Template:Kyoto/Notebook/Aug 30
From 2013.igem.org
Contents |
Aug 30
Gel Extraction
| Name | quantity | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|---|
| pT181 attenuator(2) (EcoRI & SpeI) | 305.3 | 2.2 | 1.66 | 0.36 |
| pT181 attenuator(2) (XbaI & PstI) | 217.7 | 3.9 | 1.73 | 0.04 |
| pT181 antisense(2) (EcoRI & SpeI) | 347.7 | 3.1 | 1.88 | 0.31 |
| pT181 antisense(2) (XbaI & PstI) | 376.5 | 5.9 | 0.49 | 2.36 |
| pSB1C3 (EcoRI & SpeI) | 436.6 | 30.7 | 1.03 | 1.28 |
| pSB1C3 (XbaI & PstI) | 457.4 | 7.9 | 1.65 | 0.42 |
| Spinach (EcoRI & SpeI) | 490.5 | 3.0 | 1.87 | 0.26 |
| tetR aptamer 12_1R (EcoRI & SpeI) | 451.7 | -12.8 | 5.56 | -0.04 |
Miniprep
| DNA | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| RBS-lysis2 9 | 264.6 | 1.97 | 2.21 |
| RBS-lysis2 12 | 220.0 | 1.89 | 2.46 |
| RBS-lysis2 14 | 195.3 | 1.88 | 1.53 |
Colony PCR
| Sample | base pair |
|---|---|
| 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT(8/20 LB Amp) (1)~(4) | 2143 |
| 8/29 Plux-RBS-GFP-DT(8/27 LB-CP) (1)~(2) | 1227 |
| 8/29 Pbad/araC-RBS-RFP{BBa_I13516} (8/21 LB-CP) (1)~(2) | 2256 |
| 8/29 RBS-lysis3-DT-(1) | 1210 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 2min | 30cycle |
Ethanol Precipitation
| DNA |
|---|
| pT181 attenuator-(2) (EcoRI & SpeI) |
| pT181 attenuator-(2) (XbaI & PstI) |
| pT181 antisense-(2) (EcoRI & SpeI) |
| pT181 antisense-(2) (XbaI & PstI) |
| pSB1C3 (EcoRI & SpeI) |
| pSB1C3 (XbaI & PstI) |
| Spinach (EcoRI & SpeI) |
