Team:HokkaidoU Japan/Shuffling Kit/Examples
From 2013.igem.org
| (30 intermediate revisions not shown) | |||
| Line 3: | Line 3: | ||
<div id="common-header-bottom-background"> | <div id="common-header-bottom-background"> | ||
<div class="wrapper"> | <div class="wrapper"> | ||
| - | <h1 id="common-header-title">Maestro E.coli</h1> | + | <h1 id="common-header-title">Maestro <span class="italic">E. coli</span></h1> |
| - | <h2 id="common-header-subtitle"> | + | <h2 id="common-header-subtitle">Shuffling Kit</h2> |
<img id="common-header-img" src="https://static.igem.org/mediawiki/2013/e/ea/HokkaidoU2013_Maestro_Header.png"> | <img id="common-header-img" src="https://static.igem.org/mediawiki/2013/e/ea/HokkaidoU2013_Maestro_Header.png"> | ||
</div> | </div> | ||
| Line 18: | Line 18: | ||
<h2>Promoter Selector</h2> | <h2>Promoter Selector</h2> | ||
<p>Let's select the best promoter for Kanamycin resistance by Promoter Selector.</p> | <p>Let's select the best promoter for Kanamycin resistance by Promoter Selector.</p> | ||
| - | <p>For | + | <p>For this demonstration we decided to use the expression of Kanamycin resistance. Changing the concentration of Kanamycin in agar plate, it is estimated that different promoter will be chosen by our Promoter Selector (fig.1).</p> |
| - | <p>If the concentration of Kanamycin was high, the colony with strong promoter | + | <p>If the concentration of Kanamycin was high, the colony with strong promoter would survive. Therefore, only one or two colors would appear and indicate the first and second biggest occupancy rate on the plate. |
| - | If the concentration of Kanamycin was low, colonies with weak promoters | + | If the concentration of Kanamycin was low, colonies with even weak promoters could be able to survive. So in this way many colors of colonies would appear (fig.2). |
</p> | </p> | ||
<div class="fig fig800"> | <div class="fig fig800"> | ||
| Line 33: | Line 33: | ||
</div> | </div> | ||
| - | < | + | <h3>Method</h3> |
| - | <p>Optimum concentration of Kanamycin: in LB is 50 mg/ml | + | <p>Optimum concentration of Kanamycin: in LB is 50 mg/ml <br> |
| - | We prepared 3 | + | We prepared optimum and other 3 concentration plates. |
</p> | </p> | ||
| Line 49: | Line 49: | ||
Vector: pSB1C3 | Vector: pSB1C3 | ||
</p> | </p> | ||
| - | <p>We cloned Kanamycin | + | <p>We cloned Kanamycin resistance gene from pSB3K3, by using BsaI adding primer. Used the Promoter Selector (K1084501, K1084502, K1084503, K1084504, K1084505 ).</p> |
<p> | <p> | ||
| - | Culture: 37 °C, for 48h | + | Culture: 37 °C, for 48h<br> |
</p> | </p> | ||
| - | < | + | <h3>Results</h3> |
<div class="fig fig400"> | <div class="fig fig400"> | ||
| Line 67: | Line 67: | ||
</div> | </div> | ||
| - | <p>After 48h cultivation, around 300 colonies had appeared on each LBKC (Kanamycin and Chloramphenicol) plates. We prepared | + | <p>After 48h cultivation, around 300 colonies had appeared on each LBKC (Kanamycin and Chloramphenicol) plates. We prepared LacZ&alpha expression system in Promoter Selector as negative control to estimate the success of BsaI digestion. We got 0-7 blue colonies which was expressing LacZ&alpha. |
</p> | </p> | ||
| + | |||
| + | <p>We observed mixed colored colonies. They may have been transformed by two or more Promoter Selector. The number and rate of colonies per each plate were graphed (fig.4), with rejecting these undesirable colonies. | ||
| + | </p> | ||
| + | |||
<p> | <p> | ||
| - | In | + | In fig.4, legend color corresponds to Promoter Selector’s part number. The sum of colony numbers is displayed above each bar, and rate is in these sections. Number in the table referrers the number of each Promoter Selector’s colony. These data are collected by n=1. |
</p> | </p> | ||
<div class="clearfix"></div> | <div class="clearfix"></div> | ||
| - | < | + | <h3>Conclusion</h3> |
<p> | <p> | ||
| - | + | Big difference did not appear among each Kanamaicin concentration. | |
| + | |||
| + | In this experiment, number of colonies derived from K1084405 (containing K1084010 promoter) had the largest rate on each plate. This result suggests that the colonies which had K1084505 could cost little resource of transcription and translation for expression of Kanamycin resistance gene, and the rest of resource had been spared to cell growth. Thus, the number of colonies might be the largest. | ||
</p> | </p> | ||
<p> | <p> | ||
| - | From these result and the experimental fact, the existence of Km resistance gene in Promoter Selector’s BsaI cloning section is partially confirmed. Our Promoter Selector was successfully assembled | + | The other reason of why , colonies derived from K1084405 had the largest number may be, the DNA solution of K1084505 Promoter Selector used at ligation was simply larger than other DNA solution. Although the result is collected from assay of only once , higher concentration of Kanamycin and much number of trials than this time will be needed. |
