Team:Bielefeld-Germany/Project/GldA
From 2013.igem.org
GldA
Glycerol dehydrogenase GldA - Overview
Mediators are essential for the use of Escherichia coli in Microbial Fuel Cells. The main goal when improving MFCs is to enhance the kinetics of the electron transfer between the bacterial cells and the Fuel Cell anode. Increasing the mediator concentration in the MFC is an efficient way to enhance electron transfer. In order to avoid the usage of expensive and toxic synthetic mediators, we engineered an E. coli KRX strain to overexpress the glycerol dehydrogenase (GldA). GldA produces the endogenous mediator NADH from NAD+ and glycerol, which is the main carbon source in our medium. We were able to produce high amounts of NADH with the optimized E.coli, which resulted in a more efficient electron transfer. This demonstrates that genetically introducing an appropriate oxidoreductase into E. coli via gene manipulation can greatly improve the mediator production and power generation. We could show that the increased intracellular- and extracellular NADH concentration, leads to a 20 % enhanced current production in our Microbial Fuel Cell. The overexpression of the glycerol dehydrogenase in Escherichia coli is a great genetic optimization for electron shuttle-mediated extracellular electron transfer from bacteria to electrodes.
Theory
Escherichia coli is a readily available and easily grown bacterium. It has become a popular biocatalyst used in MFCs, despite of its inability to conduct electricity by nature (Park et al., 2000). One way of transferring electrons generated from redox reactions inside E. coli cells to the anode is using mediators. Mediators shuttle the electrons from inside of the bacterial cell to the anode. There are several drawbacks to using exogenous mediators (e.g. neutral red or methylene blue), such as their price, short lifetime and toxicity to the microorganisms (Seopet al., 2006). However, when the bacteria produce their own mediators, the system can operate at a high, sustainable level of activity. Such a system is called mediator-less Microbial Fuel Cell, because exogenous mediators do not need to be added. Thus they are of great importance in MFC applications.
To significantly improve the MFC performance, a large amount of the bacteria-excreted mediators must be generated. Because of the already high metabolic stress this is a great challenge to the cell (Qiao et al., 2008).
Therefore our plan was it to overexpress the homologous glycerol dehydrogenase (GldA) from Escherichia coli in order to minimize the metabolic discrepancies which would be caused by genes foreign to the species. Most of the GldA products are small water-soluble redox molecules, which have properties similar to mediators (Kelley and Dekker, 1985). GldA has a broad substrate specificity and is involved in different metabolic pathways. The main function of this oxidoreductase (EC 1.1.1.6) is the catalyzation of the chemical reaction glycerol + NAD+ --> glycerone + NADH + H+ with NADH as the main endogenous mediator.
NAD+ is reduced to NADH by addition of two electrons (e-) and one proton (H+) and therefore very suitable as an electron mediator. (Ruzheinikov et al., 2001)
Overexpression of the glycerol dehydrogenase GldA from Escherichia coli will enhance the amount of mediator within the MFC and lead to an improved electron shuttle-mediated extracellular electron transfer. This results in a more efficient electricity generation by the microorganisms.
Genetic Approach
The gldA gene from Escherichia coli was cloned and overexpressed in E. coli KRX under the control of different promoters (Table 1).

Figure 3: pSB1C3 – <bbpart>BBa_K1172201</bbpart> GldA BioBrick (1104 bp) was examined by restriction analysis and sequencing. Map was designed with LabGenius Plasmid Mapper.
Results
Upon the expression of the glycerol dehydrogenase, the endogenous mediator production of Escherichia coli was measured. SDS-PAGE combined with MALDI-TOF MS/MS and different NADH-assays were performed to characterize the GldA BioBrick <bbpart>BBa_K1172201</bbpart>.
SDS-PAGE and MALDI-TOF
SDS-PAGE shows the protein glycerol dehydrogenase at the expected size of 40 kDa. In contrast to the Escherichia coli KRX wild type, the GldA protein under the control of the weak constitutive Anderson promoter (<bbpart>BBa_K1172205</bbpart>) can be seen by an only slightly stronger band. When the T7 (<bbpart>BBa_K1172203</bbpart>) and lac (<bbpart>BBa_K1172204</bbpart>) promoter are used a strong band can be seen, which is equated with a strong expression and overproduction of GldA.
