Template:Kyoto/Notebook/Sep 6
From 2013.igem.org
(Difference between revisions)
(→Colony PCR) |
(→Liquid Culture) |
||
| (32 intermediate revisions not shown) | |||
| Line 22: | Line 22: | ||
|8||100bp ladder | |8||100bp ladder | ||
|} | |} | ||
| - | [[File: | + | [[File:Igku Sep6 ColonyPCRN1.jpg]]<br> |
</div> | </div> | ||
| Line 39: | Line 39: | ||
|Ptet-pT181 antisense-(1~4)||604 | |Ptet-pT181 antisense-(1~4)||604 | ||
|- | |- | ||
| - | |Pconst-pT181 | + | |Pconst-pT181 attenuator-(1~4)||568 |
|- | |- | ||
|Pconst+RBS-tetR-DT-(1~4)||1121 | |Pconst+RBS-tetR-DT-(1~4)||1121 | ||
| Line 52: | Line 52: | ||
|5min||30s||30s||40s||30cycles | |5min||30s||30s||40s||30cycles | ||
|} | |} | ||
| - | |||
| + | </div> | ||
===Liquid Culture=== | ===Liquid Culture=== | ||
| Line 101: | Line 101: | ||
|13||Pcon-pT181 antisense -(4) | |13||Pcon-pT181 antisense -(4) | ||
|} | |} | ||
| - | + | ||
{| class="wikitable" | {| class="wikitable" | ||
!Lane||Sample | !Lane||Sample | ||
| Line 115: | Line 115: | ||
|5||Ptet-pT181 antisense -(4) | |5||Ptet-pT181 antisense -(4) | ||
|- | |- | ||
| - | |6||Plac-pT181 | + | |6||Plac-pT181 attenuator -(1) |
|- | |- | ||
| - | |7||Plac-pT181 | + | |7||Plac-pT181 attenuator -(2) |
|- | |- | ||
| - | |8||Plac-pT181 | + | |8||Plac-pT181 attenuator -(3) |
|- | |- | ||
| - | |9||Plac-pT181 | + | |9||Plac-pT181 attenuator -(4) |
|} | |} | ||
| - | [[File: | + | [[File:igku_Sep6_COlonyPCRN1_1a.jpg]]<br> |
| + | [[File:igku_Sep6_colonyPCR_N2_1b.jpg]]<br> | ||
| + | </div> | ||
===Miniprep=== | ===Miniprep=== | ||
| Line 143: | Line 145: | ||
|tetR aptamer-(2)||211.2||1.90||1.99 | |tetR aptamer-(2)||211.2||1.90||1.99 | ||
|- | |- | ||
| - | |DT181 | + | |DT181 attenuator||102.2||1.98||2.17 |
|- | |- | ||
|121R aptamer-DT||251.7||1.92||2.01 | |121R aptamer-DT||251.7||1.92||2.01 | ||
| Line 159: | Line 161: | ||
<span class="author">Tatsui</span> | <span class="author">Tatsui</span> | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | ! ||9/6 RBS-lysis2-DT (78.3 | + | ! ||9/6 RBS-lysis2-DT (78.3 µg/mL)||XbaI||PstI||BSA||Buffer||MilliQ||total |
