Team:BYU Provo/Notebook/SmallPhage/Springexp/Period3/Exp/7.8 Mutagen Concentration Test - Forth Protocol
From 2013.igem.org
(Difference between revisions)
| (7 intermediate revisions not shown) | |||
| Line 14: | Line 14: | ||
<font color="#333399" size="3" font face="Calibri"> | <font color="#333399" size="3" font face="Calibri"> | ||
| - | : | + | <font size = "4"> |
| + | |||
| + | : <u> '''Small Phage''' </u> </font> | ||
: [[Team:BYU Provo/Notebook/SmallPhage/Winterexp|March-April]] | : [[Team:BYU Provo/Notebook/SmallPhage/Winterexp|March-April]] | ||
| Line 30: | Line 32: | ||
<font face="Calibri" size="3"> | <font face="Calibri" size="3"> | ||
| - | <font size="5"> '''7.8 Mutagen Concentration Test - | + | <font size="5"> '''7.8 Mutagen Concentration Test - Fourth Protocol''' </font> |
<br> | <br> | ||
| Line 36: | Line 38: | ||
'''I) Purpose''' | '''I) Purpose''' | ||
| - | : To test the | + | : To test the forth protocol for applying 5-bromodeoxyuridine and inducing mutation. |
'''II) Expected Outcome''' | '''II) Expected Outcome''' | ||
| Line 64: | Line 66: | ||
: - 30uL of 5.3 phage stock was added to each centrifuge tube (add nothing to C). ''From 5.15 Titer Test on 5.3 T7 new Phage Stock, we know that 5.3 phage stock has 7E8 particles/20uL. Thus, to each of the test tubes, we are adding approximately 1.2E9 particles.'' | : - 30uL of 5.3 phage stock was added to each centrifuge tube (add nothing to C). ''From 5.15 Titer Test on 5.3 T7 new Phage Stock, we know that 5.3 phage stock has 7E8 particles/20uL. Thus, to each of the test tubes, we are adding approximately 1.2E9 particles.'' | ||
| - | : - Incubate phage on shaker at 37C | + | : - Incubate phage on shaker at 37C for 3 hours. |
| - | : - Liquid culture from the four test (excluding C) tubes is then centrifuged at 4000 rpm for 10 minutes to pellet cell debris. Supernatant containing phage particles is removed and | + | : - Added 1mL of chloroform to each test tube. Liquid culture from the four test (excluding C) tubes is then centrifuged at 4000 rpm for 10 minutes to pellet cell debris. Supernatant containing phage particles is removed and placed in a new tube. |
: - The stock solutions are now stored in 4 Celsius. | : - The stock solutions are now stored in 4 Celsius. | ||
| Line 78: | Line 80: | ||
: Plates were incubated upside down for 24 hours. | : Plates were incubated upside down for 24 hours. | ||
| + | 3) Spot Test to Confirm Phage Viability (7.12) | ||
| + | : - Add 0.75mL of BL21 overnight into 8 test tubes | ||
| + | |||
| + | : - Add 7mL of x6 top agar to each tube and plate it on top of LB plates | ||
| + | |||