| tetR aptamer 12_1R (EcoRI & SpeI) |
Restriction Enzyme Digestion
| 8/30 RBS-lysis2 (9) | EcoRI | SpeI | 10x bufferB | 100x BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 7.5 µL | 0.5 µL | 0.5 µL | 3 µL | 0.3 µL | 18.2 µL | 30 µL |
| NC | 0.5 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 8.4 µL | 10 µL |
| 8/22 pSB1C3-(2) | EcoRI | SpeI | 10x Buffer | 100x BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 8 µL | 0.5 µL | 0.5 µL | 3 µL | 0.3 µL | 17.7 µL | 30 µL |
| NC | 1 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 7.9 µL | 10 µL |
| 8/22 pSB1C3-(2) | XbaI | PstI | BufferD | BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 8 µL | 0.5 µL | 0.5 µL | 3 µL | 0.3 µL | 17.7 µL | 30 µL |
| NC | 1 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 7.9 µL | 10 µL |
| 8/21 Spinach (1) | EcoRI | SpeI | 10x BufferB | 100x BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 4 µL | 0.5 µL | 0.5 µL | 3 µL | 0.3 µL | 21.9 µL | 30 µL |
| NC | 0.5 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 8.4 µL | 10 µL |
| 8/21 tetR aptamer 12_1R (1) | EcoRI | SpeI | 10xBufferB | 100xBSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 3 µL | 0.5 µL | 0.5 µL | 3 µL | 0.3 µL | 22.7 µL | 30 µL |
| NC | 0.4 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 8.4 µL | 10 µL |
| 8/21 pT181 attenuator-(2) | EcoRI | SpeI | XbaI | PstI | 10x BufferB | 10x BufferD | 100x BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 cuts | 4 µL | 0.5 µL | 0.5 µL | 0 µL | 0 µL | 3 µL | 0 µL | 0.3 µL | 21.7 µL | 30 µL |
