Team:BYU Provo/Notebook/SmallPhage/Summerexp/9.13 Mutagen Concentration Test
From 2013.igem.org
| (10 intermediate revisions not shown) | |||
| Line 12: | Line 12: | ||
|- valign="top" | |- valign="top" | ||
| - | | style="width: | + | | style="width: 22%; background-color: transparent;"| |
<font color="#333399" size="3" font face="Calibri"> | <font color="#333399" size="3" font face="Calibri"> | ||
| Line 86: | Line 86: | ||
* Store the supernatants at 4 Celsius. | * Store the supernatants at 4 Celsius. | ||
| - | 3) Cesium Chloride Gradients | + | 3) Cesium Chloride Gradients (9.16) |
* The phage purification team ran 3 cesium chloride gradients: | * The phage purification team ran 3 cesium chloride gradients: | ||
| Line 93: | Line 93: | ||
: - One that selects for large phage | : - One that selects for large phage | ||
| - | * For their procedure, go here | + | * For their procedure, go here [[Team:BYU_Provo/Notebook/Phage_Purification/Fallexp/Period1/Exp/9.16CsClGradient|9.16 CsCl Gradient]] |
4) Spot Test of Gradient Samples (9.18) | 4) Spot Test of Gradient Samples (9.18) | ||
| Line 115: | Line 115: | ||
6) Plating to Check for Plaque Size 2 (9.23) | 6) Plating to Check for Plaque Size 2 (9.23) | ||
| - | 7) Checking for Plaque Viability from Step 5 | + | * We redid the plating from step 5, but this time added a -3 dilution for some tubes (Small 11-13 and Large 3) because no plaques had formed at -4 for them. We also used x4 agar while plating. |
| + | |||
| + | 7) Checking for Plaque Viability from Step 5 (9.23) | ||