| + | : - Divide the plates into 6 quadrants with a sharpie. 4 plates are for the new dilution (next step) and 4 plates are for the old dilution series (step 2), each labeled with their respective mutagen concentration series. | ||
| + | |||
| + | : - Using the mutagenized phage stock from step 1 above, create a 1:10 dilution series of each mutagen concentration (0ul, 100ul, 200ul, and 500ul) from 0 to -4 using 90ul LB and 10ul phage in eppendorf tubes. | ||
| + | |||
| + | : - Spot 5ul of each eppendorf tube (from both new and old dilution) onto the plates. Also spot 5ul of 5.3 phage stock on each plate as a control. | ||
'''V) Results''' | '''V) Results''' | ||
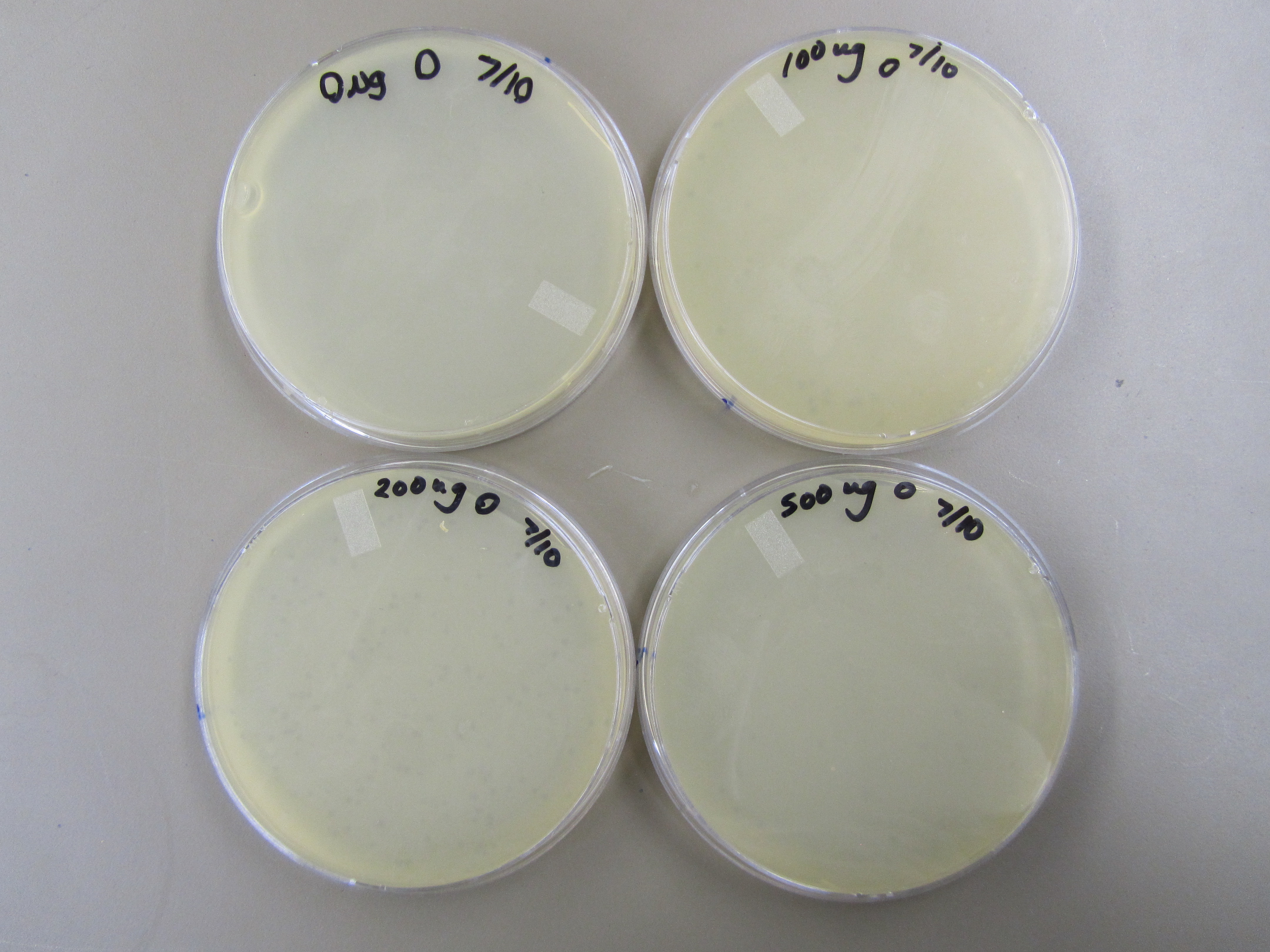
| + | 2) Titer to Determine Phage Concentration 1 | ||
| + | : - Only the 0 plates from the 100ul, 200ul, and 500ul series showed plaques. All other plates had no plaques. The 0 200ul plate had the most plaques. | ||
| - | + | [[File:BYUSuM4TiterTest.JPG|400px|center|link=]] | |
| + | 3) Spot Test to Confirm Phage Viability | ||
| - | + | : - The old dilution series plates showed plaques at 0 and 5.3. | |
| + | |||
| + | : - The new dilution series plates showed the following plaques: | ||
| + | |||
| + | :: - 0ug: 5.3, 0 | ||
| + | |||
| + | :: - 100ug: 5.3, 0, -1 | ||
| + | |||
| + | :: - 200ug: 5.3, 0, -1, -2 | ||
| + | |||
| + | :: -500 ug: 5.3, 0 | ||
| + | |||
| + | [[File:BYUSuM4SpotTest.JPG|400px|center|link=]] | ||
| + | |||
| + | '''VI) Conclusion''' | ||
| + | |||
| + | : Our results don't point to any concrete conclusion. The results try to tell us that as we add mutagen up to 200ug, phage viability increases. At 500ug, phage viability drops. We know this can't be right, especially because our control (0ug) showed an abnormal decrease in phage. This points to a possible error in our experiment. The 3 possible errors we could find were 1) 5.3 stock might be dying, 2) The overnights had their lids screwed on tight so that might have killed off some of the bacteria, and 3) It's possible that when we started overnights in different test tubes, the concentrations of bacteria could have been different. We will correct these 3 practices in our next experiments. | ||
|} | |} | ||
Latest revision as of 15:33, 9 September 2013
| Small Phage July - August Notebook: Experiments
| ||
|
|
7.8 Mutagen Concentration Test - Fourth Protocol
I) Purpose
II) Expected Outcome
III) Reagents Used
IV) Procedure 1) Applying the mutagen (7.8)
2) Titer to Determine Phage Concentration 1 (7.10)
3) Spot Test to Confirm Phage Viability (7.12)
V) Results 2) Titer to Determine Phage Concentration 1
 3) Spot Test to Confirm Phage Viability
 VI) Conclusion
| |
 "
"