Team:SydneyUni Australia/Project/Parts
From 2013.igem.org


Project Registry Parts
Lorem ipsum dolor sit amet, consectetuer adipiscing elit, sed diam nonummy nibh euismod tincidunt ut laoreet dolore magna aliquam erat volutpat. Ut wisi enim ad minim veniam, quis nostrud exerci tation ullamcorper suscipit lobortis nisl ut aliquip ex ea commodo consequat. Duis autem vel eum iriure dolor in hendrerit in vulputate velit esse molestie consequat, vel illum dolore eu feugiat nulla facilisis at vero eros et accumsan et iusto odio dignissim qui blandit praesent luptatum zzril delenit augue duis dolore te feugait nulla facilisi. Nam liber tempor cum soluta nobis eleifend option congue nihil imperdiet doming id quod mazim placerat facer possim assum. Typi non habent claritatem insitam; est usus legentis in iis qui facit eorum claritatem. Investigationes demonstraverunt lectores legere me lius quod ii legunt saepius. Claritas est etiam processus dynamicus, qui sequitur mutationem consuetudium lectorum. Mirum est notare quam littera gothica, quam nunc putamus parum claram, anteposuerit litterarum formas humanitatis per seacula quarta decima et quinta decima. Eodem modo typi, qui nunc nobis videntur parum clari, fiant sollemnes in futurum.
Gibson
Gibson Assembly *should* make for a much easier, simpler, rapid assembly of different genes than conventional PCR and cloning, plus there’s much more flexibility for optimisation through gene synthesis.
- The assembly works on fragments of DNA with ~30bp of overlapping sequence, which is exposed as 5’ single-stranded overhangs by an exonuclease. A ligase joins the overlapping regions and a polymerase fills in any gaps left by the exonuclease. These enzymes can all work together in a single reaction tube with many different overlapping fragments, making the assembly a very, very simple activity. Gibson Assembly is based on the older technique of [http://nar.oxfordjournals.org/content/32/12/e98.full| PCR Assembly], with the similar reliance on the initial construction of 200+bp fragments from smaller oligos, but with a greater degree of sequence fidelity due to less polymerase activity.
- Here’s a great introduction from IDT’s magazine, [http://www.idtdna.com/pages/decoded/decoded-articles/core-concepts/decoded/2012/01/10/isothermal-assembly-quick-easy-gene-construction| DECODED], and a more in-depth webinar.
- If you’re historically-minded or want more detail, try the [http://diyhpl.us/~bryan/papers2/bio/venter/Enzymatic%20assembly%20of%20DNA%20molecules%20up%20to%20several%20hundred%20kilobases.pdf| original paper] in which Gibson Assembly was described - or one of the coolest and most famous applications of Gibson, building a [http://www.ncbi.nlm.nih.gov/books/NBK84435/| synthetic genome].
Design
Choice of genes
Mox, chloroethanol dehydrogenase
- In sketches of our project (February-March), we planned to use Mox as the alcohol dehydrogenase converting chloroethanol to chloroacetaldehyde. After a little more research we discovered that this enzyme requires the co-factor PQQ, and unfortunately for us, this co-factor cannot be synthesised by E. Coli and would have required us to add at least one more gene to our final construct (eg, PQQ synthase, as in Khairnar et al, 2003).
- Mox had seemed like an obvious choice because it would be sourced from Xanthobacter autotrophicus GJ10, the most well-documented DCA-degrader ([http://mic.sgmjournals.org/content/133/1/85.full.pdf).| Janssen et al, 1987])
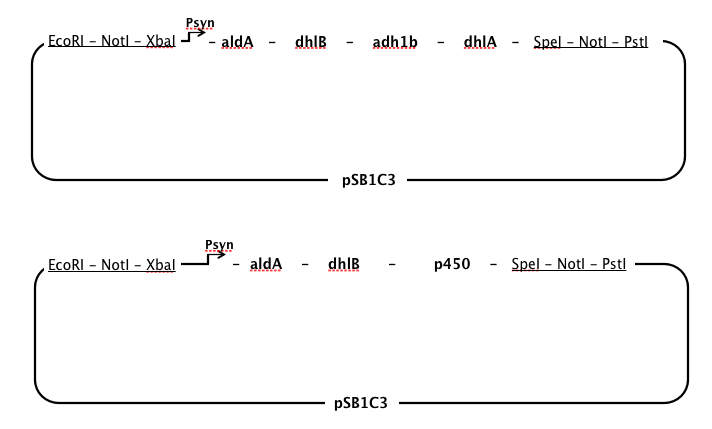
aldA, chloroacetaldehyde dehydrogenase, dhlB, haloacid dehalogenase, and dhlA, haloalkane dehalogenase
- We used a sequence for aldA from Xanthobacter autrophicus GJ10, but used sequences for dhlB and dhlA from a different strain, Xanthobacter autrophicus EL4, which was isolated and characterised in the Coleman lab. When our Gibson Assembly ran into problems, we continued to work with dhlB and dhlA from EL4 in our lab and submitted these two genes as parts.
