Team:Tokyo Tech/Project/Farming
From 2013.igem.org
| Line 90: | Line 90: | ||
<p> | <p> | ||
<div style="margin-left:80px;"> | <div style="margin-left:80px;"> | ||
| - | iP : 6-(γ, γ-Dimethylallylamino) purine, tZ : trans-zeatin<br> | + | iP : 6-(γ, γ-Dimethylallylamino) purine, tZ : trans-zeatin (both sorts of cytokinin)<br> |
The absorbances of the supernatant were read at 663.6 and 646.6 nm. Calculation of concentrations of the chlorophyll were carried out as described by Porra et al.,1989. | The absorbances of the supernatant were read at 663.6 and 646.6 nm. Calculation of concentrations of the chlorophyll were carried out as described by Porra et al.,1989. | ||
</div></p> | </div></p> | ||
Revision as of 17:16, 28 October 2013
Farming
Contents |
1. Introduction
In our story, E. ninja retires from being an undercover warrior, and turns to farming (Fig. 2-3-1). Our improved part for phosphate sensor and a new part for cytokinin synthesis will lead us to our goal, which is to create "Farmer E. coli" that could increase plant growth by synthesizing several plant hormones depending on the soil environment. As for sensing soil nutrients, we focused on phosphate and nitrate because these are known to be especially important for plants. When nutrient levels are low, plants are in danger of withering. Therefore, we are proposing E. coli to produce the plant hormone "auxin," which promotes the growth of plants' roots. Also, when the soil is rich in nutrients, plants can grow quickly. Therefore, we want to make E. coli produce the plant hormone "cytokinin," which promotes the growth of plants' shoots (Fig. 2-3-2).
Our "Farmer E. coli" can be implemented by combining our improved phosphate sensor part ([http://parts.igem.org/Part:BBa_K1139201 BBa_ K1139201]), our newly constructed cytokinin production part and two pre-existing parts which are the nitrate sensor part (Edinburgh 2009, [http://parts.igem.org/Part:BBa_K216005 BBa_K216005]) and the auxin production part (Imperial College 2011, [http://parts.igem.org/Part:BBa_K515100 BBa_K515100]) (Fig. 2-3-2). Since the existing phosphate sensor part (OUC-China 2012, [http://parts.igem.org/Part:BBa_K737024 BBa_K737024]) does not have sufficient data, we improved the part by using a different promoter. We also learned methods for quantitative analysis for cytokinin through bioassay of cucumber seed sprouts (Fig. 2-3-3) and through using ultra-performance liquid chromatography (UPLC).
In addition, to achieve the natural plants' temporal pattern for producing plant hormones in E. coli, we introduced an incoherent feed forward loop to our circuit, including a new hybrid promoter part ([http://parts.igem.org/Part:BBa_K1139150 BBa_K1139150]). Our mathematical model for this system predicted that we can create temporal patterns for the plant hormone production.
2. Phosphate sensor
We improved a phosphate sensor part by using the inducible promoter of the alkaline phosphatase gene (phoA) from E. coli (Dollard et al., 2003). This promoter is repressed by high phosphate concentrations (Fig. 2-3-4). To know more about the regulation of this promoter, please click here. We amplified the phoA promoter from E. coli (MG1655) and ligated this promoter into GFP part to construct the new part ([http://parts.igem.org/Part:BBa_K1139201 BBa_K1139201]). We then introduced this new part into E. coli (MG1655). Fig. 2-3-5 shows the result of our induction assay. It shows that the increase in phosphate concentration repressed the phoA promoter. Especially, we see that the phoA promoter is drastically repressed at phosphate concentrations of 100 to 200 µM. Though we also assayed OUC-China's phosphate sensor part including the phoB promoter ([http://parts.igem.org/Part:BBa_K737024 BBa_K737024]) by the same method as that for our phoA promoter assay, their part did not respond to the increase in phosphate concentration (Fig. 2-3-6). Thus, we concluded that we improved the phosphate sensor part. (Note that the scales of the vertical axis are different between the two results.) To know more about this assay, please see here.
