Team:Goettingen/Team/DAC
From 2013.igem.org
(→DAC Team) |
(→DAC Team) |
||
| Line 65: | Line 65: | ||
Diadenylate cyclases catalyze the condensation reaction of two molecules ATP to a single molecule c-di-AMP while releasing two pyrophosphate molecules consisting of the β-γ-phosphates of each ATP (Fig. 2). In order to analyze the cyclase activity of DacA in vitro, we performed an assay in which these pyrophosphate molecules were used as evidence for the reaction. Among other things, the assay included a pyrophosphatase that cleaves the pyrophosphate molecule in free phosphate molecules, malachite-green and molybdate. Malachite-green forms a complex with free phosphate molecules and molybdate that can be measured due to its absorption spectrum. With this in vitro assay the concentration of free phosphate molecules and thus the conversion rate of ATP to c-di-AMP was analyzed. | Diadenylate cyclases catalyze the condensation reaction of two molecules ATP to a single molecule c-di-AMP while releasing two pyrophosphate molecules consisting of the β-γ-phosphates of each ATP (Fig. 2). In order to analyze the cyclase activity of DacA in vitro, we performed an assay in which these pyrophosphate molecules were used as evidence for the reaction. Among other things, the assay included a pyrophosphatase that cleaves the pyrophosphate molecule in free phosphate molecules, malachite-green and molybdate. Malachite-green forms a complex with free phosphate molecules and molybdate that can be measured due to its absorption spectrum. With this in vitro assay the concentration of free phosphate molecules and thus the conversion rate of ATP to c-di-AMP was analyzed. | ||
| + | |||
| + | Our results confirmed that DacA Lmo2120 acts as an active enzyme in the presence of a divalent cation, ATP and a buffer system at pH 8 (in vitro), however, the catalysis rate in vivo seems to be much more efficient. | ||
| + | |||
| + | <html><a href="https://static.igem.org/mediawiki/2013/7/7f/Goe-dac-fig-2.png" target="_blank"><img src="https://static.igem.org/mediawiki/2013/7/7f/Goe-dac-fig-2.png" width="750"/></a></html> | ||
| + | |||
| + | Fig. 2. '''Cyclic di-AMP production and degradation.''' Cyclic di-AMP is produced from two molecules of ATP by diadenylyl cyclase (DAC) enzymes and degraded to pApA by phosphodiasterases. (Edited from Corrigan and Gründling, 2013) | ||
| + | |||
| + | |||
| + | Finally, we are coming to the core of our project, the protein structure of DacA! The molecular structure of DacA can lead to the discovery of new antibacterial substance classes ''in silico'', which later can be used to treat patients. | ||
| + | |||
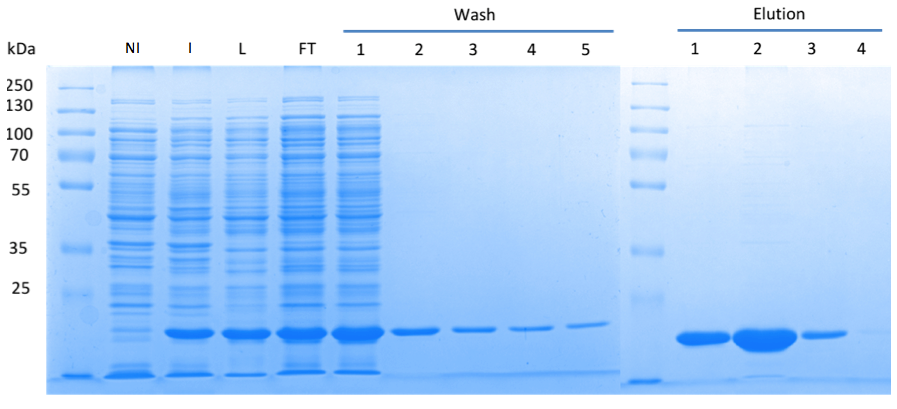
| + | In order to purify a large amount of this protein, our BioBrick [http://partsregistry.org/Part:BBa_K1045003 BBa_K1045003] with an N-terminal Strep-tag was expressed in ''E. coli'' under the control of a T7-promoter. The expression was again induced by supplementing the growth medium with IPTG. The high expression was confirmed by SDS gel electrophoresis with a resulting thick band at the size of 20 kDa (Fig. 3). The ''E. coli'' strain was grown in large scale with the result that 10μl of cell cultures were lysed and purified (Fig. 3). | ||
| + | |||
| + | <html><a href="https://static.igem.org/mediawiki/2013/f/fe/Goe-dac-fig-3.png" target="_blank"><img src="https://static.igem.org/mediawiki/2013/f/fe/Goe-dac-fig-3.png" width="750" /></a></html> | ||
| + | |||
| + | Fig. 3. '''SDS gel showing overexpression and purification of DacA.''' (A) Lane 1: Thermo Scientific PageRuler Plus Prestained Protein Ladder; NI: Non-induced, Clone contained the cyclase domain of which the activity was not induced; I: Induced, Clone contained the cyclase domain of which the activity was induced by addition of isopropyl-ß-D-1-thiogalactopyranoside (IPTG); L: Lysate; FT: Flow-through; After wash 1 our protein was purified. | ||
| + | |||
| + | Having dialyzed and concentrated the eluted protein, we obtained a protein concentration of 10 mg/ml, which was sufficient to perform the crystallization reaction. Very nice crystals were already received in a trial when testing different media (Fig. 4A). In order to find the perfect supplements for growing our crystals, the whole procedure was repeated. The crystal yields an x-ray diffraction pattern with a resolution of 2,8 Å (Fig. 4B,C). The dataset was measured at the EMBL Hamburg Beamline P13 at the PETRA III synchrotron on the DESY campus. | ||
| + | |||
| + | Finally, we were also able to obtain the protein structure (Fig. 5)! The structure shows a globular protein with a distinct ATP-binding cleft. The ribbon model demonstrates the general structure composed of α-helices and β-sheets. | ||
| + | |||
| + | <html><a href="https://static.igem.org/mediawiki/2013/a/a8/Goe-dac-fig-4.png" target="_blank"><img src="https://static.igem.org/mediawiki/2013/a/a8/Goe-dac-fig-4.png" width="750"></a></html> | ||
=== === | === === | ||
Revision as of 16:17, 1 October 2013
 "
"