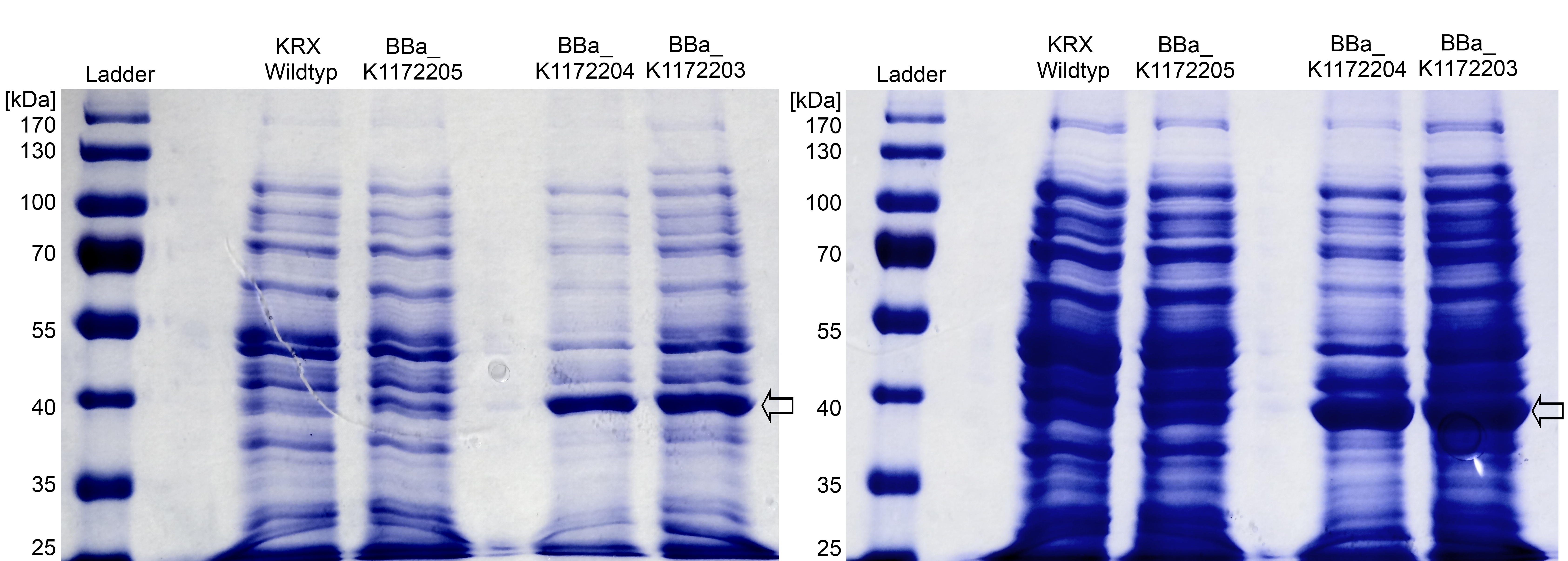
Figure 4: SDS-PAGE with [http://www.thermoscientific.com/ecomm/servlet/productsdetail_11152___13576050_-1 Prestained Protein Ladder from Thermo Scientific] as marker. Comparison of protein expression between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172205</bbpart>, <bbpart>BBa_K1172204</bbpart> and <bbpart>BBa_K1172203</bbpart> after total cell disruption. The right-hand gel was loaded with a higher protein concentration. The SDS-PAGEs display the protein glycerol dehydrogenase at the expected size of 40 kDa and enhanced overproduction of the protein with increasing promoter strength.
Furthermore, we were able to identify the overexpressed glycerol dehydrogenase (Figure 4) with MALDI-TOF MS/MS.
Tryptic digest of the gel lane for analysis with MALDI-TOF could confirm the identity of the strong band as the glycerol dehydrogenase with a Mascot Score of 266 against the Escherichia coli database.
Growth characteristics
In order to test the effect of the protein expression on the cell growth, growth was measured as increase in optical density and plotted against time.