* We selected 4 plaques (looked bigger than normal) from the small phage plates and 6 plaques from the large phage plates (looked smaller than normal) from the plates in step 5. | * We selected 4 plaques (looked bigger than normal) from the small phage plates and 6 plaques from the large phage plates (looked smaller than normal) from the plates in step 5. | ||
| - | * We picked each plaque with a pipet tip and placed them each in eppendorf | + | * We picked each plaque with a pipet tip and placed them each in eppendorf tubes with 100mL of LB. |
* We created a -1 and -2 dilution series for each plaque. | * We created a -1 and -2 dilution series for each plaque. | ||
| - | * We then | + | * We then put 0.5mL of E. coli B into 18 test tubes and infected each tube with 20uL of phage from the -1 and -2 dilution series tubes for about 10 minutes. |
| + | * We then added 6mL of x4 top agar to each tube and plated it. | ||
| + | |||
| + | * Allowed them to incubate for 19 hours at 37 Celsius. | ||
| + | |||
| + | 8) Checking for Plaque Viability 2 (9.25) | ||
| + | |||
| + | * From the plates made in step 6, we selected more large and small plaques. We followed the same procedure as step 7 in the selection process, except we only made a -2 dilution. In total, we had 21 plaques that looked really large and 10 plaques that looked really small. | ||
| Line 131: | Line 140: | ||
2) Applying the mutagen | 2) Applying the mutagen | ||
