0.12g KH2PO4 to a bottle
3. Then add dH20 to the 500ml mark
4. Titrate with HCl until pH reaches 7.4
9/11
Stock solution of DAPI
Aim: to make stock solution of DAPI
Materials:
-10mg DAPI
-2ml dH2O
-20 eppendorf tubes.
Procedure:
1. Add 2ml dH2O into 10mg DAPI vial.
2. Vortex vial.
3. Aliquot 10ul into eppendorf tubes.
4. Store in freezer. Away from light.
9/11-9/12
Vanillate and xylose induction at different concentration.
Aim: to find a concentration at which transform cells fluoresce.
Materials:
-12 well culture plate (x3)
-CB15+PxYFPC-2 preculture (from previous experiment)
-CB15+PvCFPC-4 preculture (from previous experiment)
-CB15+PvCFPC-4 pick from a plate
-PYE+KAN
-PYE+GENT
-50% xylose
-50mM vanillate
Procedure:
9/11
1. Precultures of CB15+PxYFPC-2 and CB15+PvCFPC-4 were prepared from previous induction experiment. 5ul of preculture was added to new 5ml PYE.
2. Pick a colony from pvcfpc-4 transformed plate. Prepare a negative.
3. Incubate at 200rpm 33oC
9/12
4. Put 2ml of PYE+KAN in 6 wells on a culture plate. Repeat twice for PYE+Gent on new plates. (one gent would from preculture and the other one from a new colony picked from plate)
5. Inoculate all 2ml PYE+Kan wells w/ 250ul of pxyfpc-2 preculture.
6. For Pye+Gent inoculate one plate w/ 250ul of pvcfpc-4 preculture and the other plate w/ preculure from colony picked from plate
7. Incubate at 33oC 200rpm until O.Ds of all cultures are round 0.4-0.6. Then induce according to the following scheme.
8. Incubate at 200rpm 33oC for atleast 5 hrs.
9. Look under uv lamp for floursecent
Results.
For PxYFPC-2 there seems to be fluorescent at 0.5% xylose.
For PvCFPC-4 cells fluoresce at 5mM to 7mM
Protocol for DAPI staining
Refer to http://www.igsb.org/uploads/pdf/ProtocolDAPIStaining.pdf
Aim: To stain biofilms w/ DAPI
Materials:
-4ml of 4% PFA(paraformaldehyde) look at protocol
-1X PBS: look at protocol
-4ml of 0.1% Triton-X-100: look at protocol
-4ml of DAPI(stock conc. 1ug/ml): look at protocol
-fume hood
-Nutator
-biofilm coverslip
-12 well culture plate
-paper towel and aluminum foil for disposal for PFA
Procedure:
1. Make 4% PFA, 1X PBS, 0.1% Triton-X-100, and DAPI stock conc. 1ug/ml (prepare these by looking at protocols). Do all work w/ PFA in fume hood.
2. Using a new 12 well culture plate, fill a well up w/ 4ml of DH20.
3. Take a coverslip to be analyze, wash slip by transferring it to the well w/ 4ml of dH2O for 2mins w/ gentle shaking (in order to get rid of swarmer and unattached cells.)
4. Do this next step in fume hood, since we are going to work w/ PFA. Aspirate dH2O and replace medium w/ 4ml of 4% PFA. Leave coverslip in solution for 8 minutes and place plate on a nutator. Do not leave it in there for more than 10mins.
5. Aspirate 4% PFA medium. PFA is volatile so dumped liquid waste onto paper towels that should be wrapped in aluminum foil before disposal.
6. Wash cells by inoculating 4ml of 1x PBS into well for 5mins. Place plate on a nutator. Repeat this process 2 more times (wash cells w/ 1x PBS for a total of 3 times.)
7. Remove 1X PBS and add 4ml of 0.1% Triton-X-100 (to permeabilize cells). Leave coverslip in there for 10mins. Also place plate on a nutator.