Figure 5: Growth curves for E. coli KRX with LB medium. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172201</bbpart>, <bbpart>BBa_K1172203</bbpart> and <bbpart>BBa_K1172204</bbpart>. GldA gene expression was induced for inducable promoters at OD600 = 1.0. All data are representing two biological and two technical replicates with standard deviation.
E. coli KRX wild type shows the best growth characteristics with a maximal optical density OD600 = 4.0. E. coli KRX with gldA coding plasmid shows a slightly lower growth due to the higher replication stress. E. coli KRX expressing gldA under control of lac promoter shows 14% decreased maximal cell density and E. coli KRX expressing gldA under control of T7 promoter shows a further decreased growth which was 45% reduced in comparison to E. coli KRX wild type. This lower growth is based on an increased expression of gldA and therefore on an increased NADH production, which was confirmed with different NADH assays.
NADH-Assays
An overproduction of the glycerol dehydrogenase results in an oversupply of products from the glycerol dehydrogenase. Glycerol dehydrogenase produces several mediators because of its broad substrate specificity. GldA is involved in several metabolic pathways, for example in the glycerol metabolism, which converts glycerol and NAD+ to glycerone, NADH and H+. NADH is a small, water-soluble redoxmolecule, which appears to be a great mediator for Microbial Fuel Cells. An overexpression of the glycerol dehydrogenase leads to an overproduction of NADH and therefore to an endogenous mediator production. Furthermore, GldA seems to be very interesting in combination with our preferred MFC carbon source glycerol. Thus, it is essential to get reliable data on the NADH overproduction to see how many electron shuttles are available and how efficient the electron shuttle-mediated electron transfer (EET) will be.
We could observe an enhanced NADH production in comparison to the wild type for Escherichia coli KRX containing pSB1C3 with <bbpart>BBa_K1172203</bbpart>, <bbpart>BBa_K1172204</bbpart> and <bbpart>BBa_K1172205</bbpart> with increasing promoter strength. (Table 2 and Figure 6)