|- | |- | ||
| - | |2 cuts||22 | + | |2 cuts||22 µL||1 µL||1 µL||3 µL||3 µL||0 µL||30 µL |
|- | |- | ||
| - | |NC||1.3 | + | |NC||1.3 µL||0 µL||0 µL||1 µL||1 µL||6.7 µL||10 µL |
|} | |} | ||
| - | |||
{| class="wikitable" | {| class="wikitable" | ||
| - | ! ||aptamer-DT (251.7 | + | ! ||aptamer-DT (251.7 µg/mL)||XbaI||PstI||BSA||Buffer||MilliQ||total |
|- | |- | ||
| - | |2 cuts||0.4 | + | |2 cuts||0.4 µL||1 µL||1 µL||3 µL||3 µL||14.1 µL||30 µL |
|- | |- | ||
| - | |NC||0.4 | + | |NC||0.4 µL||0 µL||0 µL||1 µL||1 µL||7.6 µL||10 µL |
|} | |} | ||
| - | |||
{| class="wikitable" | {| class="wikitable" | ||
| - | ! ||Pλ-luxI (86.4 | + | ! ||Pλ-luxI (86.4 µg/mL)||EcoRI||SpeI||BSA||Buffer||MilliQ||total |
|- | |- | ||
| - | |2 cuts||23.1µL||1µL||1 | + | |2 cuts||23.1 µL||1 µL||1 µL||3 µL||3 µL||1.9 µL||30 µL |
|- | |- | ||
| - | |NC||1.2 | + | |NC||1.2 µL||0 µL||0 µL||1 µL||1 µL||6.8 µL||10 µL |
|} | |} | ||
| - | *at 37°C, for 1h | + | *at 37 °C, for 1h |
| - | === | + | ===Electrophoresis=== |
<div class="experiment"> | <div class="experiment"> | ||
<span class="author">Hirano</span> | <span class="author">Hirano</span> | ||
{| class="wikitable" | {| class="wikitable" | ||
| - | !|Sample|| | + | !Lane||Sample |
| + | |- | ||
| + | |1||100bp ladder | ||
| + | |- | ||
| + | |2||Pconst+RBS-tetR-DT -(1) | ||
| + | |- | ||
| + | |3||Pconst+RBS-tetR-DT -(2) | ||
|- | |- | ||
| - | |Pconst+RBS-tetR-DT-( | + | |4||Pconst+RBS-tetR-DT -(3) |
|- | |- | ||
| - | |Pconst+RBS-tetR-DT-( | + | |5||Pconst+RBS-tetR-DT -(4) |
|- | |- | ||
| - | | | + | |6||Ptet+RBS-lacZα-DT -(1) |
|- | |- | ||
| - | | | + | |7||Ptet+RBS-lacZα-DT -(2) |
|- | |- | ||
| - | | | + | |8||100bp ladder |
|- | |- | ||
| - | |Ptet+RBS-lacZα-DT-( | + | |9||Ptet+RBS-lacZα-DT -(3) |
|- | |- | ||
| - | |Ptet+RBS-lacZα-DT-( | + | |10||Ptet+RBS-lacZα-DT -(4) |
|- | |- | ||
| - | | | + | |11||100bp ladder |
|} | |} | ||
| + | [[File:igku_Sep6_electrophoresis_N3.jpg]]<br> | ||
</div> | </div> | ||
