Team:BIOSINT Mexico/Safety: Kill switch
From 2013.igem.org
| (8 intermediate revisions not shown) | |||
| Line 6: | Line 6: | ||
---- | ---- | ||
---- | ---- | ||
| + | |||
| + | === Specific kill switch=== | ||
| + | |||
| + | [[File:Veneno.png|180px|left]] | ||
| + | |||
| + | :As an extra safety measure we adapt an specific kill switch in order than our probiotic was only capable to make the specific function for wich was design. This mechanism can be regulated through a sensor, so it will be activated only when it is desired. | ||
| + | |||
| + | :Plasmid recombination occurs naturally on the microbiota, so apply a kill switch could be dangerous for the others bacterial in the intestine. For that reason we development the concept of specificity, using a method that only could act in our lactobacillus strain. | ||
| + | |||
| + | So our construct could be DOUBLE SECURITY because it is strain specific and function specific. | ||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
| + | |||
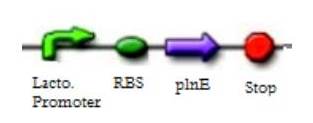
===Plantaricins=== | ===Plantaricins=== | ||
| + | ---- | ||
| - | Bacteriocins are ribosomally synthetized antimicrobial peptides produced by bacteria.4 Unlike antibiotics, bacteriocins have a narrow killing spectrum and affect only closely related strains.7 They can be classified in different groups depending on how they are produced, their components, or the bacteria they affect. | + | :Bacteriocins are ribosomally synthetized antimicrobial peptides produced by bacteria.4 Unlike antibiotics, bacteriocins have a narrow killing spectrum and affect only closely related strains.7 They can be classified in different groups depending on how they are produced, their components, or the bacteria they affect. |
| - | Plantaricins are one kind of bacteriocins which belong to the IIB group. This means they are heat stable, postranslationally unmodified non-lantibiotics, and are formed by two peptides. As their name suggests, they are produced by Lactobacillus plantarum.2 | + | |
| - | Recent studies have been developed on the identification of plantaricins, their activity, and interactions among each other. | + | :Plantaricins are one kind of bacteriocins which belong to the IIB group. This means they are heat stable, postranslationally unmodified non-lantibiotics, and are formed by two peptides. As their name suggests, they are produced by Lactobacillus plantarum.2 |
| - | Plantaricin A (PlnA) is a peptide that has both bacteriocin and pheromone activity.5 When it is produced, Plantaricin A activates five operons which are identified as plnABCD, plnEFI, plnJKLR, plnMNOP, and plnGHSTUV. The first operon codifies for pln A itself and for several proteins involved in the transduction process. Both pln EFI and pln JKLR codify for the two peptide Plantaricins. PlnE interacts with PlnF to form PlnEF while PlnJ interacts with PlnK to form PlnJK. Research suggests that certain types of strains are more susceptible to PlnEF, and others to PlnJK.1 The other subunits of this operon codify for immunity-like proteins that protect the producer against its own plantaricins. The activity of the fourth operon is still unknown, and the fifth operon has two ORF’s, one which results in PlnGH which encodes the machinery responsible of secreting and processing the Plantaricin precursors.3 | + | |
| - | PlnEF and PlnJK have strain specific antagonist activity, which is optimum when both subunits are produced equally.1 As part of our project, we will try to create a killing mechanism which produces a plantaricin (either one or the other) without producing the immunity proteins so our Lactobacillus will be susceptible to these substances once they are outside the cell membrane. As they are strain specific, they will not affect the other members of the human microbiota, nor will they be a potential risk for human health. | + | :Recent studies have been developed on the identification of plantaricins, their activity, and interactions among each other. |
