Team:Tianjin/Project/Characterization
After finishing our final construction of our Alk-Sensor, we wanted to know its more property and funtions. So we did several test around it. The Figure below showed the test we did, click the picture for more details for the test.
Contents |
Alkane Detection For Alkane Producer
Construction:
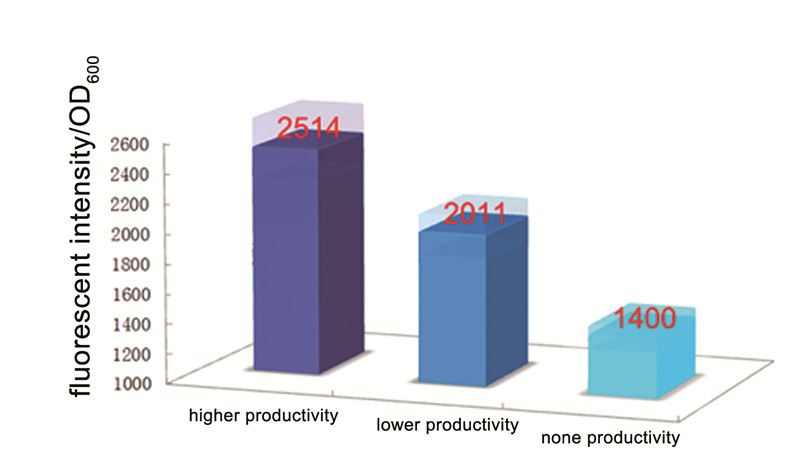
In this test, we try to use our Alk-Sensor to “mark” the alkane molecular which is produced by E. coli with different alkane productivity. In our project, we choose NPDC and AAR gene to construct the alkane producing modular. We choose a strong promoter (BBa_J23100) and a weaker promoter (BBa_J23114) to control the NPDC-AAR transcription unit, to obtain alkane modular with different alkane productivity. At the same time, we build a control plasmid which didn’t contain any part as the modular with no alkane productivity. Then we cotransform the alkane producing modular and our Alk-Sensor to E. coli BL21 to build the strains with different alkane productivity. (All construct process showed in Figure 1)

Test process and Result:
Then we fermented these three strain in M9 media with proper concentration of antibiotics(34ug/ml Cm and 50ug/ml Km) for about 48 hours. We can find that the strains which can produce alkane are red and more colorful than control, the strain with high alkane productivity is the most colorful one. And then we measured the fluorescent intensity of the 3 strain, the result showed in Figure 3. Although the control one also have a severely leaky of the expression of RFP, but the fluorescent per OD600 of strain with alkane producing modular is higher than the control, and the strain with higher alkane productivity got a higher fluorescent intensity per OD600.
Conclusion:
This test proved that our Alk-Sensor can be the “marker” which could transform the signal of Alkane which is small inconspicuous molecular to the signal (RFP) which can be detected by ourselves more easily. And the relationship between the input and output was positive.
Exogenous Alkane Induction
Although the Alk-Sensor can detect the alkane produce by engineered strain, but it is also difficult to measure the concentration of alkane just according to the fluorescent intensity, so the Alk-Sensor needs more characterization. So we did two test to characterize the Alk-Sensor through exogenous alkane induction, these results will help us get more information of the relationship between input (Alkane) and output (RFP).
a. Alkane specific of our Alk-Sensor
The alkane biosynthesized in E. coli is a mixture of alkane with different length, so it is important to know the specific alkane of the Alk-Sensor for building a accurate relationship between the input and output. So we test the response of Alk-Sensor to different kinds of alkanes. We choose octane, decane, dodecane, tetradecane and hexadecane as our targets and use full dose alkane to induce the expression of RFP. The result shows in Figure 4 and Figure 5. The respond to the different alkane is C8>C12>C14>C10>C16>control. Because of the low solubility of alkane especially the longer chain alkane, the response of Alk-Sensor may be influenced. So the response to the hexadecane is nearly to the control.