| Line 236: | Line 243: | ||
|3 | |3 | ||
|} | |} | ||
| + | [[File:igku_Sep6_gel_extraction_N4_2.jpg]]<br> | ||
| + | [[File:igku_Sep6_gel_extraction_N4_3.jpg]]<br> | ||
{| class="wikitable" | {| class="wikitable" | ||
!Name||concentration[µg/mL]||260/280||260/230 | !Name||concentration[µg/mL]||260/280||260/230 | ||
| Line 245: | Line 254: | ||
|Pλ-luxI (EcoRI & SpeI)||4.5||1.79||0.31 | |Pλ-luxI (EcoRI & SpeI)||4.5||1.79||0.31 | ||
|} | |} | ||
| + | [[File:igku_Sep6_gel_extraction_N5_1.jpg]]<br> | ||
| + | [[File:igku_Sep6_gel_extraction_N5_2.jpg]]<br> | ||
</div> | </div> | ||
| Line 253: | Line 264: | ||
!Sample||medium | !Sample||medium | ||
|- | |- | ||
| - | |9/ | + | |9/5 Pconst-spinach-DT-(1)||Plusgrow medium(+Amp&CP) |
|- | |- | ||
| - | |9/ | + | |9/5 Ptet-pT181 antisense-(1)||Plusgrow medium(+CP) |
|- | |- | ||
| - | |9/ | + | |9/5 Pconst+RBS-tetR-DT-(1)||Plusgrow medium(+Amp&CP) |
|- | |- | ||
| - | |9/ | + | |9/5 Ptet+RBS-lacZα-DT-(1)||Plusgrow medium(+CP) |
|- | |- | ||
| - | |9/ | + | |9/5 Pcon-pT181 attenuator-(1)||Plusgrow medium(+CP) |
|- | |- | ||
| - | |9/ | + | |9/4 pT181 antisense-(1)||Plusgrow medium(+CP) |
|- | |- | ||
| - | |9/ | + | |9/4 aptamer 12_1R-(1)||Plusgrow medium(+CP) |
|} | |} | ||
</div> | </div> | ||
| Line 279: | Line 290: | ||
|9/5 Pcon-antisense-(5~12)||468 | |9/5 Pcon-antisense-(5~12)||468 | ||
|- | |- | ||
| - | |9/5 Plac- | + | |9/5 Plac-attenuator-(5~12)||664 |
|- | |- | ||
|9/5 pT181 antisense+Plac-(1)||- | |9/5 pT181 antisense+Plac-(1)||- | ||
Latest revision as of 03:56, 28 September 2013
Contents |
Sep 6
Electrophoresis
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | 9/4 RBS-lysis3-DT -3 |
| 3 | 9/4 RBS-lysis3-DT -4 |
| 4 | 9/4 RBS-lysis3-DT -5 |
| 5 | 9/4 RBS-lysis1-DT -3 |
| 6 | 9/4 Ptrc-KaiC -1 |
| 7 | 9/4 pSB4K5 -1 |
| 8 | 100bp ladder |
Colony PCR
| Sample | base pair |
|---|---|
| Pconst-spinach-DT-(1~4) | 563 |
| Plac-spinach-DT-(1~4) | 659 |
| Pcon-pT181 antisense-(1~4) | 468 |
| Ptet-pT181 antisense-(1~4) | 604 |
| Pconst-pT181 attenuator-(1~4) | 568 |