| + | </p> | ||
| + | <p> | ||
| + | From these result and the experimental fact, the existence of Km resistance gene in Promoter Selector’s BsaI cloning section is partially confirmed. Our Promoter Selector was successfully assembled. However it was not adopted to all colonies. As a result of assembling, we succeeded in making colorful colonies appear on one plate. | ||
</p> | </p> | ||
<h2>RBS Selector</h2> | <h2>RBS Selector</h2> | ||
<h3>4 colors</h3> | <h3>4 colors</h3> | ||
<p>Let’s create all combinations by two reporter genes and make various colors on one plate!</p> | <p>Let’s create all combinations by two reporter genes and make various colors on one plate!</p> | ||
| - | |||
<p> | <p> | ||
| Line 99: | Line 107: | ||
</div> | </div> | ||
| - | <p>When the RBS | + | <p>When the RBS of mRFP1 was stronger than that of LacZα, the colony would be red. When in the opposite case, the colony would be blue. And when the strengths of both RBSs were same, colony would be white or purple (fig.6).</p> |
| Line 109: | Line 117: | ||
<h4>Method</h4> | <h4>Method</h4> | ||
<ul> | <ul> | ||
| - | <li> | + | <li>Assembled promoter1 (BBa_K1084001), SD2 (BBa_K1084101), SD4 (BBa_K1084102).</li> |
<li>Spread X-GAL(250 mg)on LBC plate.</li> | <li>Spread X-GAL(250 mg)on LBC plate.</li> | ||
| - | <li>Cultured | + | <li>Cultured at 37 °C for 26h.</li> |
</ul> | </ul> | ||
| Line 120: | Line 128: | ||
<h4>Results</h4> | <h4>Results</h4> | ||
<p> | <p> | ||
| - | We got | + | We got colorful colonies; red, blue, white, and purple. |
| Line 131: | Line 139: | ||
<p> | <p> | ||
| - | We can say that our RBS Selector worked!! | + | We can say that our RBS Selector worked exactly!! |
| - | The RBSs | + | The RBSs upstream of 2 genes were randomly assembled and they had different expression level. |
</p> | </p> | ||
| + | |||
<h3>64 colors</h3> | <h3>64 colors</h3> | ||
| + | |||
| + | <p> | ||
| + | Let’s create all combinations by three reporter gene and using tandem RBS, make various colors on one plate! | ||
| + | </p> | ||
| + | |||
| + | <p> | ||
| + | As the tandem RBS and the RBS selector demonstration, we randomized 64 patterns of RBS-CDS combination. By adding tandem RBS, three kinds of CDSs and GGA VACTOR, One-pot Golden Gate Assembly had done for making 64 kinds of different constructs. | ||
| + | </p> | ||
<div class="fig fig800"> | <div class="fig fig800"> | ||
<img src="https://static.igem.org/mediawiki/2013/1/1d/64demo2_HokkaidoU_2013.png"> | <img src="https://static.igem.org/mediawiki/2013/1/1d/64demo2_HokkaidoU_2013.png"> | ||
| - | <div><span class="bold">fig.8.</span></div> | + | <div><span class="bold">fig.8 The 64 patterns of RBS-CDS combination constructed by RBS Selector.</span></div> |
</div> | </div> | ||
| + | |||
| + | <p> | ||
| + | <h4>Method</h4> | ||
| + | <ul> | ||
| + | <li>Used tandem RBS (BBa_K1084302), GGA VECTOR that BsaI and overhang was added by PCR from K1084401, eforRed (BBa_K592012), aeBlue (BBa_K864401) and amilGFP (BBa_K592010) are assembled by Golden Gate Assembly.</li> | ||
| + | <li>Spread on LBC plate.</li> | ||
| + | <li>Cultured at 37 °C, 48h.</li> | ||
| + | </ul> | ||
| + | </p> | ||
| + | |||
| + | <div class="fig fig400"> | ||
| + | <img src="https://static.igem.org/mediawiki/2013/5/55/HokkaidoU_2013_64ROK_result_1.JPG"> | ||
| + | <div><span class="bold">fig.9 The result plate of 64 pattern randomizing.</span></div> | ||
| + | </div> | ||
| + | |||
| + | <h4>Results</h4> | ||
| + | |||
| + | <p> | ||
| + | |||
| + | |||
| + | </p> | ||
| + | |||
| + | <p> | ||
| + | |||
| + | Many yellowish green (amilGFP) and red color (mRFP1) colonies appeared, but these colonies are undesirable colonies.They did not contain inserts of all three color proteins. As a Golden Gate assembly result, green color (strong aeBlue and amilGFP expression) appeared, and the insert DNA length was confirmed that the insert DNA has same length of Golden Gate Assembly. | ||
| + | Other color colony, white, weak green, weak amilGFP expression colonies were observed. We did not have time to confirm all inserts were ligated. | ||
| + | </p> | ||
| + | <p></p> | ||
| + | <h4>Conclusion</h4> | ||
| + | |||
| + | |||
| + | <p> | ||
| + | mRFP1 expression might be caused by the remaining of mRFP1 expression DNA used as template for GGA VECTOR producing by PCR (K1084401). | ||
| + | </p> | ||
| + | |||
| + | <p> | ||
| + | However, the blue and green colonies appeared on the plate had the insert DNA length same with ideal Golden Gate Assembling product. Our Shuffling kit have partially worked! | ||
| + | </p> | ||
Latest revision as of 03:59, 29 October 2013
Maestro E. coli
Shuffling Kit