Table 2: Results of the NADH-assay. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172205</bbpart>, <bbpart>BBa_K1172204</bbpart> and <bbpart>BBa_K1172203</bbpart>. NADH concentration with standard deviation and concentration in comparison to the wild type is shown. Two biological and at least three technical replicates were analyzed.
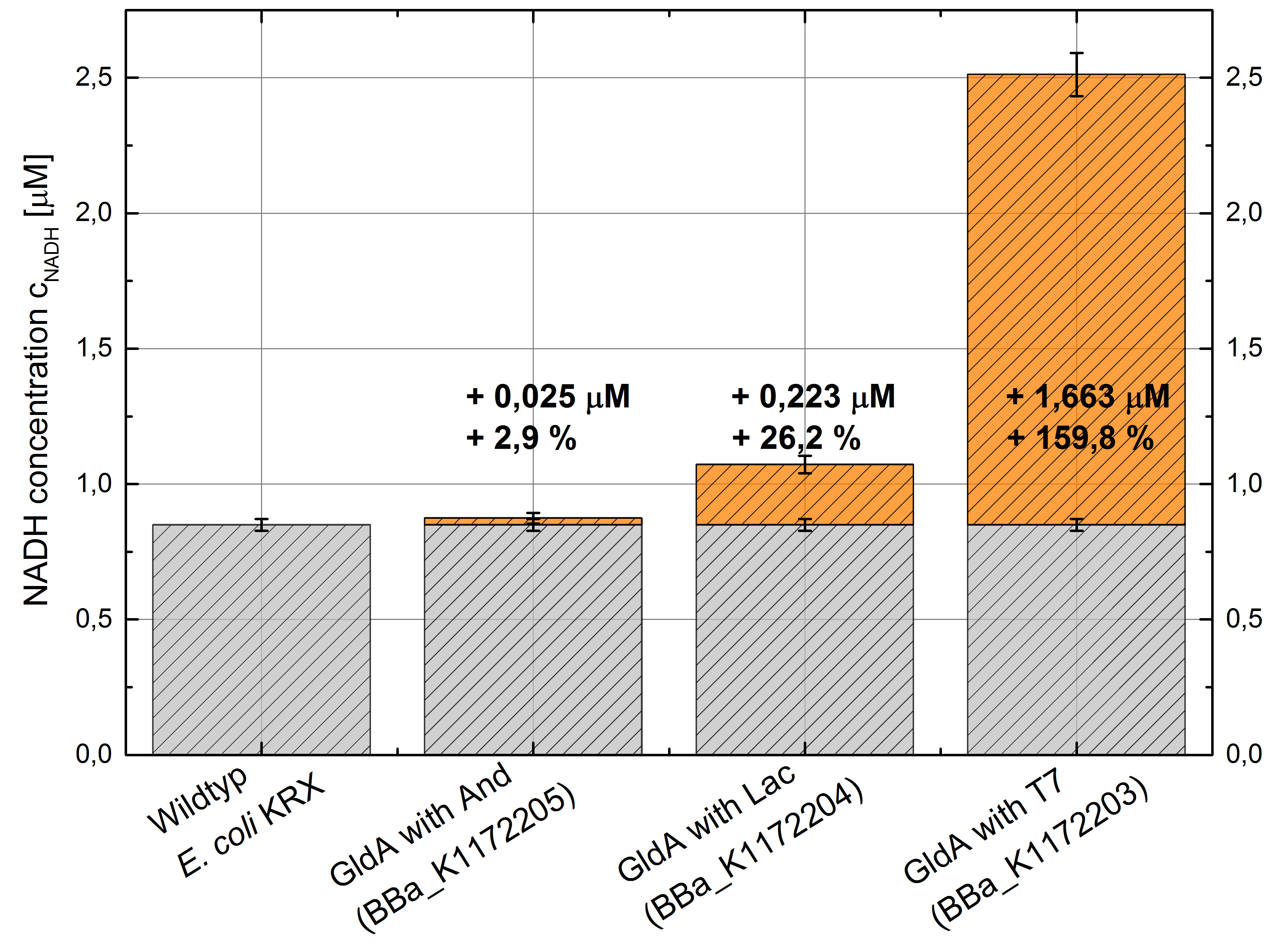
Figure 6: Column chart with results of the NADH-assay. Comparison between Escherichia coli KRX wild typ and Escherichia coli KRX with <bbpart>BBa_K1172205</bbpart>, <bbpart>BBa_K1172204</bbpart> and <bbpart>BBa_K1172203</bbpart>. NADH concentration with standard deviation and concentration in comparison to the wild type is shown. Two biological and at least three technical replicates were analyzed.
Compared to the Escherichia coli KRX wild type, Escherichia coli carrying gldA and T7 promoter (<bbpart>BBa_K1172203</bbpart>) shows 1.7 μM NADH overproduction and gldA with lac promoter (<bbpart>BBa_K1172204</bbpart>) shows 0.2 μM NADH overproduction. The gldA expression by the Anderson promoter (<bbpart>BBa_K1172205</bbpart>) is too weak for a significant NADH overproduction.
For all following tests, E. coli with gldA under control of the T7 promoter (<bbpart>BBa_K1172203</bbpart>) was used, because this strain by far shows the highest NADH production rate.
Prior MFC tests showed, that glycerol is the best carbon source for growth of Escherichia coli under anaerobic respiration in our Microbial Fuel Cell. Therefore it is important to see, which effect an increased glycerol concentration has on the mediator production. To test these parameters, LB medium was supplemented with different amounts of glycerol (Table 3).
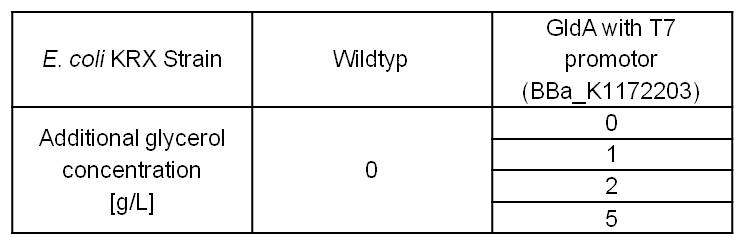
Table 3: Experimental design of the glycerol dependent NADH-assay. Supplementation of different amounts of glycerol to LB medium should show the effect of glycerol on NADH overproduction.