| Pconst+RBS-tetR-DT-(1~4) | 1121 |
| Ptet+RBS-lacZα-DT-(1~4) | 1162 |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 40s | 30cycles |
Liquid Culture
| Sample | medium |
|---|---|
| 9/4 RBS-Lysis3+DT3 | Plusgrow medium(+CP) |
| 9/4 RBS-Lysis1+DT3 | Plusgrow medium(+CP) |
| 9/4 pSB4K5 | Plusgrow medium(+Kan) |
Electrophoresis
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | Pconst-spinach-DT -(1) |
| 3 | Pconst-spinach-DT -(2) |
| 4 | Pconst-spinach-DT -(3) |
| 5 | Pconst-spinach-DT -(4) |
| 6 | Plac-spinach-DT -(1) |
| 7 | Plac-spinach-DT -(2) |
| 8 | Plac-spinach-DT -(3) |
| 9 | Plac-spinach-DT -(4) |
| 10 | Pcon-pT181 antisense -(1) |
| 11 | Pcon-pT181 antisense -(2) |
| 12 | Pcon-pT181 antisense -(3) |
| 13 | Pcon-pT181 antisense -(4) |
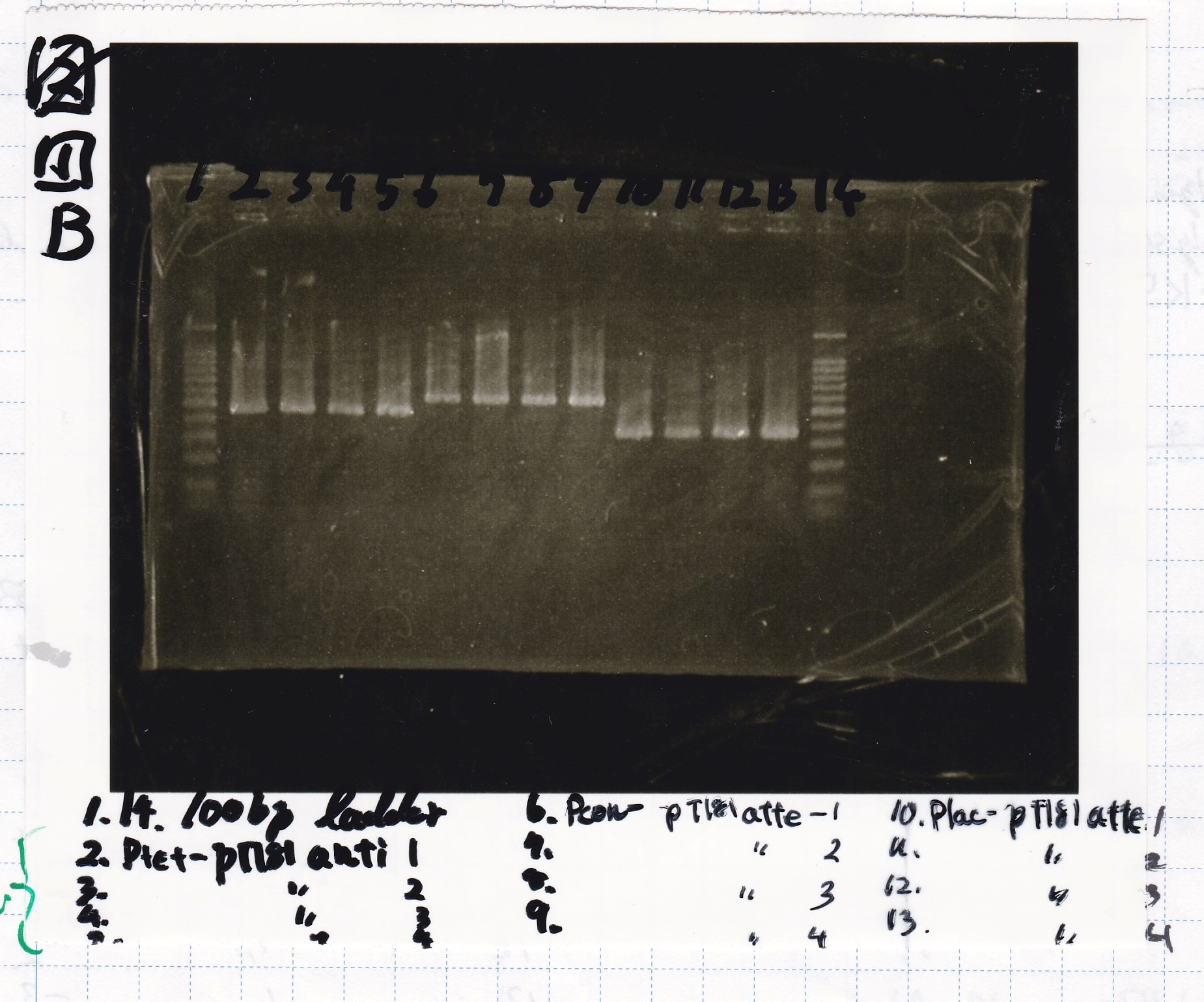
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | Ptet-pT181 antisense -(1) |
| 3 | Ptet-pT181 antisense -(2) |
| 4 | Ptet-pT181 antisense -(3) |
| 5 | Ptet-pT181 antisense -(4) |
| 6 | Plac-pT181 attenuator -(1) |
| 7 | Plac-pT181 attenuator -(2) |
| 8 | Plac-pT181 attenuator -(3) |
| 9 | Plac-pT181 attenuator -(4) |
Miniprep
| DNA | concentration [µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| pT181 antisense-(1) | 238.7 | 1.68 | 1.44 |
| pT181 antisense-(2) | 248.8 | 1.83 | 1.77 |
| pT181 antisense-(3) | 223.2 | 1.78 | 1.61 |
| pT181 antisense-(4) | 220.8 | 1.89 | 1.92 |
| tetR aptamer-(1) | 252.5 | 1.89 | 1.90 |
| tetR aptamer-(2) | 211.2 | 1.90 | 1.99 |
| DT181 attenuator | 102.2 | 1.98 | 2.17 |
| 121R aptamer-DT | 251.7 | 1.92 | 2.01 |
| pSB4K5 | 212.9 | 1.80 | 1.90 |
| RBS-lacZα-DT | 185.7 | 1.78 | 1.79 |
| RBS-lysis2-DT-(1) | 84.8 | 1.85 | 2.24 |
Restriction Enzyme Digestion
| 9/6 RBS-lysis2-DT (78.3 µg/mL) | XbaI | PstI | BSA | Buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 22 µL | 1 µL | 1 µL | 3 µL | 3 µL | 0 µL | 30 µL |
| NC | 1.3 µL | 0 µL | 0 µL | 1 µL | 1 µL | 6.7 µL | 10 µL |
| aptamer-DT (251.7 µg/mL) | XbaI | PstI | BSA | Buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 0.4 µL | 1 µL | 1 µL | 3 µL | 3 µL | 14.1 µL | 30 µL |