From our results above, we determined parameters for the induction mechanism by fitting the results to the following Hill equation (Fig. 2-3-7). α denotes the maximum GFP expression rate in this construct. m denotes the phosphate concentration at which the GFP expression rate is half of α. β denotes the hill coefficient. Those parameters (Fig. 2-3-8) can be used in future modeling. Plants are reported to be in phosphate starvation when its concentration is below 1 mM (Hoagland et al., 1950). Our part can sense the concentration below 1 mM, too (Fig. 2-3-9). Therefore, our improved part is useful for our farming circuit. Moreover, a sensor for phosphate concentration is valuable for various studies in synthetic biology.
3. Cytokinin synthesis
In order to construct the cytokinin production part, we focused on AtIPT, a plant enzyme which catalyzes the synthesis of cytokinin. The mechanisms of cytokinin synthesis are shown in Fig. 2-3-10 (Takei et al., 2001). We ordered two DNA sequences of AtIPT (AtIPT4 and AtIPT7) derived from A. thaliana and constructed the part including one of them.
AtIPT, a plant enzyme, catalyzes the synthesis of iPRMP from DMAPP and AMP. According to the previous research, DMAPP and AMP are provided by the authentic metabolism in E. coli . It is also expected in the report that E. coli also has enzymes which catalyze the synthesis of iP or tZ (both sorts of cytokinin) from iPRMP.