E.coli (<bbpart>BBa_K1172203</bbpart>) grown in glycerol-supplemented medium shows enhanced NADH production in contrast to the Escherichia coli KRX wild type and also in contrast to the Escherichia coli KRX with <bbpart>BBa_K1172203</bbpart> and no further glycerol supplementation (Table 4 and Figure 7).
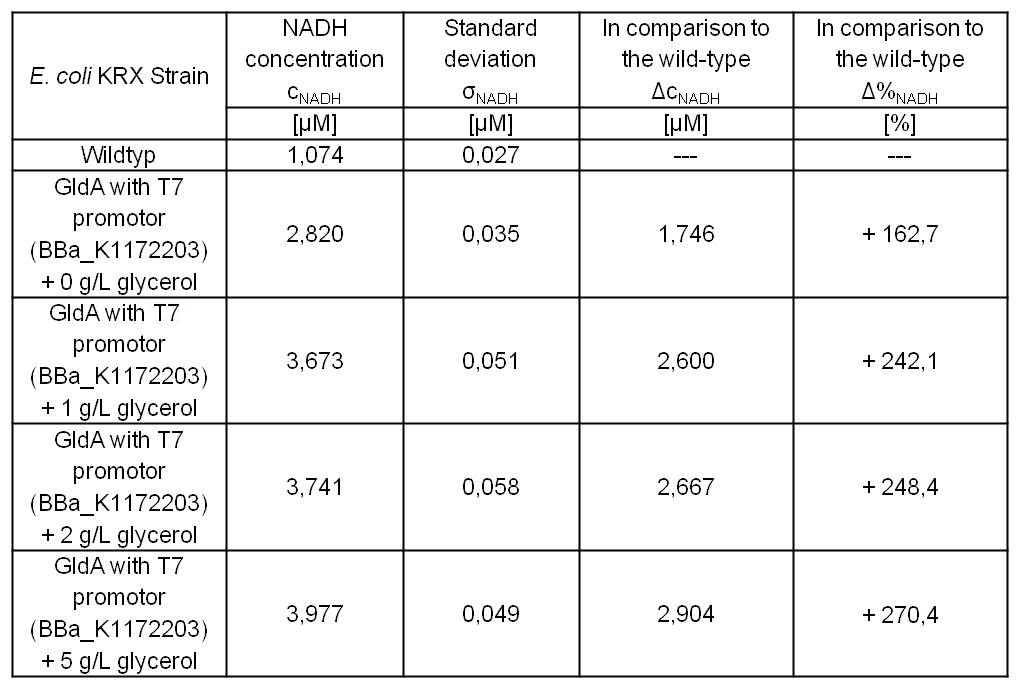
Table 4: Results of the NADH-assay. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172203</bbpart> supplemented with different amounts of glycerol to LB medium. NADH concentration with standard deviation and concentration in comparison to the wild type is shown. Two biological and at least three technical replicates were analyzed.
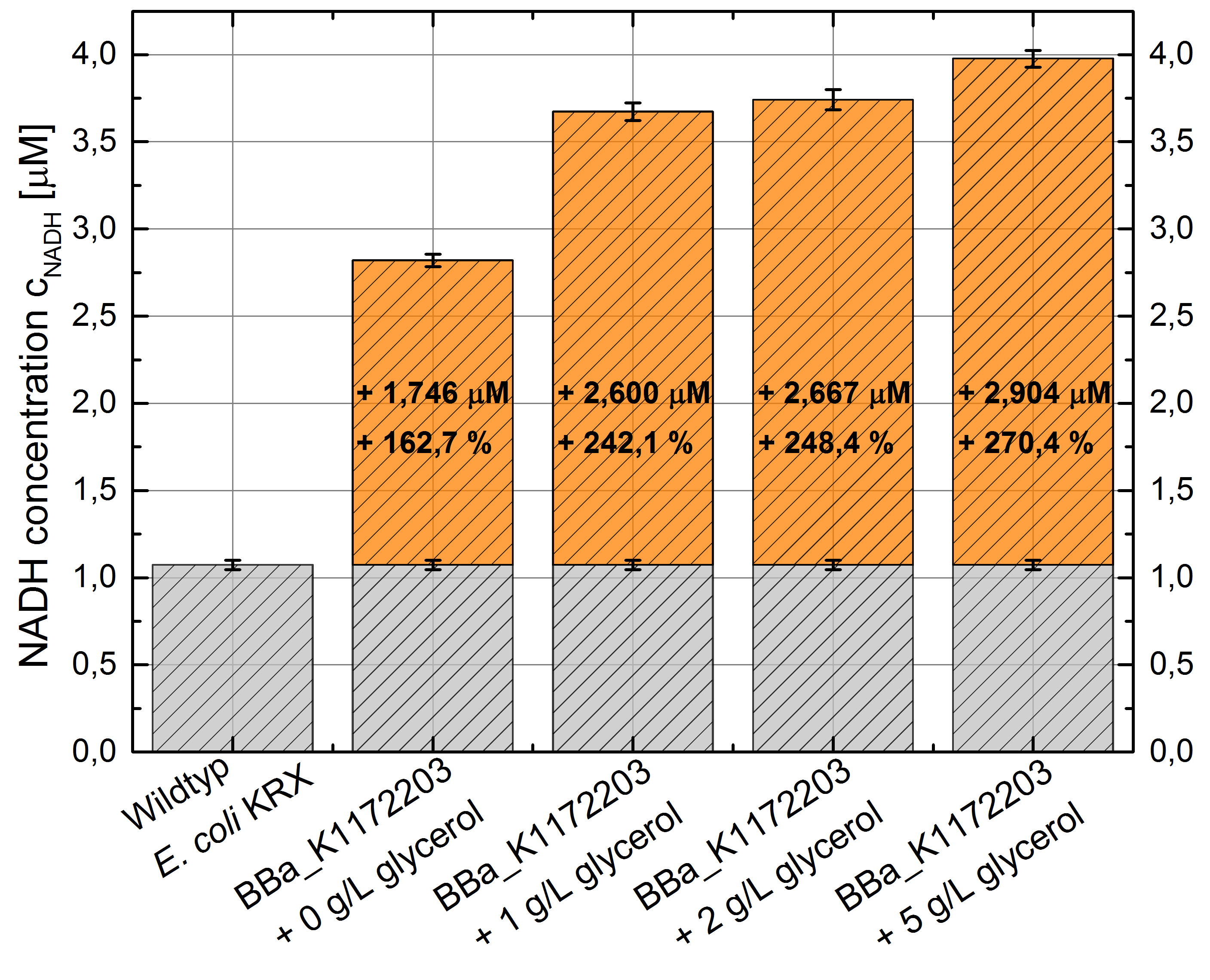
Figure 7: Column chart with results of the NADH-assay. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172203</bbpart> supplemented with different amounts of glycerol to LB medium. NADH concentration with standard deviation and concentration in comparison to the wild type is shown. Two biological and at least three technical replicates were analyzed.
The glycerol dependent NADH-assay shows the expected NADH overproduction for <bbpart>BBa_K1172203</bbpart> when supplemented with glycerol. It was possible to increase the NADH production by 270 % in comparison to the Escherichia coli KRX wild type and up to 100 % in comparison to the E. coli KRX carrying <bbpart>BBa_K1172203</bbpart>, without glycerol supplementation. However, the supplemented glycerol concentration does not have a considerable impact on the NADH production. These characteristics in production improvement are initially only valid for LB medium with glycerol supplementation.
With previous NADH-assays we only observed the effects of GldA on the intracellular NADH concentration of E. coli. To additionally investigate the extracellular NADH concentrations, we tested the Escherichia coli KRX wild type strain and the E. coli KRX with pSB1C3 and <bbpart>BBa_K1172203</bbpart> by cultivation with different glycerol concentrations in M9 medium. (Table 5)

Table 5: Experimental design of the glycerol dependent NADH-assay. Different concentrations of glycerol in M9 medium should show the effect of glycerol on NADH overproduction.
Cultivating Escherichia coli <bbpart>BBa_K1172203</bbpart> with increasing glycerol concentration in M9 medium shows enhanced intracellular and also enhanced extracellular NADH concentrations in contrast to the Escherichia coli KRX wild type. (Table 6 and Figure 8)

Table 6: Results of the NADH-assay. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172203</bbpart> grown in M9 medium with different concentrations of glycerol. NADH concentration with standard deviation and concentration in comparison to the wild type is shown for cell disruption and supernatant fraction. Two biological and at least three technical replicates were analyzed.

