Team:Newcastle/Project/shape shifting
From 2013.igem.org

Contents |
Shape Shifting
Background
Most bacteria have evolved to have a cell wall, and those which do not, have cell walled ancestry (Trachtenberg 1998)(Errington 2013). The cell wall is a rigid structure which protects the bacteria from the variety of environmental hazards such as mechanical stress, osmotic rupture and lysis. The cell wall often serves as a docking point to many proteins including various receptors and adherence sites. In addition to these features, the cell wall provides the cell with a rigid boundary and helps bacteria to acquire and preserve their shape. In Bacillus subtilis a group of proteins termed Penicillin Binding Proteins (Pbp),along with other proteins, usually anchored in the cell wall, along with other proteins, are involved in the formation of the rod shape. When the cells lose their cell wall they automatically lose these proteins to the environment as they are being made. The cells lose their support and turn into a sphere as it is the most energetically favourable state (ratio of surface area to volume is minimal, and membrane curvature is more-or-less constant).
The idea
It has been previously observed that these cell wall-less bacteria can become elongated and 'squeeze' into the spaces with a smaller diameter than theirs. This fact sparked our interest and led us to ask some questions. L-forms are an improved secretory machine; the cell wall hinders excretion of some proteins. Could L-forms be used as a delivery vehicle in small or out-of-reach areas? Out of reach areas include intercellular spaces and micro-cracks in solid materials, which are smaller than 1 μm. B.subtilis in L-form state, can grow to large sizes before they divide. Is it possible to fill spaces of various shapes and sizes with L-forms?
To research into these areas, we were to manipulate L-forms microfluidics. Microfluidics allows manipulation of single cells via precise movement of fluids in the micro scale. Microfluidic chips were designed on autoCAD which were to be used to produce silicon wafer master moulds. Unfortunately these were not created in time.
The idea was to use directed flow within the microfluidic wafer to physically manoeuvre L-form cells into designed chambers of different shapes, shown in figure 1. The membranes of the L-forms were to be stained with FM.595 and in this way we would be able to visualise whether the L-forms were able to adopt different shapes.
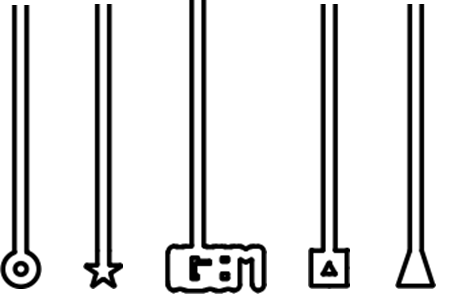 Figure 1. The design of the chambers which L-forms would be forced into. The L-forms would potentially adopt the shape of the terminal part of the chambers.
Figure 1. The design of the chambers which L-forms would be forced into. The L-forms would potentially adopt the shape of the terminal part of the chambers.
Modelling
To show the flexibility of the L-forms we have planned to trap them in a microfluidics chamber whose shape differs from that of a normal L-form e.g. a star, square or triangle.
Before we even had begun to design the experiments, to illustrate the process which we predict to occur inside of the terminal chamber as the cell grows we have constructed a predicted model of the cell behaviour as it grows inside a square, based on the knowledge that we have about the processes inside the cell which are involved in membrane synthesis and growth. We would like to thank Dr. David Swailes from the School of Mechanical Engineering at Newcastle University for his massive help with the mathematical side of the modelling. We couldn't have done what we have without his help.
For the purposes of the study the complex model of the growing cell inside of the confined space can be broken down to simpler models of the system at two phases. The first phase would be a constantly growing cell, followed by a model of the cell, gradually adopting the shape of the boundaries.
The full description of the model can be found on this page.
This model can be improved by conducting experiments which would allow us to find parameters such as the rate of membrane synthesis and maximum membrane torsion, or to make the model free of a few assumptions i.e. find out the maximum size the cell could grow to before dividing, measure the effects of nutrient depletion, and develop a protocol which would allow us to test our hypothesis on the cells which are in the growth phase of their cell cycle. It can also be expanded in terms of factoring in the bending energy of the membrane in a particular shape and then comparing the models of different shapes and evaluating which would me more readily assumed.
It would also be interesting to see whether the state of the cell (i.e. l-form or rod) before it enters the chamber would have any difference on the way it fills the space.
References
Trachtenberg S. (1998) Mollicutes-wall-less bacteria with internal cytoskeletons. Journal of Structural Biology. 124, 244-256.
Errington J. (2013) L-form bacteria, cell walls and the origins of life. Open Biology, 3, 120143
 "
"