[[Image:#.png|thumb|600px|center|Figure 4.]]
b. Sensitive internal determination
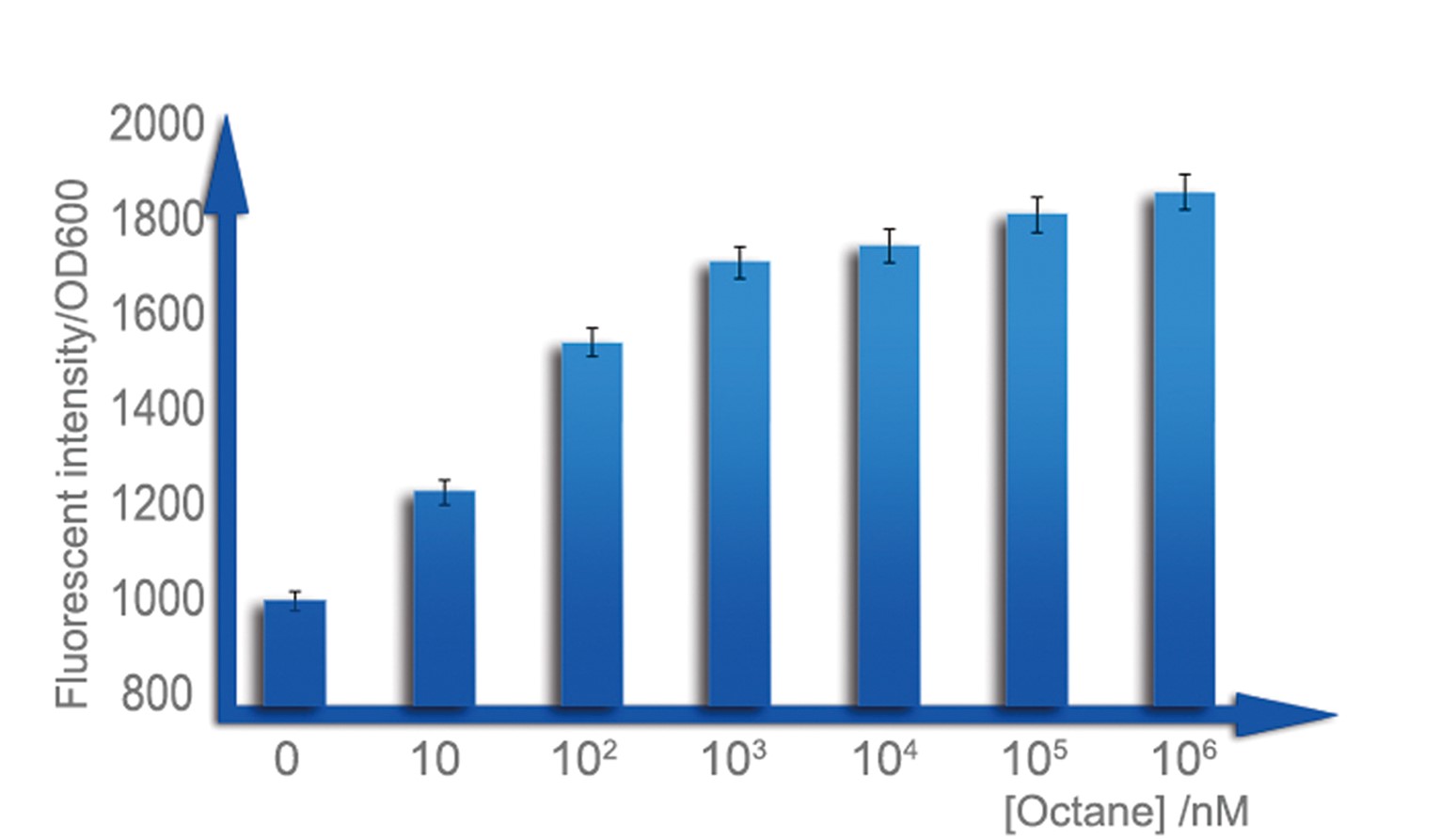
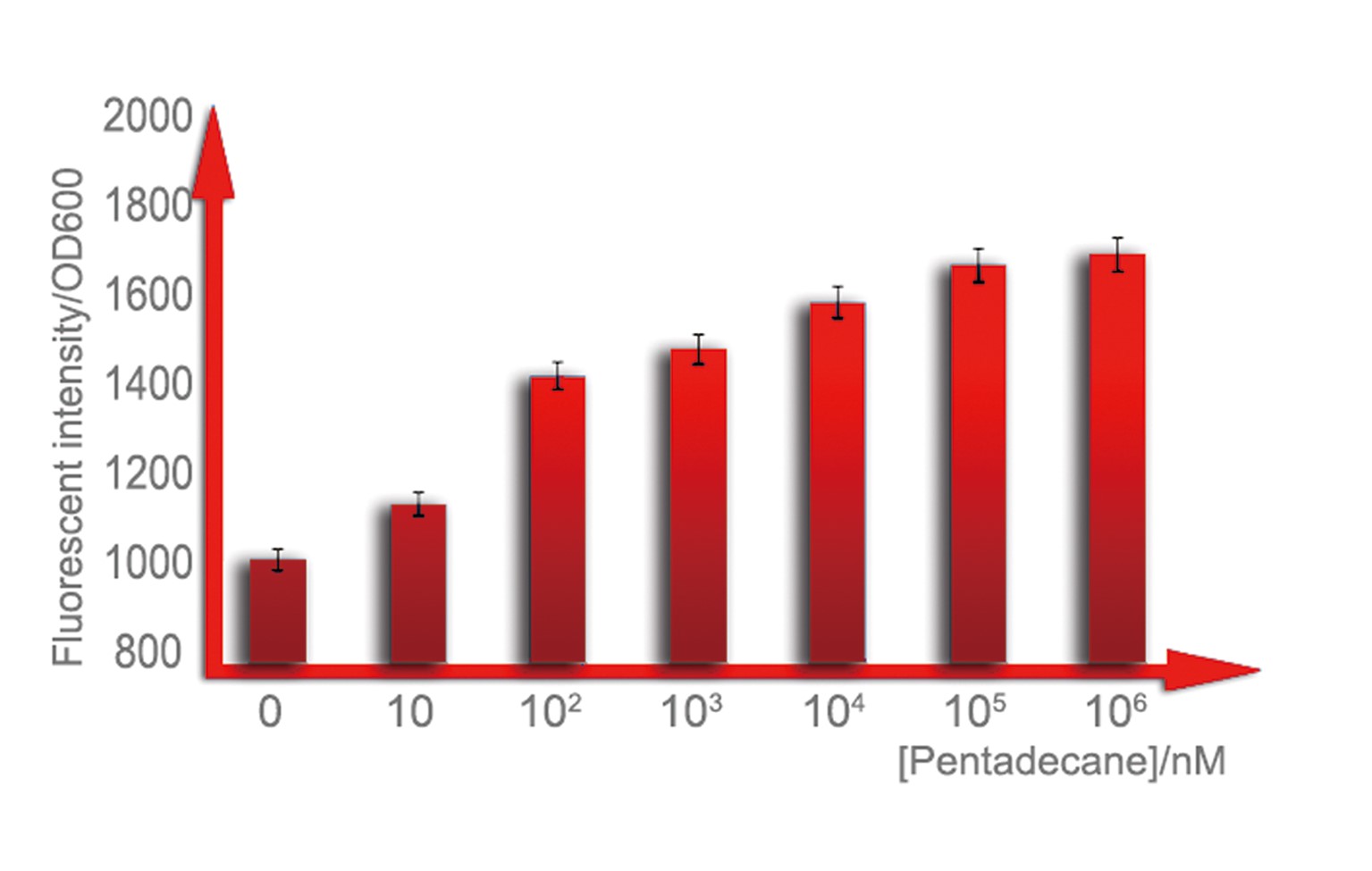
For a biosensor, it is important to know its sensitive internal. Different strength of input can be distinguished accurately only when they are in the sensitive internal. To determination the sensitive internal of Alk-Sensor, we first use different concentration of octane and pentadecane to induce the expression of RFP. In the first round of induction, the concentrations of octane we set are 10nM, 100nM, 1uM, 10uM, 100uM and 1mM. The alkane we used is dissolved in the ethanol, and to improve the absorption of the alkane into the cell, we added 1% DMSO to increase the permeability of the cell memebrane. The result shows in Figure 6 and Figure 7. According to the data, we find the Alk-sensor is more sensitive when the concentration of the alkane is below 100nM.
Then we choose 20nM, 40nM, 60nM, 80nM and 100nM alkane to induce the Alk-Sensor again, to find the working curve in the sensitive internal. The result showed in Figure 8. This data may help us calculate the concentration of alkane according to the fluorescent intensity.
[[Image:#.png|thumb|600px|center|Figure 7. We culture our strain in 3mL LB medium with 1%DMSO, which can facilitate solution of alkane in water, until OD reaches 0.6, add pentadecane of a concentration range from100nmol/L to 1mmol/L, over-night cultured. All of them are utilized to measure fluorescence intensity of cells.]]
 "
"