- These genes are shared by a few different species of bacteria that degrade DCA (Janssen et al, 1994), and most have been conventionally characterised by extraction and heterologous expression of a single gene at a time.
ToMO, toluene-o-xylene monooxygenase
- Early in our project we showed that ToMO can degrade DCA. This would not only eliminate the need for something like PQQ-synthase, but also make for a shorter, less complex pathway involving just 3 enzymes (a monooxygenase, dehydrogenase and dehalogenase).
- We initially planned to synthesise ToMO, allowing us to remove forbidden restriction sites in the process (EcoRI and PstI). However, due to the sheer size of the monooxygenase cluster (~5kb) we could not afford to have this gene and all of our others synthesised.
- The gene we worked with initially (and the sequence we would have optimised before synthesis) was itself already the result of protein engineering by [http://fenske.che.psu.edu/faculty/wood/group/publications/pdf/ToMO%20TCE%20mutagenesis%20Vardar[1].pdf| Varder & Wood (2005)].
p450, cytochrome p450 monooxygenase
- Early in 2013 it was discovered that a monooxygenase from a Polaromonas sp. (JS666) was responsible for the initial steps of DCE and DCA degradation by heterologous expression in E. Coli ([http://aem.asm.org/content/79/7/2263.short)| Nishino et al, 2013]). As far as we know, this enzyme effectively substitutes for ToMO in our pathway. Due to the shorter length of the three-gene p450 complex (~3kb) we decided to synthesise this enzyme instead of ToMO.
- The strain that carries this enzyme was first isolated from chloroethene contaminated sites by our primary supervisor, Nick Coleman, in the Stone Age ([http://aem.asm.org/content/68/6/2726.full| Coleman et al, 2002]).
adh1b2, human liver alcohol dehydrogenase
- Human liver alcohol dehydrogenases have been shown to be active on a broad range of substrates, including haloalcohols and haloaldehydes ([http://pubs.acs.org/doi/pdf/10.1021/bi00870a034| Blair & Vallee, 1966]). They’ve also been expressed in E. Coli before (eg, [http://onlinelibrary.wiley.com/store/10.1111/j.1530-0277.1993.tb00849.x/asset/j.1530-0277.1993.tb00849.x.pdf?v=1&t=hm3swoes&s=48df141f12d9dea31a52423907067e2f1e28bdee&systemMessage=Wiley+Online+Library+will+be+unavailable+for+approximately+4+hours+between+09%3A00+EDT+and+14%3A00+EDT+on+Saturday%2C+28+September+2013+as+we+make+upgrades+to+improve+our+services+to+you.+There+will+also+be+some+delays+to+online+publishing+between+25+to+28+September+2013.+We+apologize+for+the+inconvenience+and+appreciate+your+patience.+Thank+you+for+using+Wiley+Online+Library%21| Zheng et al, 1993]) using cDNA, but we were able to use a modified mRNA sequence from GenBank ([http://www.ncbi.nlm.nih.gov/nuccore/BC033009| BC033009.2]). We were also able to choose between many types and classes of human liver enzyme, so we picked the one with the greatest turnover on ethanol as substrate (Edenberg, 2007).
Order of gene expression
- We chose to have a single, strong constitutive promoter in our pathway (see below, promoter), so it made sense to place our genes in order from last to first in order that at every step in the pathway there would be an excess of the enzyme required for the substrate, thus minimising the build-up of toxic intermediates.
- However, chloroacetaldehyde appears to be the most toxic of the intermediates produced while DCA is degraded (Janssen et al, 1994). It seemed necessary to position the enzyme specific to aldA immediately after the promoter to ensure that chloroacetaldehyde was rapidly removed from the cell.
References
- Blair, A. H., & Vallee, B. L. (1966). Some Catalytic Properties of Human Liver Alcohol Dehydrogenase*. Biochemistry, 5(6), 2026-2034.
- Edenberg, H. J. (2007). The genetics of alcohol metabolism: role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Research & Health.
- Janssen, D. B., Pries, F., & Van der Ploeg, J. R. (1994). Genetics and biochemistry of dehalogenating enzymes. Annual Reviews in Microbiology, 48(1), 163-191.
- Khairnar, N. P., Misra, H. S., & Apte, S. K. (2003). Pyrroloquinoline–quinone synthesized in< i> Escherichia coli</i> by pyrroloquinoline–quinone synthase of< i> Deinococcus radiodurans</i> plays a role beyond mineral phosphate solubilization. Biochemical and biophysical research communications, 312(2), 303-308.
- Vardar, G., & Wood, T. K. (2005). Protein engineering of toluene-o-xylene monooxygenase from Pseudomonas stutzeri OX1 for enhanced chlorinated ethene degradation and o-xylene oxidation. Applied microbiology and biotechnology, 68(4), 510-517.
- Zheng, C. F., Wang, T. T., & Weiner, H. (1993). Cloning and Expression of the Full‐Length cDNAS Encoding Human Liver Class 1 and Class 2 Aldehyde Dehydrogenase. Alcoholism: Clinical and Experimental Research, 17(4), 828-831.
 "
"