Team:UGent/Experiments
From 2013.igem.org
| Line 22: | Line 22: | ||
<h2> Experiment 2 </h2> | <h2> Experiment 2 </h2> | ||
<p> Next is finding a way to administer the toxin to the cells. This will be done by putting the gene coding for the toxin on a plasmid (a linear piece of DNA) and transferring this plasmid into the bacterial cell. In experiment 2 a plasmid containing ccdB (toxin) under control of a T7 promotor will be constructed. The T7 promoter allows us to control the expression of ccdB by regulating the amount of IPTG added to the cells. </p> | <p> Next is finding a way to administer the toxin to the cells. This will be done by putting the gene coding for the toxin on a plasmid (a linear piece of DNA) and transferring this plasmid into the bacterial cell. In experiment 2 a plasmid containing ccdB (toxin) under control of a T7 promotor will be constructed. The T7 promoter allows us to control the expression of ccdB by regulating the amount of IPTG added to the cells. </p> | ||
| + | <center><img src="https://static.igem.org/mediawiki/2013/4/44/UGent_2013_Exp1.jpg" width="700"></center> <br> | ||
<A HREF="https://static.igem.org/mediawiki/2013/4/46/UGent_2013_Experiment_2.pdf" target="_blank">Protocol experiment 2</A> | <A HREF="https://static.igem.org/mediawiki/2013/4/46/UGent_2013_Experiment_2.pdf" target="_blank">Protocol experiment 2</A> | ||
<h2> Experiment 3 </h2> | <h2> Experiment 3 </h2> | ||
Revision as of 14:16, 1 October 2013
|
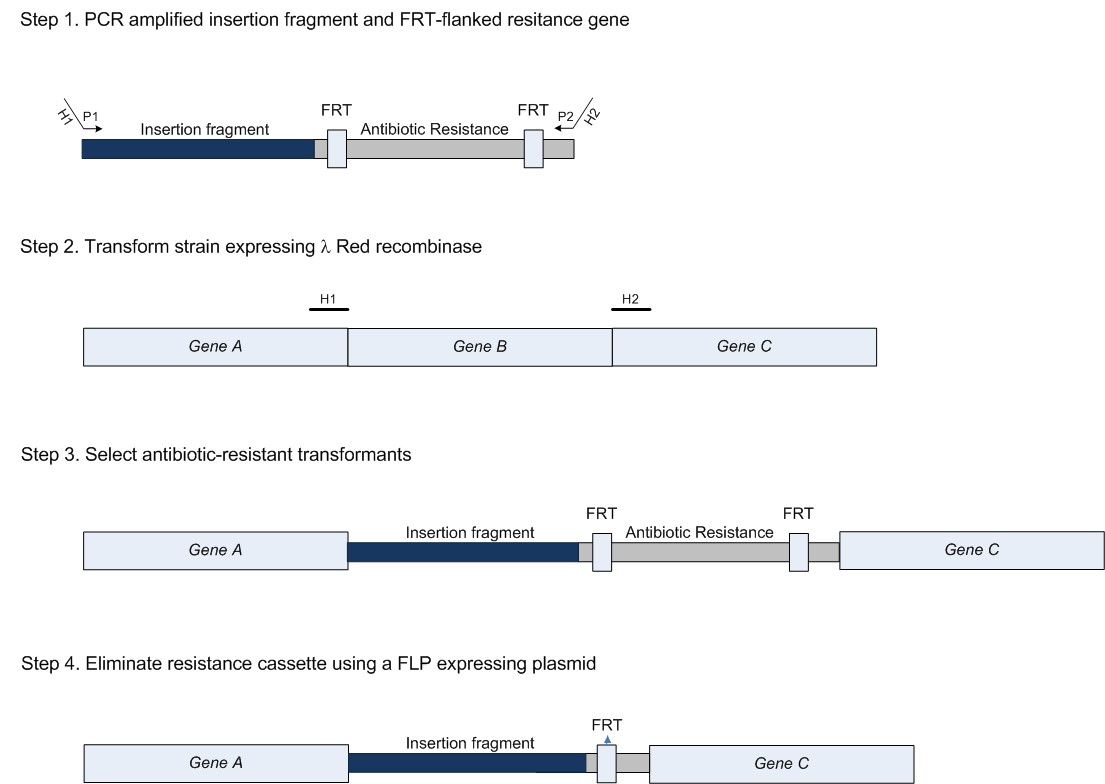
ExperimentsTo test our idea we are conducting 6 experiments. These are described below. Experiment 1Experiment 1 is the knock-in (KI) of the construct containing ccdA, GFP and the homologous regions (this was constructed beforehand). This will be done by using the method of Datsenko & Wanner [PNAS 2000], based on homologous recombination. The principle of the KO/KI method is depicted below:
Experiment 2Next is finding a way to administer the toxin to the cells. This will be done by putting the gene coding for the toxin on a plasmid (a linear piece of DNA) and transferring this plasmid into the bacterial cell. In experiment 2 a plasmid containing ccdB (toxin) under control of a T7 promotor will be constructed. The T7 promoter allows us to control the expression of ccdB by regulating the amount of IPTG added to the cells.  Protocol experiment 2 Experiment 3Once we have constructed the plasmid with ccdB and a T7 promoter, it has to be transferred into the cells in which CIChE will be performed. This “transformation” (the process of putting DNA into a cell) will be carried out in experiment 3. Protocol experiment 3Experiment 4Strains constructed in experiment 3 will be used to perform CIChE. Tandem gene replication of reporter protein GFP will be induced by replicating the antitoxin ccdA as a response on titration of the toxin ccdB. Titration of ccdB under inducible T7-promoter will be accomplished by different levels of IPTG [0.01 mM – 0.5mM] and different plasmid copy numbers [p5, p10 and p20]. Protocol experiment 4Experiment 5Experiment 6In this experiment, a new part, called BBa_K1105000, will be constructed by cloning Laciq-T7ccdB with standard Biobrick prefix and suffix in pSB1C3. Laciq-T7ccdB is derived from the plasmid p10-LacIq-T7ccdB, which originally comes from a mini F-plasmid positive strains (such as E. coli F+). The function of this part is to produce CcdB (in control of a T7 promotor), which interferes with the topoisomerase unit gyrA. Protocol experiment 6
|

Tweets van @iGEM_UGent |
||
|
|
 "
"