| - | Generally speaking plantaricin will be produced inside the cell and exported outside the cell through ABC transporters.6 Once outside the cell, they will adopt an amphiphilic alpha helical structure which will allow them to interact with the cell membrane and permeabilize it.5 Up until now, the only known facts are that these plantaricins create pores in the membrane and dissipate both the transmembrane electrical potential (∆Ψ) and ∆pH gradient.6 | + | |
| - | + | :Plantaricin A (PlnA) is a peptide that has both bacteriocin and pheromone activity.5 When it is produced, Plantaricin A activates five operons which are identified as plnABCD, plnEFI, plnJKLR, plnMNOP, and plnGHSTUV. The first operon codifies for pln A itself and for several proteins involved in the transduction process. Both pln EFI and pln JKLR codify for the two peptide Plantaricins. PlnE interacts with PlnF to form PlnEF while PlnJ interacts with PlnK to form PlnJK. Research suggests that certain types of strains are more susceptible to PlnEF, and others to PlnJK.1 The other subunits of this operon codify for immunity-like proteins that protect the producer against its own plantaricins. The activity of the fourth operon is still unknown, and the fifth operon has two ORF’s, one which results in PlnGH which encodes the machinery responsible of secreting and processing the Plantaricin precursors.3 | |
| + | |||
| + | :PlnEF and PlnJK have strain specific antagonist activity, which is optimum when both subunits are produced equally.1 As part of our project, we will try to create a killing mechanism which produces a plantaricin (either one or the other) without producing the immunity proteins so our Lactobacillus will be susceptible to these substances once they are outside the cell membrane. As they are strain specific, they will not affect the other members of the human microbiota, nor will they be a potential risk for human health. | ||
| + | |||
| + | :Generally speaking plantaricin will be produced inside the cell and exported outside the cell through ABC transporters.6 Once outside the cell, they will adopt an amphiphilic alpha helical structure which will allow them to interact with the cell membrane and permeabilize it.5 Up until now, the only known facts are that these plantaricins create pores in the membrane and dissipate both the transmembrane electrical potential (∆Ψ) and ∆pH gradient.6 | ||
| + | |||
| + | [[File:ks.JPG|center|200px]] | ||
| + | |||
| + | ===Safety forms=== | ||
---- | ---- | ||
| + | |||
| + | |||
| + | |||
| + | |||
| Line 25: | Line 55: | ||
1Anderssen, Erlend L.; Diep, Dzung Bao; Nes, Ingolf F.; Eijsink, Vincent G. H. and Nissen-Meyer, Jon. (1998). Antagonistic Activity of Lactobacillus plantarum C11: Two New Two-Peptide Bacteriocins, Plantaricins EF and JK, and the Induction Factor Plantaricin A. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC106311/ on April 10, 2013. | 1Anderssen, Erlend L.; Diep, Dzung Bao; Nes, Ingolf F.; Eijsink, Vincent G. H. and Nissen-Meyer, Jon. (1998). Antagonistic Activity of Lactobacillus plantarum C11: Two New Two-Peptide Bacteriocins, Plantaricins EF and JK, and the Induction Factor Plantaricin A. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC106311/ on April 10, 2013. | ||
| + | |||
2Cintas, L.M.; Casaus, M.P.; Herranz, C.; Nes, I.F. and Hernández, P.E. (2013). Review: Bacteriocins of Lactic Acid Bacteria. [Online]. Retrieved from http://fst.sagepub.com/content/7/4/281.abstract on April 10, 2013. | 2Cintas, L.M.; Casaus, M.P.; Herranz, C.; Nes, I.F. and Hernández, P.E. (2013). Review: Bacteriocins of Lactic Acid Bacteria. [Online]. Retrieved from http://fst.sagepub.com/content/7/4/281.abstract on April 10, 2013. | ||
| + | |||
