Header
Smart drug delivery systems should administer drugs at the right place, at the right time and the right concentration. Thus diminish the side effects and the amount of drugs needed. It could eventually be used to treat a wide range of diseases, but especially tumors. The idea to target the drug delivery is to increase the concentraton of the drug at the targetsite while keeping it to a minimum in the rest of the organism. One big drawback of smart drug delivery is that it doesn't exist for many drugs, it needs to be developed for each drug separately and that is quite expensive.
Introduction
Our team aims to create a highly adaptable smart drug delivery system, which could be used for several applications and alternatives in disease treatment. The design makes it so flexible that it could even be possible to adapt it to the patient’s individual needs. By being highly adaptive, the system we propose would also reduce the cost of targeted drug delivery because, instead of creating a new system for each drug and each patient, one could simply use the basic part that we made, modify them slightly and assemble them.
A Proof of Principle
Our effort is limited to a proof of principle. We used E.coli as model organism. Using the principles of synthetic biology we aimed to engineer an E.coli Bacterium that expresses streptavidin on its surface and contains a pH sensing promoter that controls the expression of gelatinases. Those bacteria would be able to bind biotinylated nanoparticles.
The Nanoparticles
The nanoparticles are made out of gelatin. This kind of nanoparticles has been shown to be able to transport proteins, organic molecules or DNA. They were biotinylated using activated biotin. We invite you to read more about Nanoparticles. To assemble the nanoparticles with the bacteria, they only need to be in the same tube, because the interaction between biotin and streptavidin is very strong and spontaneous.
Cell surface display
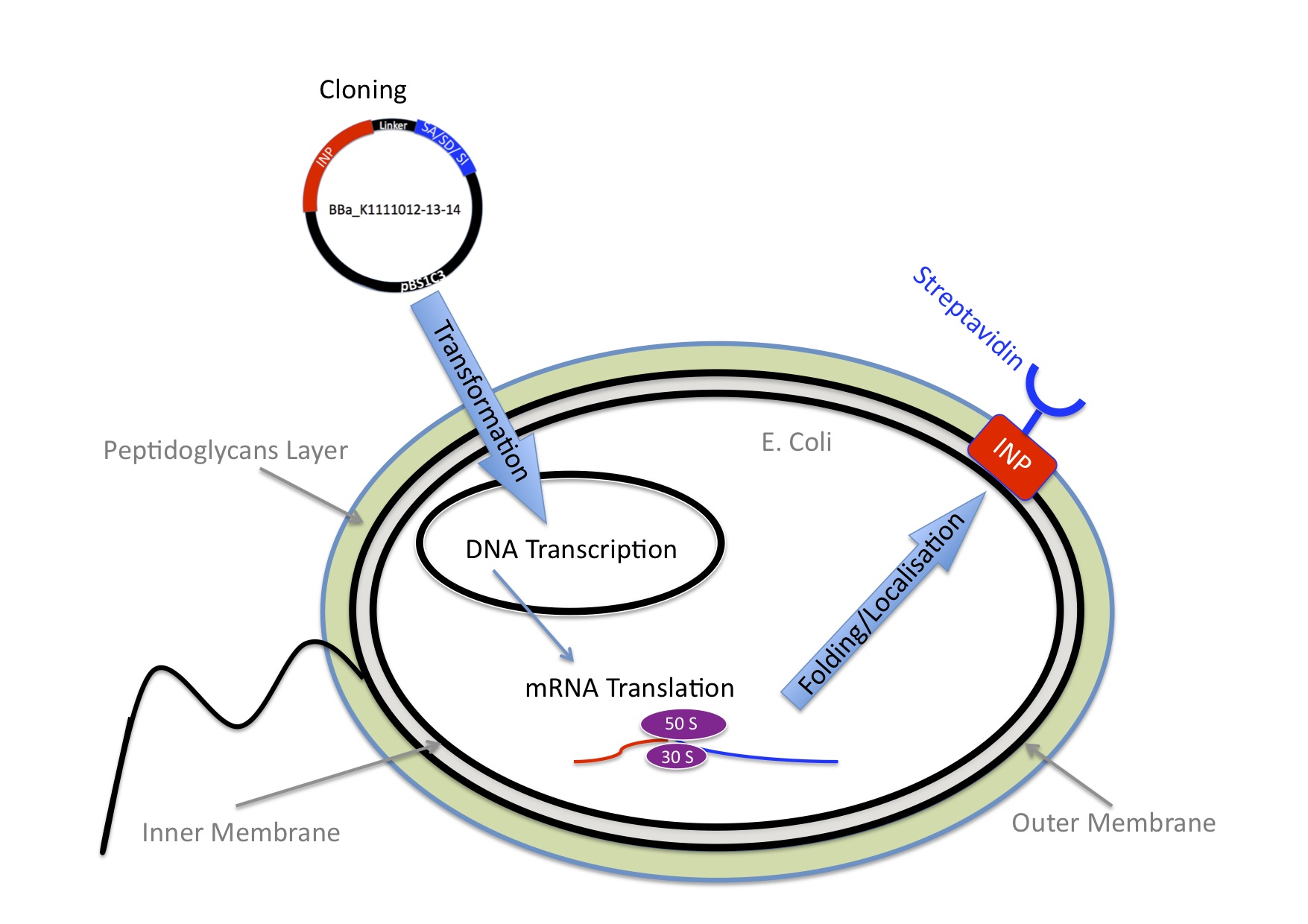
To make the bacteria express streptavidin on their surface, we engineered a plasmid that encodes a fusion protein between streptavidin and ice nucleation protein from pseudomonas syringae, which serves as an anchor for the streptavidin to the outer membrane. Read more about Cell Surface Display . At this point the bacteria would be able to transport nanoparticles, but on its own this wouldn’t make any sense.
Sensing
The crucial point of our project is that E.coli was genetically engineered to sense a specific trigger in the environment. Read more about Sensing. Sensing such a trigger induces the production of enzymes that degrade the nanoparticles, GelE gelatinase from Enterococcus Fecalis and Matrix Metalloprotease MMP2 from H. Sapiens. So the drug will be released exactly where it is needed to treat the patient.
As mentioned before, we only aimed to make a proof of principle. Eventually, it would be necessary to develop the system in another organism, to add safety mechanisms and develop different sensing plasmids. Read more about the future applications we imagined.
 "
"