| NC | 0.5 µL | 0 µL | 0 µL | 0 µL | 0 µL | 1 µL | 0 µL | 0.1 µL | 8.4 µL | 10 µL |
| 2 cuts | 4 µL | 0 µL | 0 µL | 0.5 µL | 0.5 µL | 0 µL | 3 µL | 0.3 µL | 21.7 µL | 30 µL |
| NC | 0.5 µL | 0 µL | 0 µL | 0 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 8.4 µL | 10 µL |
| 8/21 pT181 antisense-(2) | EcoRI | SpeI | XbaI | PstI | 10x BufferB | 10x BufferD | 100x BSA | MilliQ | total | |
|---|---|---|---|---|---|---|---|---|---|---|
| 2 cuts | 9 µL | 0.5 µL | 0.5 µL | 0 µL | 0 µL | 3 µL | 0 µL | 0.3 µL | 16.7 µL | 30 µL |
| NC | 1 µL | 0 µL | 0 µL | 0 µL | 0 µL | 1 µL | 0 µL | 0.1 µL | 7.9 µL | 10 µL |
| 2 cuts | 9 µL | 0 µL | 0 µL | 0.5 µL | 0.5 µL | 0 µL | 3 µL | 0.3 µL | 16.7 µL | 30 µL |
| NC | 1 µL | 0 µL | 0 µL | 0 µL | 0 µL | 0 µL | 1 µL | 0.1 µL | 7.9 µL | 10 µL |
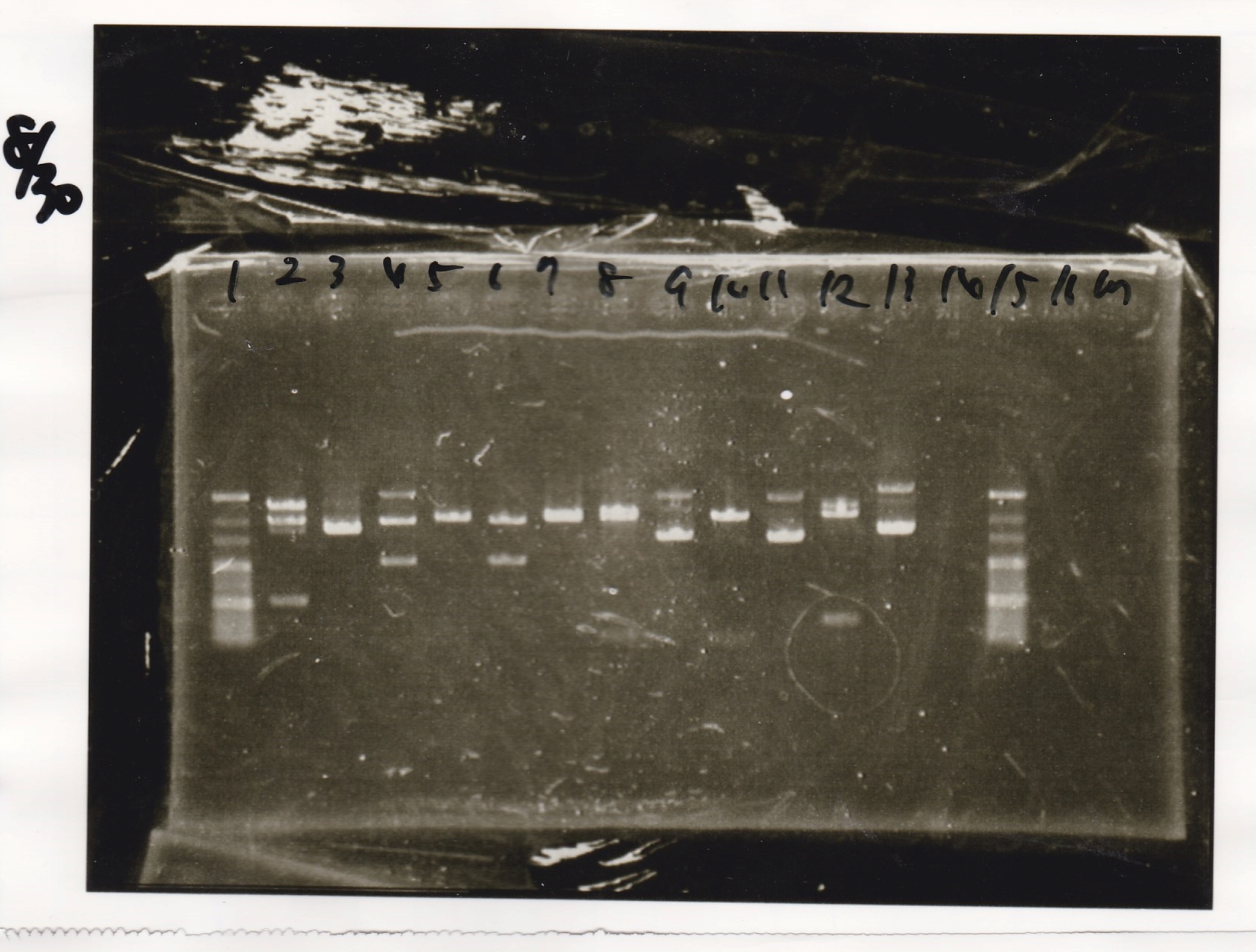
Electrophoresis
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | RBS-lysis2-(9) | EcoRI | SpeI |
| 3 | RBS-lysis2-(9) | -- | -- |
| 4 | pSB1C3-(2) | EcoRI | SpeI |
| 5 | pSB1C3-(2) | -- | -- |
| 6 | pSB1C3-(2) | XbaI | PstI |
| 7 | pSB1C3-(2) | -- | -- |
| 8 | tRNA-Spinach-(1) | EcoRI | SpeI |
| 9 | tRNA-Spinach-(1) | -- | -- |
| 10 | tetR aptamer12_1R | EcoRI | SpeI |
| 11 | tetR aptamer12_1R | -- | -- |
| 12 | pT181 attenuator | EcoRI | SpeI |
| 13 | pT181 attenuator | EcoRI | SpeI |
| 14 | 100bp ladder | -- | -- |
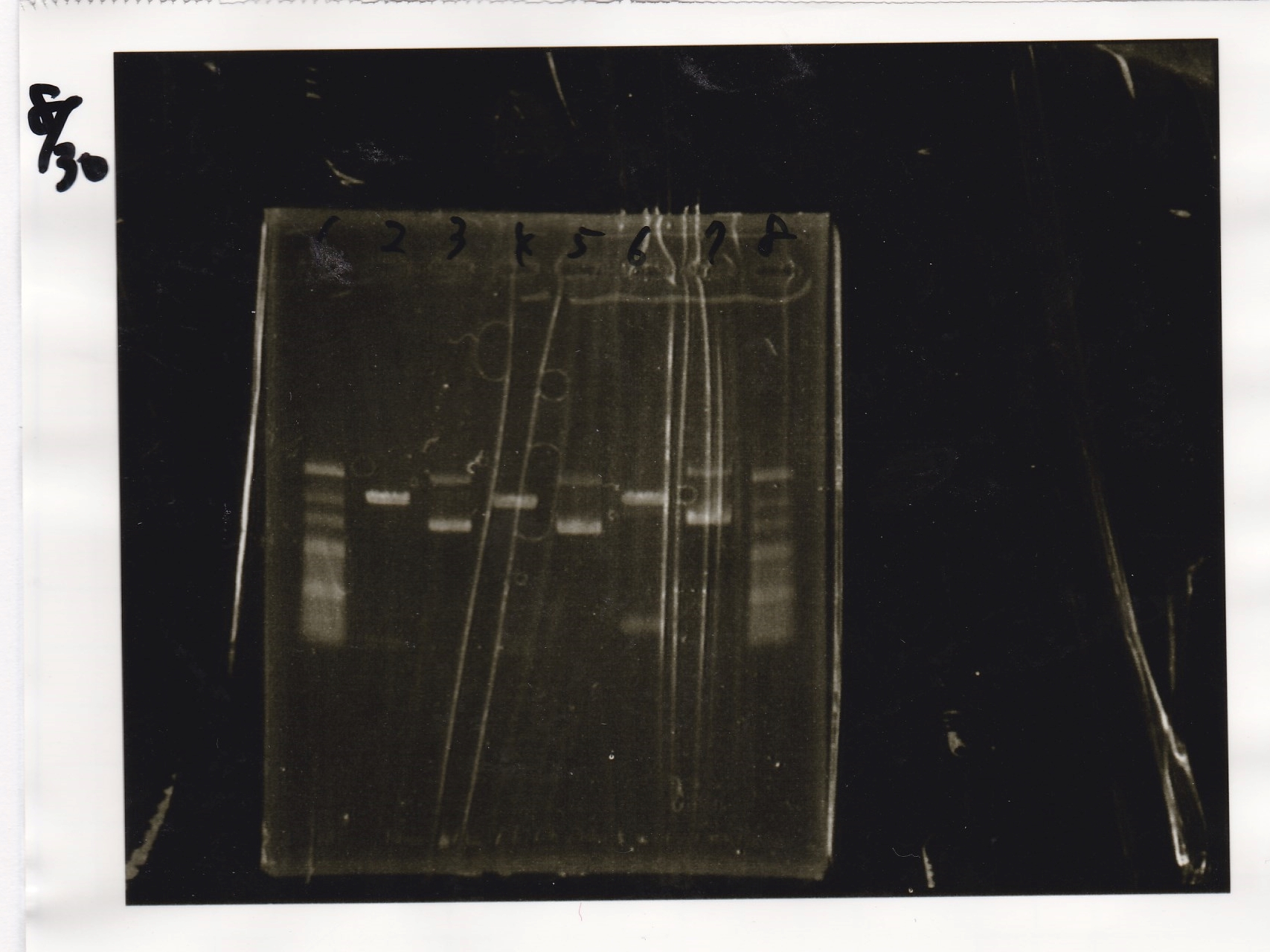