Demonstrations for Usecase Example
We will show some interesting demonstrations of our kits, Promoter Selector and RBS Selector!
Promoter Selector
Let's select the best promoter for Kanamycin resistance by Promoter Selector.
For this demonstration we decided to use the expression of Kanamycin resistance. Changing the concentration of Kanamycin in agar plate, it is estimated that different promoter will be chosen by our Promoter Selector (fig.1).
If the concentration of Kanamycin was high, the colony with strong promoter would survive. Therefore, only one or two colors would appear and indicate the first and second biggest occupancy rate on the plate. If the concentration of Kanamycin was low, colonies with even weak promoters could be able to survive. So in this way many colors of colonies would appear (fig.2).


Method
Optimum concentration of Kanamycin: in LB is 50 mg/ml
We prepared optimum and other 3 concentration plates.
- Plate A: Kanamycin 125 mg per plate
- Plate B: Kanamycin 250 mg per plate
- Plate C: Kanamycin 500 mg per plate (optimum concentration)
- Plate D: Kanamycin 1000 mg per plate
Gene Vector: pSB1C3
We cloned Kanamycin resistance gene from pSB3K3, by using BsaI adding primer. Used the Promoter Selector (K1084501, K1084502, K1084503, K1084504, K1084505 ).
Culture: 37 °C, for 48h
Results


