Team:Virginia/Applications
From 2013.igem.org
Polysialic Acid

The K1 strain of E. coli produces and encapsulates itself in α-2,8-linked polysialic acid (PSA), a polysaccharide that biochemically mimics the human glycoprotein Neural Cell Adhesion Molecule (NCAM). This similarity renders virulent K1 E. coli poorly immunogenic in humans because they aren’t seen as foreign and are therefore cloaked from antibody opsonization and complement-mediated killing. Clearly, equipping our minicells with such protection would be highly advantageous for improving their successful delivery rate and reducing harmful immune responses.

A significant amount of machinery (14 genes in total) is required to generate the PSA capsule in K1 E. coli and transferring this ability over to the comparatively docile K12 E. coli is quite the feat. Thankfully, we were able to find previous work by Dr. Eric Vimr that accomplished just that and obtain a single pSR23 plasmid that contains all the required genes. We are currently in the process of confirming the presence of PSA on our K12 E. coli via antibody-labeled fluorescence microscopy.
Sources:
Ail
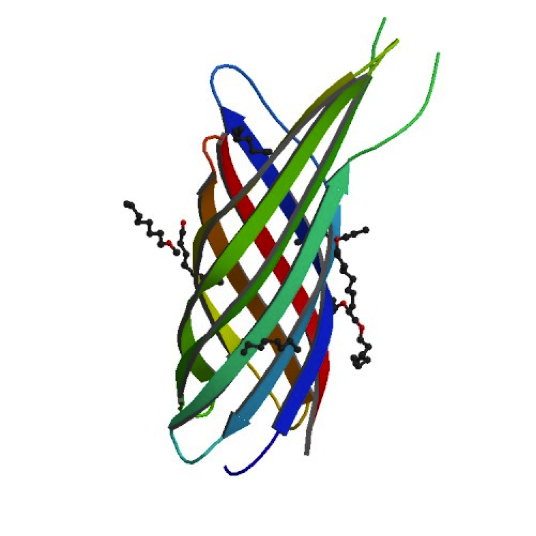
Ironically, in order to make our minicells as non-toxic and safe as possible we had to look toward the likes of the bacteria that caused the Black Plague. While it may seem strange, bacteria that have such success when infecting the human body have developed the best ways to avoid the traps of the immune system. For our innocuous minicells, the only source of harm to the body would come from an overreaction of its own immune system. Therefore, equipping our minicells with the immunoevasive tools of virulent bacteria actually makes them less harmful.

Our research led us to the Ail protein from Yersinia enterocolitica. Ail is just one of three invasion proteins cloned from Yersinia species, invasin probably being the most widely known. When present on E. coli, Ail confers a phenotype of attachment and invasion, though invasion is primarily limited to epithelial tissue. These qualities already make Ail an attractive protein for enhancing minicells role as a delivery vehicle, but they are not the primary reasons we chose to use it. Ail was also found to have both structural and functional homology with outer membrane proteins like Rck of Salmonella typhimurium, which grants high-level serum resistance. The serum resistance Ail grants to E. coli helps prevent the activation of the complement cascade—a primary defense mechanism of the immune system. The binding of complement proteins to bacteria usually spells their end: it triggers inflammation, draws phagocytic cells, facilitates phagocytosis, induces cell lysis, and coordinates opsonization by antibodies. By protecting our minicells from destruction, we can improve the chances of successful payload delivery and simultaneously reduce the risk of septic shock.
Sources:
Targeting

Previous researchers who have worked on minicells have attached Protein A/G Fc receptors to an OmpA autotransporter located on the surface of minicells. This patented approach allowed them to add antibodies specific to their target host cell to purified minicells, thereby creating targeted minicells. Additionally, the antibody coating serves to protect the minicells from the host immune system and induce endocytosis when binding to certain target cell receptors. Such a modular targeting system is ideal for such a versatile vector. We hope to create a similar system that allows our minicells utilize the vast library of commercial antibodies available as highly specific receptors for a variety of applications.
Sources:
 "
"