| - | * The OD reading was | + | * The OD reading was 0.435A, which indicates there are 2.175E8 bacteria/ml. Because there are 10ml in each tube, there is roughly 2.175E9 bacteria per tube. |
| + | 3) Cesium Chloride Gradients | ||
| + | |||
| + | * The cesium chloride gradient didn't have a visible band. Therefore, we will rely on spot tests of the gradient to determine where the phage are located. | ||
| + | |||
| + | 4) Spot Test of Gradient Samples | ||
| + | |||
| + | * For the 2nd gradient, which selects for small phage, 11 tubes went to -4, while tubes 4, 6, 7, 8, 9 only went to -2. This tells us that our mutant phage are probably in tubes 2-5. | ||
| + | |||
| + | [[File:SpotTestSmall.JPG|300px|center]] | ||
| + | |||
| + | * For the 3rd gradient, which selects for large phage, 12 tubes went to -4, while tubes 5, 6, and 7 only went to -2. This tells us that our mutant phage are probably in tubes 10-15. | ||
| + | |||
| + | [[File:SpotTestLarge.JPG|300px|center]] | ||
| + | |||
| + | 5) Plating to Check for Plaque Size | ||
| + | |||
| + | * These are the plates that we selected plaques from: | ||
| + | |||
| + | [[File:5Small.JPG|300px|center]] | ||
| + | |||
| + | |||
| + | |||
| + | [[File:5Large.JPG|300px|center]] | ||
| + | |||
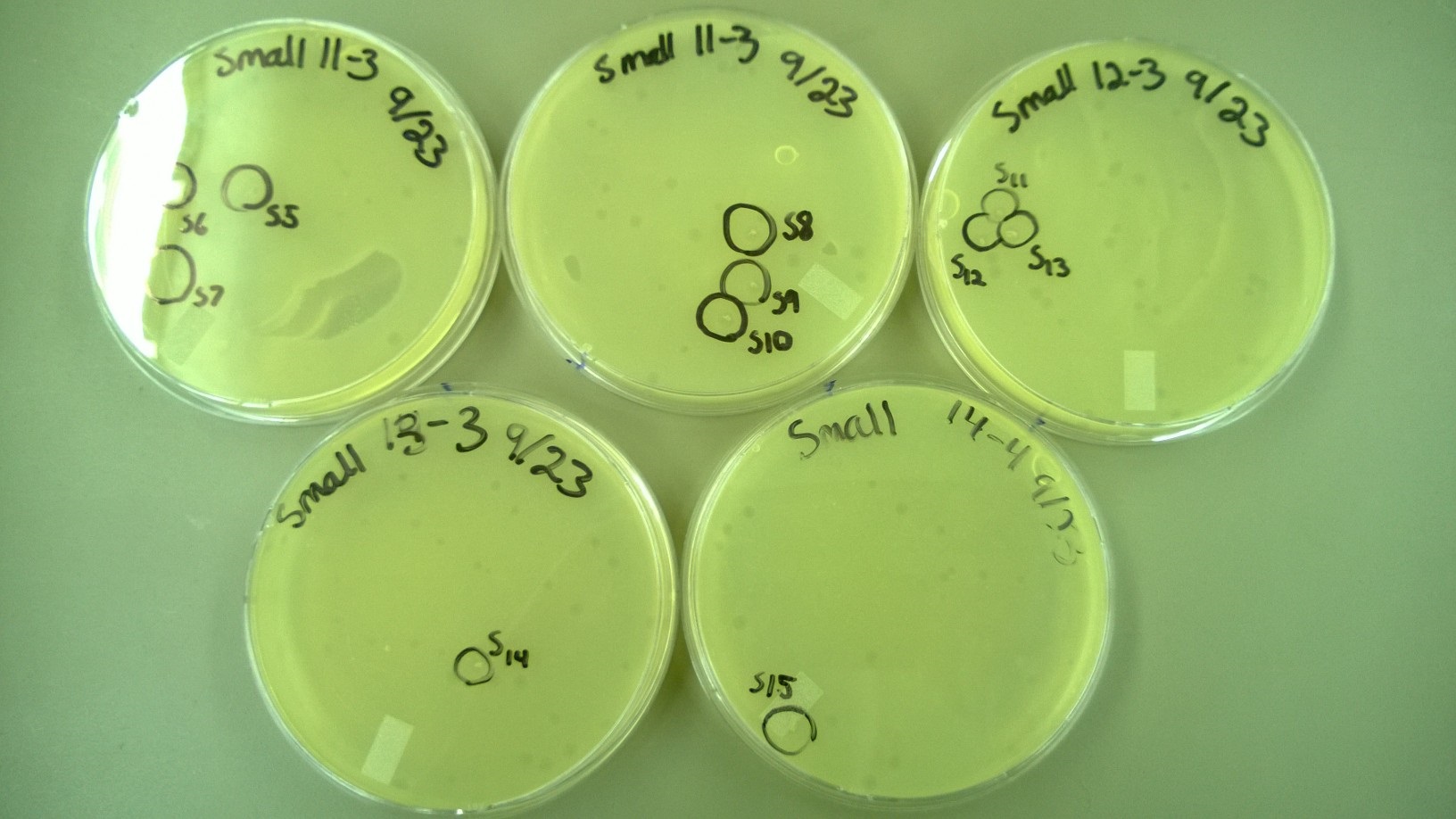
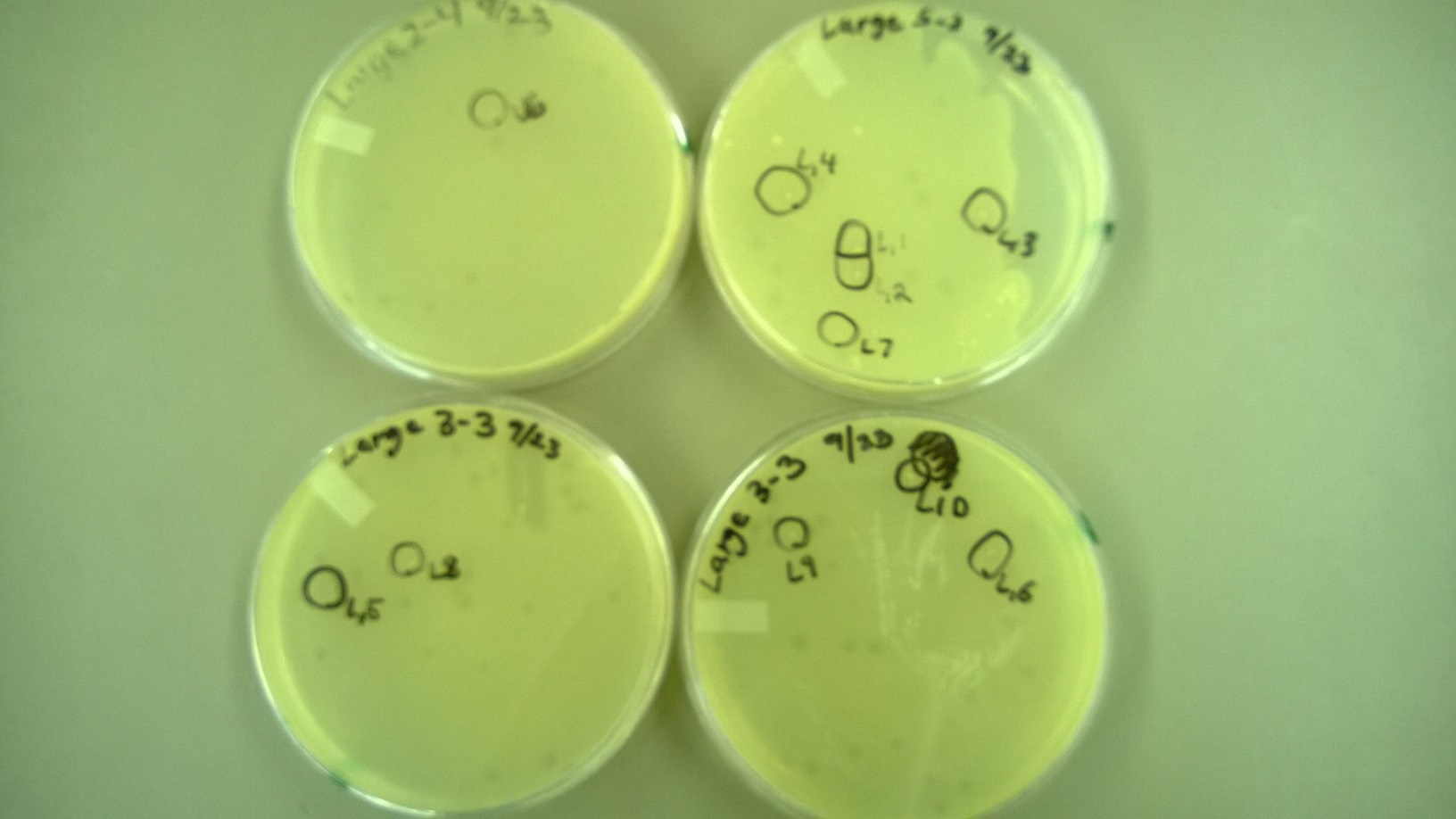
| + | 6) Plating to Check for Plaque Size 2 | ||
| + | |||
| + | * These are the plates that we selected plaques from: | ||
| + | |||
| + | [[File:6Small.JPG|300px|center]] | ||
| + | |||
| + | |||
| + | |||