After 48h cultivation, around 300 colonies had appeared on each LBKC (Kanamycin and Chloramphenicol) plates. We prepared LacZ&alpha expression system in Promoter Selector as negative control to estimate the success of BsaI digestion. We got 0-7 blue colonies which was expressing LacZ&alpha.
We observed mixed colored colonies. They may have been transformed by two or more Promoter Selector. The number and rate of colonies per each plate were graphed (fig.4), with rejecting these undesirable colonies.
In fig.4, legend color corresponds to Promoter Selector’s part number. The sum of colony numbers is displayed above each bar, and rate is in these sections. Number in the table referrers the number of each Promoter Selector’s colony. These data are collected by n=1.
Conclusion
Big difference did not appear among each Kanamaicin concentration. In this experiment, number of colonies derived from K1084405 (containing K1084010 promoter) had the largest rate on each plate. This result suggests that the colonies which had K1084505 could cost little resource of transcription and translation for expression of Kanamycin resistance gene, and the rest of resource had been spared to cell growth. Thus, the number of colonies might be the largest.
The other reason of why , colonies derived from K1084405 had the largest number may be, the DNA solution of K1084505 Promoter Selector used at ligation was simply larger than other DNA solution. Although the result is collected from assay of only once , higher concentration of Kanamycin and much number of trials than this time will be needed.
From these result and the experimental fact, the existence of Km resistance gene in Promoter Selector’s BsaI cloning section is partially confirmed. Our Promoter Selector was successfully assembled. However it was not adopted to all colonies. As a result of assembling, we succeeded in making colorful colonies appear on one plate.
RBS Selector
4 colors
Let’s create all combinations by two reporter genes and make various colors on one plate!
The RBS Selector we made, can randomize the strength of RBSs in the operon. For a demonstration, we decided to create all combinations by two genes; mRFP1 (BBa_E1010) and LacZα (BBa_I732006) (fig.5). LacZα makes the colony blue. mRFP1 makes the colony red.

When the RBS of mRFP1 was stronger than that of LacZα, the colony would be red. When in the opposite case, the colony would be blue. And when the strengths of both RBSs were same, colony would be white or purple (fig.6).

Method
- Assembled promoter1 (BBa_K1084001), SD2 (BBa_K1084101), SD4 (BBa_K1084102).
- Spread X-GAL(250 mg)on LBC plate.
- Cultured at 37 °C for 26h.

Results
We got colorful colonies; red, blue, white, and purple.
Conclusion
We can say that our RBS Selector worked exactly!! The RBSs upstream of 2 genes were randomly assembled and they had different expression level.
64 colors
Let’s create all combinations by three reporter gene and using tandem RBS, make various colors on one plate!
As the tandem RBS and the RBS selector demonstration, we randomized 64 patterns of RBS-CDS combination. By adding tandem RBS, three kinds of CDSs and GGA VACTOR, One-pot Golden Gate Assembly had done for making 64 kinds of different constructs.
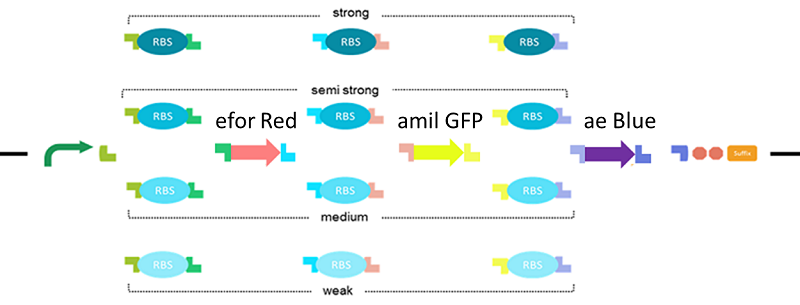
Method
- Used tandem RBS (BBa_K1084302), GGA VECTOR that BsaI and overhang was added by PCR from K1084401, eforRed (BBa_K592012), aeBlue (BBa_K864401) and amilGFP (BBa_K592010) are assembled by Golden Gate Assembly.
- Spread on LBC plate.
- Cultured at 37 °C, 48h.

Results
Many yellowish green (amilGFP) and red color (mRFP1) colonies appeared, but these colonies are undesirable colonies.They did not contain inserts of all three color proteins. As a Golden Gate assembly result, green color (strong aeBlue and amilGFP expression) appeared, and the insert DNA length was confirmed that the insert DNA has same length of Golden Gate Assembly. Other color colony, white, weak green, weak amilGFP expression colonies were observed. We did not have time to confirm all inserts were ligated.
Conclusion
mRFP1 expression might be caused by the remaining of mRFP1 expression DNA used as template for GGA VECTOR producing by PCR (K1084401).
However, the blue and green colonies appeared on the plate had the insert DNA length same with ideal Golden Gate Assembling product. Our Shuffling kit have partially worked!
 "
"