3Diep, D. B.; Havarstein, L. S. and Nes, I.F. (1996). Characterization of the locus responsible for the bacteriocin production in Lactobacillus plantarum C11. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC178213/ on April 10, 2013. | 3Diep, D. B.; Havarstein, L. S. and Nes, I.F. (1996). Characterization of the locus responsible for the bacteriocin production in Lactobacillus plantarum C11. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC178213/ on April 10, 2013. | ||
| + | |||
4Dobson, A., Cotter, P.D., Ross, R.P., Hill, C. (2012) Bacteriocin Production: a Probiotic Trait? [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255625/ on April, 10, 2013. | 4Dobson, A., Cotter, P.D., Ross, R.P., Hill, C. (2012) Bacteriocin Production: a Probiotic Trait? [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255625/ on April, 10, 2013. | ||
| + | |||
5Hildeng Hauge, H., Mantzilas, D., Eijsink, V.G.H., Nissen-Meyer, J. (1999) Membrane-Mimicking Entities Induce Structuring of the Two-Peptide Bacteriocins Plantaricin E/F and Plantaricin J/K. [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93438/ on April, 10, 2013. | 5Hildeng Hauge, H., Mantzilas, D., Eijsink, V.G.H., Nissen-Meyer, J. (1999) Membrane-Mimicking Entities Induce Structuring of the Two-Peptide Bacteriocins Plantaricin E/F and Plantaricin J/K. [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93438/ on April, 10, 2013. | ||
| + | |||
6Moll, Gert N.; van den Akker, Emile; Hauge, Havard H.; Nissen-Meyer, Jon; Nes, Ingolf F.; Konings, Wil N. and Driessen, Arnold J. M. (1999). Complementary and Overlapping Selectivity of the Two-Peptide Bacteriocins Plantaricin EF and JK. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93971/ on April 10, 2013. | 6Moll, Gert N.; van den Akker, Emile; Hauge, Havard H.; Nissen-Meyer, Jon; Nes, Ingolf F.; Konings, Wil N. and Driessen, Arnold J. M. (1999). Complementary and Overlapping Selectivity of the Two-Peptide Bacteriocins Plantaricin EF and JK. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93971/ on April 10, 2013. | ||
| + | |||
7Riley, M.A., Wertz,J.E. (2002) BACTERIOCINS: Evolution, Ecology, and Application. [On-line] Retrieved from http://www.bio.umass.edu/biology/riley/sites/www.bio.umass.edu.biology.riley/files/2002c%20Bacteriocins-%20Evolution,%20Ecology,%20and%20Application_002.pdf on April, 10, 2013. | 7Riley, M.A., Wertz,J.E. (2002) BACTERIOCINS: Evolution, Ecology, and Application. [On-line] Retrieved from http://www.bio.umass.edu/biology/riley/sites/www.bio.umass.edu.biology.riley/files/2002c%20Bacteriocins-%20Evolution,%20Ecology,%20and%20Application_002.pdf on April, 10, 2013. | ||
Latest revision as of 03:49, 28 September 2013
Safety: Kill switch
Specific kill switch
- As an extra safety measure we adapt an specific kill switch in order than our probiotic was only capable to make the specific function for wich was design. This mechanism can be regulated through a sensor, so it will be activated only when it is desired.
- Plasmid recombination occurs naturally on the microbiota, so apply a kill switch could be dangerous for the others bacterial in the intestine. For that reason we development the concept of specificity, using a method that only could act in our lactobacillus strain.
So our construct could be DOUBLE SECURITY because it is strain specific and function specific.
Plantaricins
- Bacteriocins are ribosomally synthetized antimicrobial peptides produced by bacteria.4 Unlike antibiotics, bacteriocins have a narrow killing spectrum and affect only closely related strains.7 They can be classified in different groups depending on how they are produced, their components, or the bacteria they affect.
- Plantaricins are one kind of bacteriocins which belong to the IIB group. This means they are heat stable, postranslationally unmodified non-lantibiotics, and are formed by two peptides. As their name suggests, they are produced by Lactobacillus plantarum.2
- Recent studies have been developed on the identification of plantaricins, their activity, and interactions among each other.