| + | [[File:6Large.JPG|300px|center]] | ||
| + | |||
| + | 7) Checking for Plaque Viability from Step 5 | ||
| + | * We couldn't tell if plaque size was held constant from the plaques we picked because we had used different types of plates and different concentrations of top agar. | ||
| + | |||
| + | 8) Checking for Plaque Viability 2 | ||
| + | |||
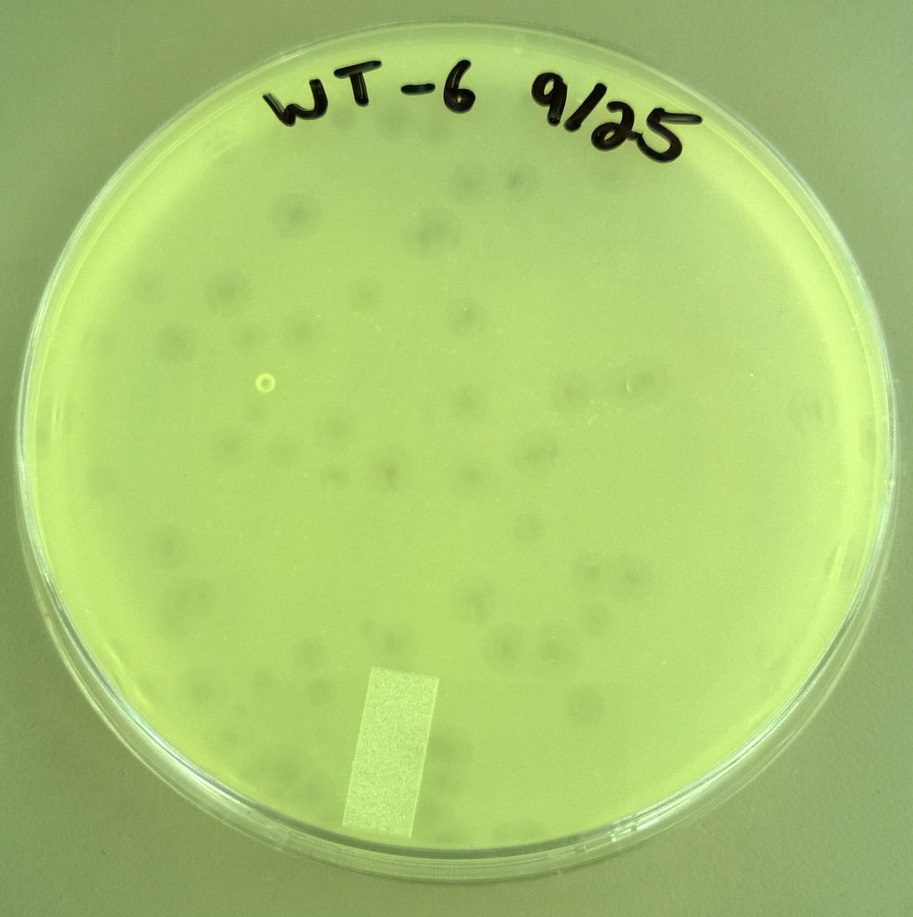
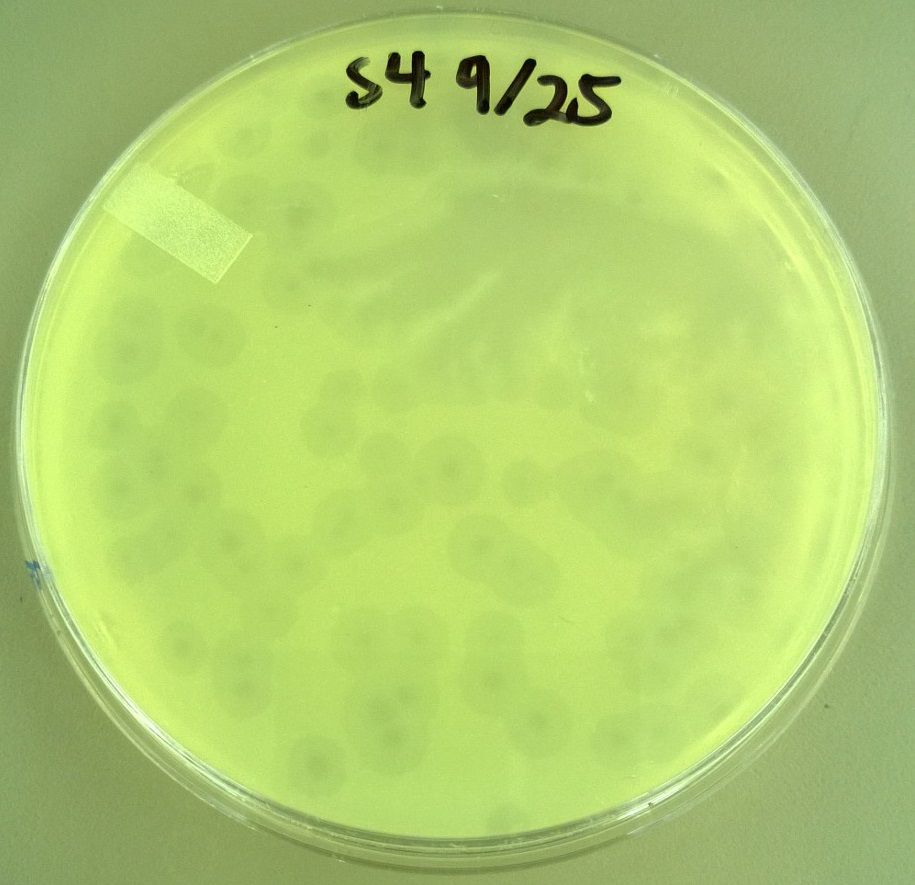
| + | * One small phage plate had considerably larger plaques than wild type phage. Another large phage plate had much smaller plaques than the wild type phage. The plaques were also about the same size as the plaques the phages were picked from, showing that the phage maintained their phenotype of forming large plaques. | ||
| + | |||
| + | * T7 Wild type: Average plaque diameter of 0.388cm +/- 0.0997cm | ||
| + | |||
| + | [[File:T7WT927.JPG|300px|center]] | ||
| + | |||
| + | * T7 Small phage: Average plaque diameter of 0.592cm +/- 0.0735cm | ||
| + | |||
| + | [[File:S4.JPG|300px|center]] | ||
| + | |||
| + | * T7 Large phage: Average plaque diameter of 0.264cm +/- 0.0867cm. | ||
| + | |||
| + | [[File:L8.JPG|300px|center]] | ||
| + | |||
| + | * There were also many plates that seemed like they could have mutant phage, but we have to run further experiments to confirm them. | ||
'''VI) Conclusion''' | '''VI) Conclusion''' | ||
| + | |||