4. Quantitative analysis for cytokinin by bioassay
Before constructing genetic parts for cytokinin synthesis, we learned methods for quantitative analysis for cytokinin through a bioassay of cucumber seed sprouts (Fletcher et al., 1971; Porra et al., 1989) and through using ultra-performance liquid chromatography (UPLC).
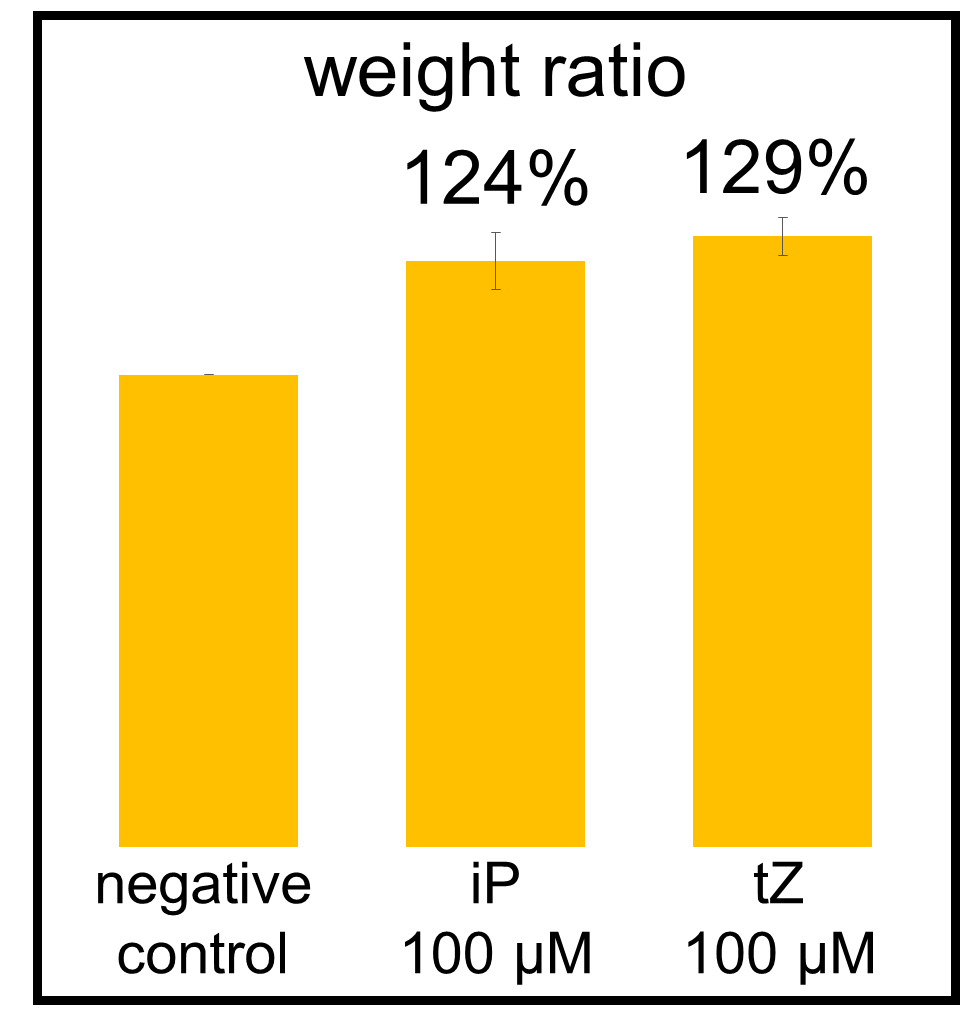
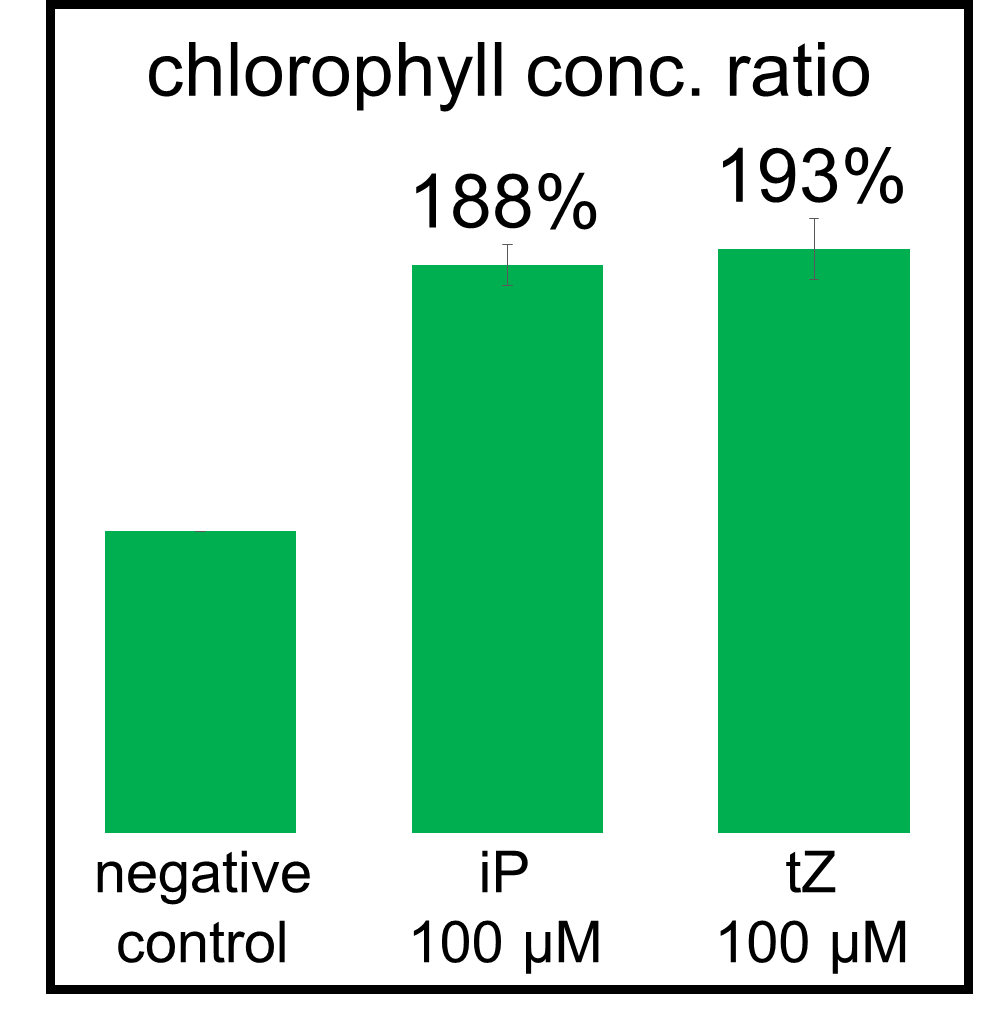
In our bioassay of cucumber seed sprouts, we learned how to detect cytokinin and found which concentration of cytokinin can act on plants. We planted the seeds and germinated for 5 days. Then, we cultivated the sprouts in standard cytokinin sample solutions. After 24 hours in the dark and 24 hours in the light, we measured the weight of the sprouts and also homogenized them to measure the concentration of chlorophyll (Fig. 2-3-12). Fig. 2-3-11 shows the result that the sprouts in the cytokinin solution are larger and greener than the negative controls, which are the sprouts in the solution without cytokinin. The quantitative results (Fig. 2-3-13 and Fig. 2-3-14) also show that cytokinin increases the weight of the sprouts and the concentration of chlorophyll (iP, tZ are both sorts of cytokinin). To know more about this assay, please see here.