8. After 10mins, repeat step 6. Wash cells 3 times w/ 1X PBS (5mins/wash.)
9. Stain cells by inoculating well w/ 4ml of DAPI for 5mins. Place plate on a nutator.
10. Wash cells w/ 1X PBS for 5 mins.
11. Coverslip is now ready to be analyzed
12. See microscopy protocol.
9/16-9/20
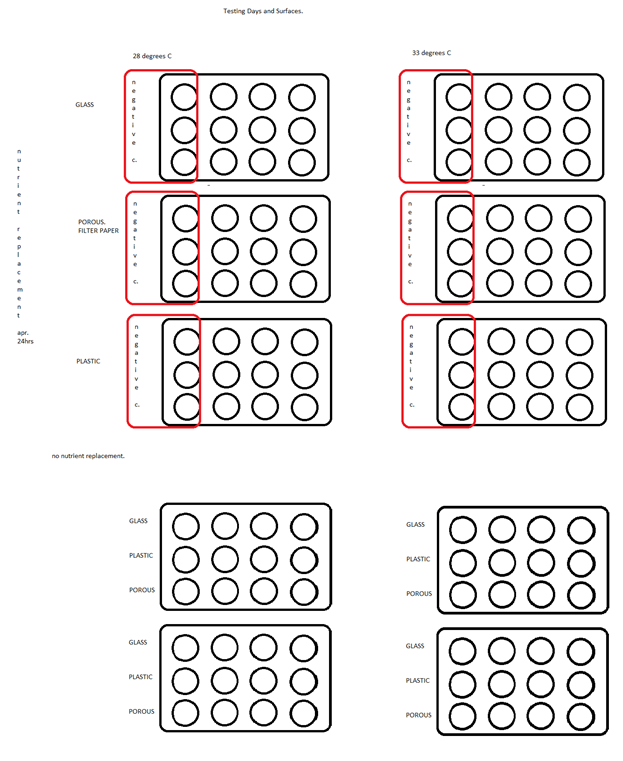
Production of Biofilm
Aim : To grow a high density uniform monolayer under varying parameters. We will culture the bacteria for 4 days taking data points at various time intervals. The cultures will be grown on glass cover slips, micro-filter paper, and plastic.
Materials :
C. crescentus CB15 (pre-culture)
PYE + ampicillin
Clorox
Ethanol
Milli HQ water
12 Well Tissue Culture Plate x 10
40 Glass coverslips 15mm
40 Filter Paper 0.22 micron
40 Plastic coverslips
forceps
Protocol :
1) Confirm if supply of pre-culture is available.
Preparing a pre culture: Using a pipette tip pick a colony from CB15 plate (from Stanford) dip it in 5ml PYE+amp. Incubate this at 200rpm and 33degrees C overnight.
2) Prepare glass cover slips by cleaning with w/ ethanol. There’s 10% ethanol in lab if not we can make 10% ethanol by adding 100ml of ethanol to 900ml dH20. Squirt some ethanol on coverslips and wipe them away with KimWipes.
3) Next sterilize glass pieces, PYE, dH2O, and forceps by autoclave at 120°C for 40 mins on fluid cycle
4) Sterilize plastic pieces by using ethanol and rinsing off w/ sterile dH20
See figure for layout (these steps done for each different surface type)
5) Place a cover slip in each well of tissue culture plate. We will have a total of 10 culture plates testing 3 different parameters (temps, days, and replacement of nutrients) See figure for experiment layout.
6) Add 2mL of PYE to each well.
7) Identify negative control wells.
8) Before inoculating check OD600 of preculture.
Procedure for spectrometer: Turn on the spectrometer, there’s a switch on the back of the machine. Hit option 4 Cell Count. Zero the machine out w/ a blank (do this by filling a cuvette w/ PYE, place it in the machine, and hit auto zero.) There should be an arrow on the cuvette, turn the arrow until it faces left and place it in the spectrometer. Read numbers off the top right. It should say 600nm and 0.00. Next pipette out 200ul of culture into another cuvette, place it in the machine, and read off the top right corner or press start and it’ll pop out a number (this is the OD at 600nm).
9) Inoculate each well that is to receive culture with 10uL of pre culture (CB15)
10) Incubate at 33°C at 300rpm in incubator
To take a data point
11 Remove the coverslip to be analyzed. Use sterile forcep to handle coverslip. Wash coverslip to remove swarmer and unattached stalked cells by preparing a petri dish filled w/ 15ml PBS (can also use a well and fill it w/ 2ml dh20). Place coverslip in PBS water for 2mins with gentle shaking.
12) return culture plate to incubator.
13) analyze removed cover slip under light microscopy. (see protocol)
9/23
Protocol for 0.1% Triton-X-100
Aim: To make 0.1% Triton-X-100 for DAPI staining
Materials:
-100% Triton-X-100
-250ml of sterile PBS
-sterile glass bottle for 250ml solution
Procedure:
1. Add 250ul of 100% Triton X-100 and 249.75ml of dH20 to a glass bottle
2. Storage: Store in tightly closed containter in a cool well-ventillated (area around 25C).
MSDS: https://fscimage.fishersci.com/msds/24515.htm
http://www.sigmaaldrich.com/etc/medialib/docs/Sigma/Product_Information_Sheet/1/t8532pis.Par.0001.File.tmp/t8532pis.pdf
 "
"