Team:TU Darmstadt/result/molecular engineering
From 2013.igem.org
(Created page with "<html> <style type="text/css"> body { margin:0; padding:15px 0 0; background-color: #ececec; background-image:url("https://static.igem.org/mediawiki/2013/1/14/Background_(3000x180_P...")
Newer edit →
Revision as of 00:01, 5 October 2013
Our results
LSSmOrange:
LSSmOrange is an orange fluorescent protein that we want to use for FRET, we us it as a donor. In a recent paper Daria M. Shcherbakova [1] showed that LSSmOrange has an excitation maximum by 437 nm and an emission maximum by 572 nm. In the following diagram (graph 1) one can see our results with our own expressed LSSmOrange. The colored dots show the different maximums. The excitation maximum is at 415 nm, another at 453 nm and our measured emission maximum is at 564 nm. This aberration changes nothing on the FRET system. The excitation maximum is big enough to stimulate mKate, the other fluorescent protein. Of course, we also checked the emission and excitation of our BL21(DE3) cells (data not shown) and we can eliminate the theories that these cells disturb this florescence measurement.
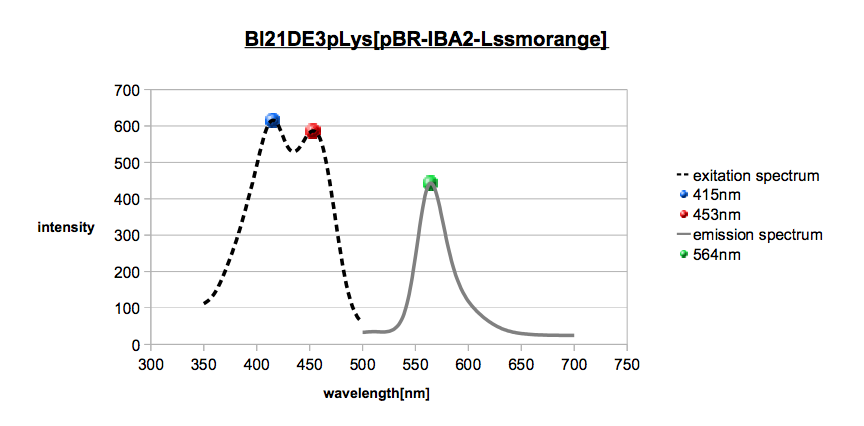
Excitation Spectrum (dashed line) and emission spectrum (solid line) from LSSmOrange with marked maximums.

Pellet of E.coli Bl21(DE3) cells with expressed LSSmOrange. Left side: without UV radiation, right side: with UV radiation.
mKate:
mKate2 is a red fluorescent protein that we want to use as a acceptor for FRET. According to Evrogen [2]
mKate2 has an excitation maximum at 588 nm and an emission maximum at 633 nm. The
diagram below (graph 2, colored dots) shows that the excitation maximum is at 579 nm and the
emission maximum is 611 nm. The first peak does not look exactly like a single peak, but we can see a
clear maximum. The problem in our FRET system is the very low intensity which results
in the next diagram (graph 3) in a huge difference of intensity of the two fluorescence
proteins.
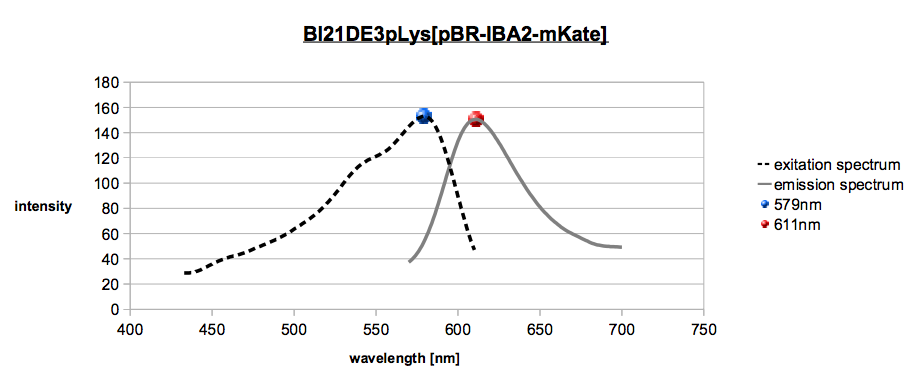
Excitation Spectrum (dashed line) and emission spectrum (solid line) of mKate with marked maximums

Pellet of E.coli Bl21(DE3) cells with expressed mKate. Left side: without UV radiation, right side: with UV radiation.
FRET:
Both diagrams in an overlay shows us the FRET system. LSSmOrange is the donor and mKate is the
acceptor. For an optimum in FRET, it should display a stronger overlap between the emission maximum of LSSmOrange and the excitation maximum of mKate.
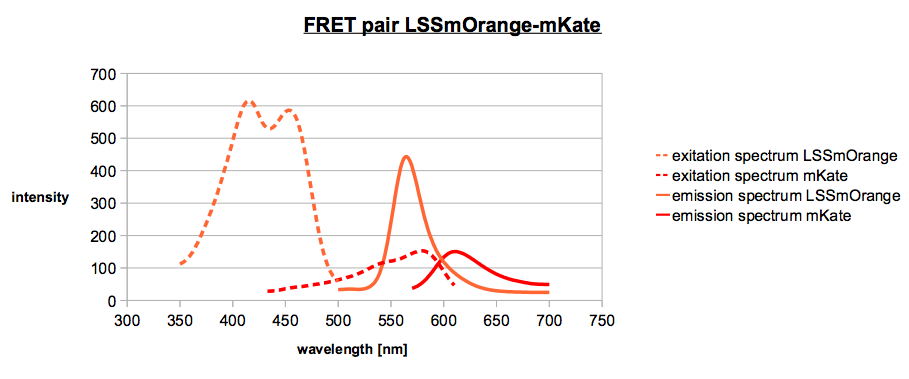
Overlay of figure 1 and figure 2.