iP : 6-(γ, γ-Dimethylallylamino) purine, tZ : trans-zeatin (both sorts of cytokinin)
The absorbances of the supernatant were read at 663.6 and 646.6 nm. Calculation of concentrations of the chlorophyll were carried out as described by Porra et al.,1989.
Through ultra-performance liquid chromatography (UPLC), we planned to confirm that our "Farmer E. coli" actually produces cytokinin. Before completing the system to produce cytokinin in E. coli, we determined the retention times of the authentic samples of iP and tZ by using UPLC. In addition, we confirmed that iP and tZ were able to be detected from the mixture of the E. coli culture medium and the cytokinin solution (Fig. 2-3-15). To see the detail method of this experiment, please click here.
5. Hybrid promoter for temporal pattern generation
In order to achieve the natural plants' temporal pattern for producing plant hormones in E. coli, we introduced an incoherent feed forward loop (Mangan et al., 2006) to our circuit, including a new hybrid promoter part ([http://parts.igem.org/Part:BBa_K1139150 BBa_K1139150]). Plants produce their hormones transiently rather than steadily (Takei et al., 2001). Moreover, continuous overexpression of hormones is harmful to plants (Thiman, 1937). Thus, we thought that it should be important to achieve this transient temporal pattern for producing plant hormones in E. coli, too. Our designed system with an incoherent feed forward loop is shown in Fig. 2-3-16. We newly developed the RM/lac hybrid promoter, which is activated by CI and repressed by LacI (Fig. 2-3-17). We planned to ligate a hormone synthase part downstream of this hybrid promoter. Our mathematical model (Fig. 2-3-18) shows the pulse wave which the temporal pattern for plant hormone production should achieve. While the RM/lac hybrid promoter activation by CI is a single-step reaction, the repression by LacI is a two-step reaction. Thus, the activation of the RM/lac hybrid promoter is faster than the repression. This time lag between the activation and the repression is important for generating a temporal pattern of plant hormone production (details about our mathematical model can be found here).
As a first step to achieve this system with an incoherent feed forward loop, we constructed a circuit shown in Fig. 2-3-19 to confirm that our new RM/lac hybrid promoter actually works. We set GFP as an output of RM/lac hybrid promoter and introduced the circuit into E. coli . Through an induction assay (Fig. 2-3-20), we saw that our new RM/lac hybrid promoter was actually activated by CI and repressed by LacI. Details about this assay can be found here.
6. Applications
Our project can be applied to studying the plants' response to external plant hormones. By applying the incoherent feed forward loop to our circuit, we can achieve pulse-generation of the output (Basu et al., 2003). Moreover, the pulse wave can be customized with changing the parameters, for example, intensities of promoters or various interactions. Various artificial genetic circuits which can achieve the temporal pattern of the output, such as incoherent feed forward loop, have been reported (Elowitz et al., 2000; Fung et al., 2005). By combining plant hormone synthesis genes to these artificial genetic circuits, we will be able to make E. coli produce plant hormones in various temporal patterns. Analysis of the plants' response to plant hormones given in temporal change leads to elucidate the abilities of plants in signal transduction. Findings in plant science using this strategy will be important for contributing to new approaches in farming. We believe that such basic research can help to solve worldwide food shortages. We hope to meet public expectations by realizing agriculture aid by bacteria, though there may be some difficulties.
 "
"