Figure 8: Column chart with results of the NADH-assay. Comparison between Escherichia coli KRX wild type and Escherichia coli KRX with <bbpart>BBa_K1172203</bbpart> grown in M9 medium with different concentrations of glycerol. NADH concentration with standard deviation and concentration in comparison to the wild type is shown for cell disruption (Intracellular) and supernatant (Extracellular) fraction. Two biological and at least three technical replicates were analyzed.
In addition to the previously observed increase in intracellular NADH concentration, we were able to show an enhanced extracellular NADH concentration. Homologous expression of GldA <bbpart>BBa_K1172203</bbpart> in E. coli seems to be an appropriate way for endogenous mediator production. Furthermore glycerol could be confirmed as a suitable substrate for the NADH production.
Intracellular NADH concentration increases up to 223 % in comparison to Escherichia coli KRX wild type with increasing promoter strength. It is also very interesting to see that the extracellular NADH concentration is 7 times higher than the intracellular concentration for all strains. Overexpression of GldA <bbpart>BBa_K1172203</bbpart> in E. coli causes a 2.5 times higher extracellular NADH concentration compared with Escherichia coli KRX wild type.
However, it is surprising that the extracellular concentration of NADH is 7 times higher than the intracellular concentration. Therefore, we cannot be sure that the complete extracellular fluorescence is only due to the endogenous mediator NADH. There might be a substance that is directly or indirectly produced with the overexpression of GldA and secreted into the medium. Thus, we will analyze the extracellular fraction with LC ESI MS/MS, in order to identify potential compounds.
These data show that NADH is an adequate mediator for Microbial Fuel Cells. NADH can be transported across the cell membrane, which is indicated by much higher extracellular NADH concentration and thus allow NADH-mediated electron transfer (EET).
Microbial Fuel Cell Measurement
All NADH-assays showed great enhanced NADH overproduction for the GldA strain. Therefore higher electron transport efficiency should result in an improved bioelectricity output.
For testing the genetic engineered system in the Microbial Fuel Cell, we used Escherichia coli KRX with gldA and lac promoter (<bbpart>BBa_K1172204</bbpart>) in contrast to the Escherichia coli KRX wild type. Certainly NADH-assays determined Escherichia coli KRX with gldA and T7 promoter (<bbpart>BBa_K1172203</bbpart>) as the best endogenous mediator producing strain. Unfortunately we could not use this strain due to cultivation problems.
According to our assumptions, the extracellular electron transfer mediated by NADH is improved in the GldA strain resulting in an increased bioelectricity output. (Figure 9 and 10)

Figure 9: Microbial Fuel Cell results from cultivation of Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) in contrast to Escherichia coli KRX wild type. Voltage curve from Escherichia coli KRX wild type and Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) is shown over time over a resistance of 200 Ω. M9-medium was used with no supplementation of mediators.

Figure 10: Microbial Fuel Cell results from cultivation of Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) in contrast to Escherichia coli KRX wild type. Electric charge curve from Escherichia coli KRX wild type and Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) is shown over time over a resistance of 200 Ω. M9-medium was used with no supplementation of mediators.

Figure 11: Microbial Fuel Cell results from cultivation of Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) in contrast to Escherichia coli KRX wild type. Average electric power bar chart from Escherichia coli KRX wild type and Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>). M9-medium was used with measurement over a resistance of 200 Ω.
Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>) shows a 27 % higher maximal voltage than the Escherichia coli KRX wild type. Over the whole cultivation, voltage was improved by about 20%. The calculation of the electric charge confirms the described results. Electric charge is equivalent to the number of transported electrons and 20 % enhanced for Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>). The maximal electric charge of 135 mC shows that the overexpression of the glycerol dehydrogenase enables endogenous mediator production and a more efficient electron shuttle-mediated electron transfer.
Finally, the calculated electric power bar chart shows 40% enhanced average electric power for Escherichia coli KRX with gldA (<bbpart>BBa_K1172204</bbpart>).