| Lane | Sample | Enzyme1 | Enzyme2 |
|---|---|---|---|
| 1 | 100bp ladder | -- | -- |
| 2 | pT181 antisense | EcoRI | SpeI |
| 3 | pT181 antisense | -- | -- |
| 4 | pT181 antisense | XbaI | PstI |
| 5 | pT181 antisense | -- | -- |
| 6 | pT181 attenuator | XbaI | PstI |
| 7 | pT181 attenuator | -- | -- |
| 8 | 100bp ladder | -- | -- |
| Lane | Sample |
|---|---|
| 1 | 1kb ladder |
| 2 | 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(1)(Colony PCR product) |
| 3 | 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(2)(Colony PCR product) |
| 4 | 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(3)(Colony PCR product) |
| 5 | 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(4)(Colony PCR product) |
| 6 | Pbad/araC-RBS-RFP-DT-(1)(Colony PCR product) |
| 7 | Pbad-araC-RBS-RFP-DT-(2)(Colony PCR product) |
Liquid Culture
| Sample | medium |
|---|---|
| 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(1) | Plusgrow medium(+Amp) |
| 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(2) | Plusgrow medium(+Amp) |
| 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(3) | Plusgrow medium(+Amp) |
| 8/29 Pcon-RBS-GFP-DT-Pcon-RBS-luxR-DT-(4) | Plusgrow medium(+Amp) |
| 8/29 Plux-RBS-GFP-DT-(1) | Plusgrow medium(+CP) |
| 8/29 Plux-RBS-GFP-DT-(2) | Plusgrow medium(+CP) |
| 8/29 Pbad/araC-RBS-RFP-DT-(1) | Plusgrow medium(+CP) |
| 8/29 Pbad-araC-RBS-RFP-DT-(2) | Plusgrow medium(+CP) |
| RBS-lysis3-DT-(1) | Plusgrow medium(+CP) |
 "
"