The next figure is from the paper of M. Shcherbakova. In this figure one can
clearly see the overlap between donor and acceptor in an optimal FRET system.
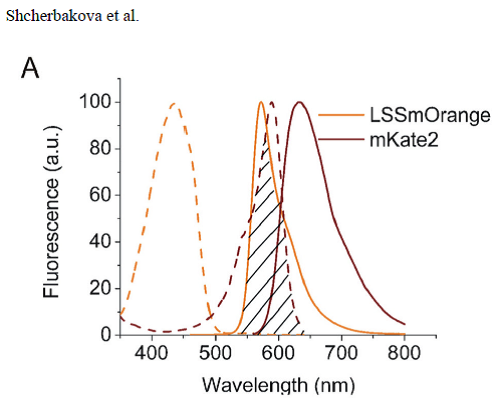
(dashed line = excitation spectrum, solid line = emission spectrum)
To determine the influence of the induced constructs mKate and LSSmOrange on the BL21DE3, growth curves were used. During a time period of 8 h the OD of the samples was measured at a wavelength of 600 nm in time steps of 15 min. The measurement was started at an OD600 nm value of 0.3. With the help of the following formulas the growth rate k and the doubling time td were calculated:
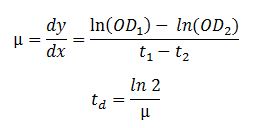
The calculated values of k and td of the competent cells BL21DE3 containing the mKate or LSSmOrange construct were compared to the level of the wild type strain, to reveal possible toxicity of the constructs. In the following diagrams the OD600, respectively the ln OD600, values are plotted against the time and in the table below the resulting values of the growth rates k and the doubling times td are listed.
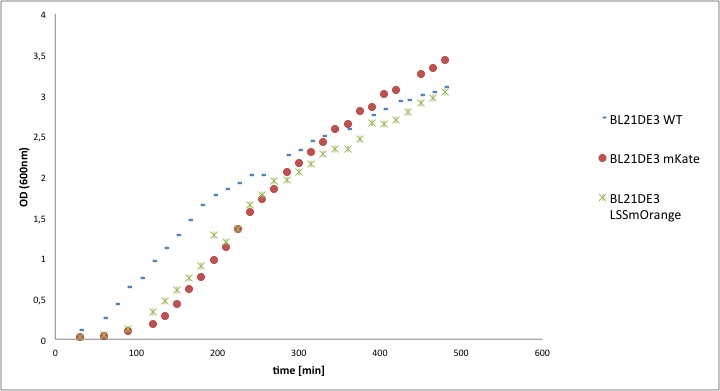
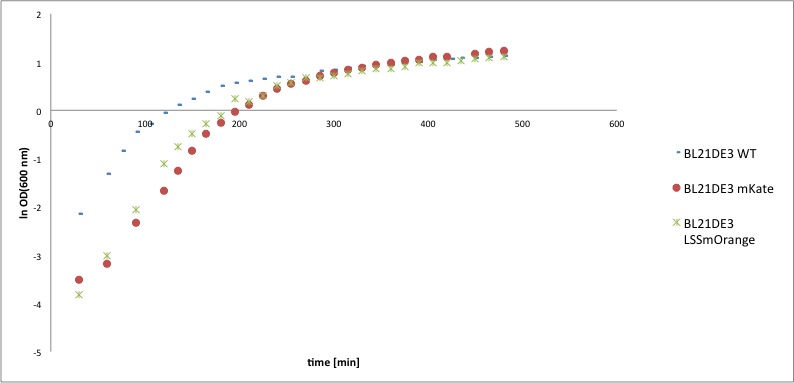
Table1: The growth rate k and the doubling time td of BL21DE3 WT, BL21DE3 mKate and BL21DE3LSSmOrange.
| BL21DE3(pPR-IBA2) | BL21DE3[pPR-IBA2-LSSmOrange] | BL21DE3[pPR-IBA2-mKate] | |
| growth rate µ [min-1] | 0,0286 | 0,0287 | 0,0232 |
| doubling time td [min] | 24,2459 | 24,1514 | 29,877 |
As can be seen in the two diagrams the course of the growth curves are rather similar. The growth rate as well as the doubling time of the wild type BL21DE3 and the one including the LSSmOrange construct are almost identical. The growth rate of the strain including the mKate construct is slightly lower and therefore the doubling time is around 5 min higher. All in all, the differences of the samples including the constructs compared to the wild type BL21DE2 are negligibly low. Therefore it can be assumed that the construct mKate as well as LSSmOrange are not toxic for the BL21DE3 cells and have no or a negligibly low influence on the growth of the cells.
References
- Daria M. Shcherbakova et al. (2012) An Orange Fluorescent Protein with a Large Stokes Shift for Single-Excitation Multicolor FCCS and FRET Imaging. J. Am. Chem. Soc. 134 (18), 7913–7923
- http://www.evrogen.com/products/basicFPs.shtml
 "
"