- Plantaricin A (PlnA) is a peptide that has both bacteriocin and pheromone activity.5 When it is produced, Plantaricin A activates five operons which are identified as plnABCD, plnEFI, plnJKLR, plnMNOP, and plnGHSTUV. The first operon codifies for pln A itself and for several proteins involved in the transduction process. Both pln EFI and pln JKLR codify for the two peptide Plantaricins. PlnE interacts with PlnF to form PlnEF while PlnJ interacts with PlnK to form PlnJK. Research suggests that certain types of strains are more susceptible to PlnEF, and others to PlnJK.1 The other subunits of this operon codify for immunity-like proteins that protect the producer against its own plantaricins. The activity of the fourth operon is still unknown, and the fifth operon has two ORF’s, one which results in PlnGH which encodes the machinery responsible of secreting and processing the Plantaricin precursors.3
- PlnEF and PlnJK have strain specific antagonist activity, which is optimum when both subunits are produced equally.1 As part of our project, we will try to create a killing mechanism which produces a plantaricin (either one or the other) without producing the immunity proteins so our Lactobacillus will be susceptible to these substances once they are outside the cell membrane. As they are strain specific, they will not affect the other members of the human microbiota, nor will they be a potential risk for human health.
- Generally speaking plantaricin will be produced inside the cell and exported outside the cell through ABC transporters.6 Once outside the cell, they will adopt an amphiphilic alpha helical structure which will allow them to interact with the cell membrane and permeabilize it.5 Up until now, the only known facts are that these plantaricins create pores in the membrane and dissipate both the transmembrane electrical potential (∆Ψ) and ∆pH gradient.6
Safety forms
REFERENCES:
1Anderssen, Erlend L.; Diep, Dzung Bao; Nes, Ingolf F.; Eijsink, Vincent G. H. and Nissen-Meyer, Jon. (1998). Antagonistic Activity of Lactobacillus plantarum C11: Two New Two-Peptide Bacteriocins, Plantaricins EF and JK, and the Induction Factor Plantaricin A. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC106311/ on April 10, 2013.
2Cintas, L.M.; Casaus, M.P.; Herranz, C.; Nes, I.F. and Hernández, P.E. (2013). Review: Bacteriocins of Lactic Acid Bacteria. [Online]. Retrieved from http://fst.sagepub.com/content/7/4/281.abstract on April 10, 2013.
3Diep, D. B.; Havarstein, L. S. and Nes, I.F. (1996). Characterization of the locus responsible for the bacteriocin production in Lactobacillus plantarum C11. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC178213/ on April 10, 2013.
4Dobson, A., Cotter, P.D., Ross, R.P., Hill, C. (2012) Bacteriocin Production: a Probiotic Trait? [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255625/ on April, 10, 2013.
5Hildeng Hauge, H., Mantzilas, D., Eijsink, V.G.H., Nissen-Meyer, J. (1999) Membrane-Mimicking Entities Induce Structuring of the Two-Peptide Bacteriocins Plantaricin E/F and Plantaricin J/K. [On-line] Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93438/ on April, 10, 2013.
6Moll, Gert N.; van den Akker, Emile; Hauge, Havard H.; Nissen-Meyer, Jon; Nes, Ingolf F.; Konings, Wil N. and Driessen, Arnold J. M. (1999). Complementary and Overlapping Selectivity of the Two-Peptide Bacteriocins Plantaricin EF and JK. [Online]. Retrieved from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC93971/ on April 10, 2013.
7Riley, M.A., Wertz,J.E. (2002) BACTERIOCINS: Evolution, Ecology, and Application. [On-line] Retrieved from http://www.bio.umass.edu/biology/riley/sites/www.bio.umass.edu.biology.riley/files/2002c%20Bacteriocins-%20Evolution,%20Ecology,%20and%20Application_002.pdf on April, 10, 2013.
 "
"