| NC | 0.4 µL | 0 µL | 0 µL | 1 µL | 1 µL | 7.6 µL | 10 µL |
| Pλ-luxI (86.4 µg/mL) | EcoRI | SpeI | BSA | Buffer | MilliQ | total | |
|---|---|---|---|---|---|---|---|
| 2 cuts | 23.1 µL | 1 µL | 1 µL | 3 µL | 3 µL | 1.9 µL | 30 µL |
| NC | 1.2 µL | 0 µL | 0 µL | 1 µL | 1 µL | 6.8 µL | 10 µL |
- at 37 °C, for 1h
Electrophoresis
| Lane | Sample |
|---|---|
| 1 | 100bp ladder |
| 2 | Pconst+RBS-tetR-DT -(1) |
| 3 | Pconst+RBS-tetR-DT -(2) |
| 4 | Pconst+RBS-tetR-DT -(3) |
| 5 | Pconst+RBS-tetR-DT -(4) |
| 6 | Ptet+RBS-lacZα-DT -(1) |
| 7 | Ptet+RBS-lacZα-DT -(2) |
| 8 | 100bp ladder |
| 9 | Ptet+RBS-lacZα-DT -(3) |
| 10 | Ptet+RBS-lacZα-DT -(4) |
| 11 | 100bp ladder |
Gel Extraction
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 100bp ladder | -- |
| 2 | RBS-lysis2-DT | XbaI & PstI |
| 3 | ||
| 4 | -- | -- |
| 5 | aptamer 12_1R-DT | XbaI & PstI |
| 6 |
| Lane | DNA | Enzyme |
|---|---|---|
| 1 | 100bp ladder | -- |
| 2 | Pλ-luxI | EcoRI & SpeI |
| 3 |
| Name | concentration[µg/mL] | 260/280 | 260/230 |
|---|---|---|---|
| RBS-lysis2-DT (XbaI & PstI) | 7.1 | 1.88 | 0.07 |
| aptamer 12_1R-DT (XbaI & PstI) | 15.3 | 1.93 | 0.70 |
| Pλ-luxI (EcoRI & SpeI) | 4.5 | 1.79 | 0.31 |
Liquid Culture
| Sample | medium |
|---|---|
| 9/5 Pconst-spinach-DT-(1) | Plusgrow medium(+Amp&CP) |
| 9/5 Ptet-pT181 antisense-(1) | Plusgrow medium(+CP) |
| 9/5 Pconst+RBS-tetR-DT-(1) | Plusgrow medium(+Amp&CP) |
| 9/5 Ptet+RBS-lacZα-DT-(1) | Plusgrow medium(+CP) |
| 9/5 Pcon-pT181 attenuator-(1) | Plusgrow medium(+CP) |
| 9/4 pT181 antisense-(1) | Plusgrow medium(+CP) |
| 9/4 aptamer 12_1R-(1) | Plusgrow medium(+CP) |
Colony PCR
| Sample | base pair |
|---|---|
| 9/5 Plac-spinach-DT-(5~12) | 563 |
| 9/5 Pcon-antisense-(5~12) | 468 |
| 9/5 Plac-attenuator-(5~12) | 664 |
| 9/5 pT181 antisense+Plac-(1) | - |
| PreDenature | Denature | Annealing | Extension | cycle |
|---|---|---|---|---|
| 94°C | 94°C | 55°C | 68°C | -- |
| 5min | 30s | 30s | 42s | 30cycles |
 "
"