Team:Dundee/Project/DetectorPKRC
From 2013.igem.org
Kyleharrison (Talk | contribs) |
Kyleharrison (Talk | contribs) |
||
| Line 52: | Line 52: | ||
<p>We hope that when the microcystin binds to PP1 this will still result in activation of the downstream pathways controlled by the native PrkC receptor. Additionally, we hope to have our <i> B. subtilis</i> strain constitutively expressing green fluorescent protein so that when it is relieved from dormancy it will fluoresce and this will hopefully be detectable with our electronic Moptopus device.</p> | <p>We hope that when the microcystin binds to PP1 this will still result in activation of the downstream pathways controlled by the native PrkC receptor. Additionally, we hope to have our <i> B. subtilis</i> strain constitutively expressing green fluorescent protein so that when it is relieved from dormancy it will fluoresce and this will hopefully be detectable with our electronic Moptopus device.</p> | ||
| + | |||
| + | <h2>Progress so far...</h2> | ||
| + | <p>We are currently in the process of cloning this receptor and we are having some difficulty. We have successfully cloned the N-terminal part of the receptor and are currently in the process of adding on the PP1s which we will be doing by suicide ligation. The final step after adding the PP1s will be to add on the C-terminal domain. </p><br><br> | ||
</div> | </div> | ||
| Line 71: | Line 74: | ||
<p><br><i>Figure 2. .</i></p> | <p><br><i>Figure 2. .</i></p> | ||
</div> | </div> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
</div> | </div> | ||
Latest revision as of 16:28, 14 August 2013
The Detector
Aim: To engineer the B. subtilis receptor PrkC to respond to microcystin
B. subtilis forms resistant structures called spores in order to survive harsh environmental conditions. In order for the spores to recognise that the conditions have again become favourable for growth the spores have to monitor the extracellular environment. This is done via a number of inner-membrane receptors described as germinant receptors. PrkC is an example of a germinant receptor and it binds to cell wall associated peptides.
Sensing cell wall peptides & conditions that are permissive for growth
Actively growing cells turnover cell wall components and these can thus be found in the extracellular milieu. So by sensing cell wall components, through the PrkC receptor, the spore can tell that other cells are growing in the nearby environment. This is how the PrkC receptor can signal to the spore that conditions are permissive for growth.
PrkC receptor activation
PrkC receptor activation triggers a process called germination which is the conversion of the spore back into an actively growing cell.
PrkC receptor
The PrkC receptor has 4 extracellular domains PASTA 1, 2 and 3 which are capped by a C-terminal domain and this sits on the outside of the spore inner membrane. The 3 PASTA domains are implicated in binding of the cell wall components and are thus described as the ligand binding domains (fig 1).
PrkC receptor to detect microcystin
We hope to detect microcystin by replacing the 3 ligand binding domains with the human protein- protein phosphatase 1 (PP1) (fig 2).
We hope that when the microcystin binds to PP1 this will still result in activation of the downstream pathways controlled by the native PrkC receptor. Additionally, we hope to have our B. subtilis strain constitutively expressing green fluorescent protein so that when it is relieved from dormancy it will fluoresce and this will hopefully be detectable with our electronic Moptopus device.
Progress so far...
We are currently in the process of cloning this receptor and we are having some difficulty. We have successfully cloned the N-terminal part of the receptor and are currently in the process of adding on the PP1s which we will be doing by suicide ligation. The final step after adding the PP1s will be to add on the C-terminal domain.
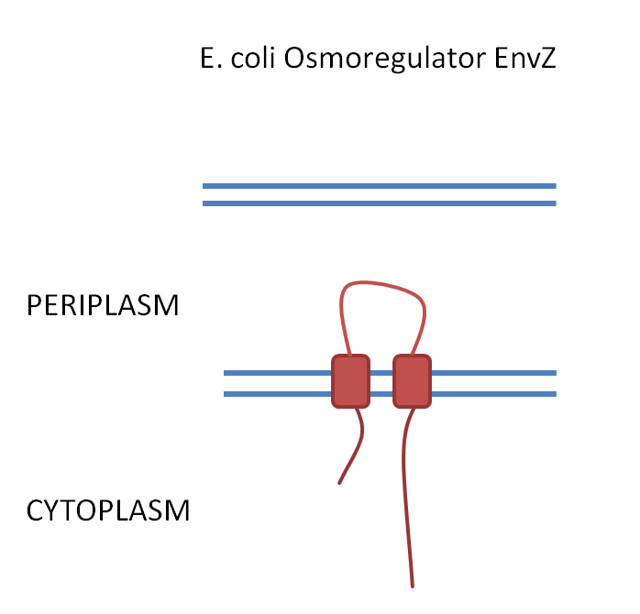
Figure 1. .
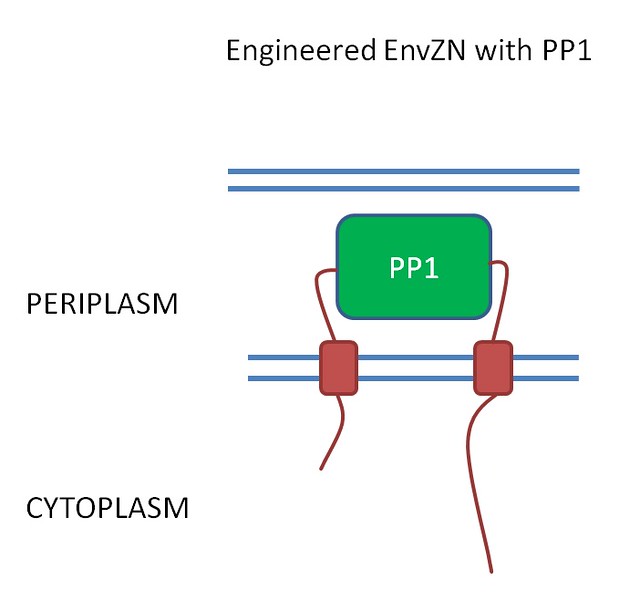
Figure 2. .
 "
"