A further optimization by using Escherichia coli KRX with gldA and T7 promoter (<bbpart>BBa_K1172203</bbpart>) instead of lac promoter (<bbpart>BBa_K1172203</bbpart>) would improve electricity output according to the NADH-assay results (Table 2 and Figure 6) of about 134 %. These results demonstrate that the redox mediator NADH from Escherichia coli facilitates the electron transfer between cell and electrode and shows a starting point for reducing costs of expensive and toxic exogenous mediators.
Conclusion
Mediators are essential for the use of Escherichia coli in Microbial Fuel Cells. The main goal of improving MFCs is to enhance kinetics of the electron transfer between the bacterial cells and the fuel cell anode. In order to decrease the usage of expensive and toxic synthetic mediators (exogenous mediators like methylene blue or neutral red), we produced the endogenous mediator NADH by overexpression of the glycerol dehydrogenase.
Looking at our project in its entirety, the overproduction of the glycerol dehydrogenase seems to be a great way for endogenous mediator production. The use of glycerol as our main carbon source for Escherichia coli in the MFC further enhances the efficiency of NADH production. We can show a 270 % higher intracellular NADH concentration (4 μM) for Escherichia coli KRX with GldA plasmid and a 170 % higher extracellular NADH concentration (24 μM) in contrast to the Escherichia coli KRX wild type.
The overexpression of GldA is a great genetic strategy to optimize mediator production as well as electricity generation in Microbial Fuel Cells. The most suitable and efficient GldA device for Escherichia coli is a combination with the rhamnose inducible T7 promoter (<bbpart>BBa_K1172203</bbpart>).
References
Kelley JJ, Dekker EE (1985) Identity of Escherichia coli D-1-amino-2-propanol: NAD+ oxidoreductase with E. coli glycerol dehydrogenase but not with Neisseria gonorrhoeae 1,2-propanediol:NAD+ oxidoreductase. [http://jb.asm.org/content/162/1/170.short| J. Bacteriol. 162: 170].
Mulichak AM (2005) Crystal structure of glycerol dehydrogenase. [http://www.rcsb.org/pdb/images/1ta9_bio_r_500.jpg| Research Collaboratory for Structural Bioinformatics (RCSB) Protein Data Band (PDB)].
Park DH, Kim SK, Shin IH, Jeong YJ (2000) Electricity production in biofuel cell using modified graphite electrode with Neutral Red. [http://link.springer.com/article/10.1023/A:1005674107841#page-1| Biotechnol. Lett 22: 1301].
Qiao Y, Li CM, Bao SJ, Lu ZS, Hong YH (2008) Electrocatalysis in microbial fuel cells - from electrode material to direct electrochemistry. [http://pubs.rsc.org/en/content/articlehtml/2010/ee/b923503e| Chem. Commun. 1290].
Ruzheinikov SN, Burke J, Sedelnikova S, Baker PJ, Taylor B, Bullough PA, Muir NM, Gore MG, Rice DW (2001) Glycerol Dehydrogenase: Structure, Specificity, and Mechanism of a Family III Polyol Dehydrogenase. [http://www.sciencedirect.com/science/article/pii/S0969212601006451|Structure 9: 789–802].
Seop CI, Moon H, Bretschger O, Jang JK, Park HI, Nealson KH, Kim BH (2006) Electrochemically Active Bacteria (EAB) and Mediator-Less Microbial Fuel Cells. [http://www.microbialfuelcell.org/Publications/EBL/1-10/8_2006_Chang%20I.%20S_%20Electrochemically%20active%20bacteria%20%28EAB%29%20and%20mediator-less%20microbial%20fuel%20cells.pdf|J. Microbiol. Biotechnol. 16 (2): 163–177].
Xiang K, Qiao Y, Ching CB, Li CM (2009) GldA overexpressing-engineered E. coli as superior electrocatalyst for microbial fuel cells. [http://www.sciencedirect.com/science/article/pii/S1388248109002835| Electrochemistry Communications 11: 1593–1595].
Zhang T, Cui C, Chen S, Ai X, Yang H, Shen P, Peng Z (2006) A novel mediatorless microbial fuel cell based on direct biocatalysis of Escherichia coli. [http://pubs.rsc.org/en/content/articlehtml/2006/cc/b600876c| Chem Commun 11: 2257–2259].
 "
"