| + | From the average plaque diameters in step 8, we conclude that we have found a smaller phage and a larger phage! The small phage made larger plaques, while the large phage made smaller plaques. The standard deviation is relatively high, but that is most likely due to the already high variability of plaque sizes. Our next steps will be to take a picture of the small and large T7 phage using an electron microscope and sequence their genomes. | ||
|} | |} | ||
Latest revision as of 00:34, 14 October 2013
| ||
|
|
9.13 Mutagen Concentration Test - Ninth Protocol
I) Purpose
II) Expected Outcome
III) Reagents Used
IV) Procedure 1) Overnight (The day before) (9.13)
2) Applying the mutagen (9.14)
3) Cesium Chloride Gradients (9.16)
4) Spot Test of Gradient Samples (9.18)
5) Plating to Check for Plaque Size (9.20)
6) Plating to Check for Plaque Size 2 (9.23)
7) Checking for Plaque Viability from Step 5 (9.23)
8) Checking for Plaque Viability 2 (9.25)
2) Applying the mutagen
3) Cesium Chloride Gradients
4) Spot Test of Gradient Samples
5) Plating to Check for Plaque Size
6) Plating to Check for Plaque Size 2
7) Checking for Plaque Viability from Step 5
8) Checking for Plaque Viability 2
VI) Conclusion From the average plaque diameters in step 8, we conclude that we have found a smaller phage and a larger phage! The small phage made larger plaques, while the large phage made smaller plaques. The standard deviation is relatively high, but that is most likely due to the already high variability of plaque sizes. Our next steps will be to take a picture of the small and large T7 phage using an electron microscope and sequence their genomes. | |
 "
"