Team:ETH Zurich/Experiments 3
From 2013.igem.org
| Line 12: | Line 12: | ||
<p align="justify">Numerous enzyme substrates have been designed following this natural product example, giving rise to many colored phenols that are used to detect enzyme activities.<br><br> | <p align="justify">Numerous enzyme substrates have been designed following this natural product example, giving rise to many colored phenols that are used to detect enzyme activities.<br><br> | ||
As shown in Figure 1, some of the hydrolases used in the Colisweeper reporter system can catalyze hydrolysis of various substrates, with different chromophores that give rise to a wide range of colors. This variety of substrates and color outputs enables change of positions and function of these enzymes. Other possible substrates that can be used for the enzymes of the Colisweeper reporter system can be found in the [https://2013.igem.org/Team:ETH_Zurich/Experiments_7 Hydrolases] section. | As shown in Figure 1, some of the hydrolases used in the Colisweeper reporter system can catalyze hydrolysis of various substrates, with different chromophores that give rise to a wide range of colors. This variety of substrates and color outputs enables change of positions and function of these enzymes. Other possible substrates that can be used for the enzymes of the Colisweeper reporter system can be found in the [https://2013.igem.org/Team:ETH_Zurich/Experiments_7 Hydrolases] section. | ||
| - | + | The set of enzyme-substrate pairs chosen for Colisweeper is described below.</p> | |
<br clear="all"/> | <br clear="all"/> | ||
<h1>Acetyl esterase (Aes)</h1> | <h1>Acetyl esterase (Aes)</h1> | ||
| - | [[File:Aes_colored.png|frame|right|200px| <b>Figure 2.</b> Liquid culture | + | [[File:Aes_colored.png|frame|right|200px| <b>Figure 2.</b> Liquid culture of the triple knockout <i>Escherichia coli</i> strain overexpressing Aes; after addition of magenta-butyrate.]] |
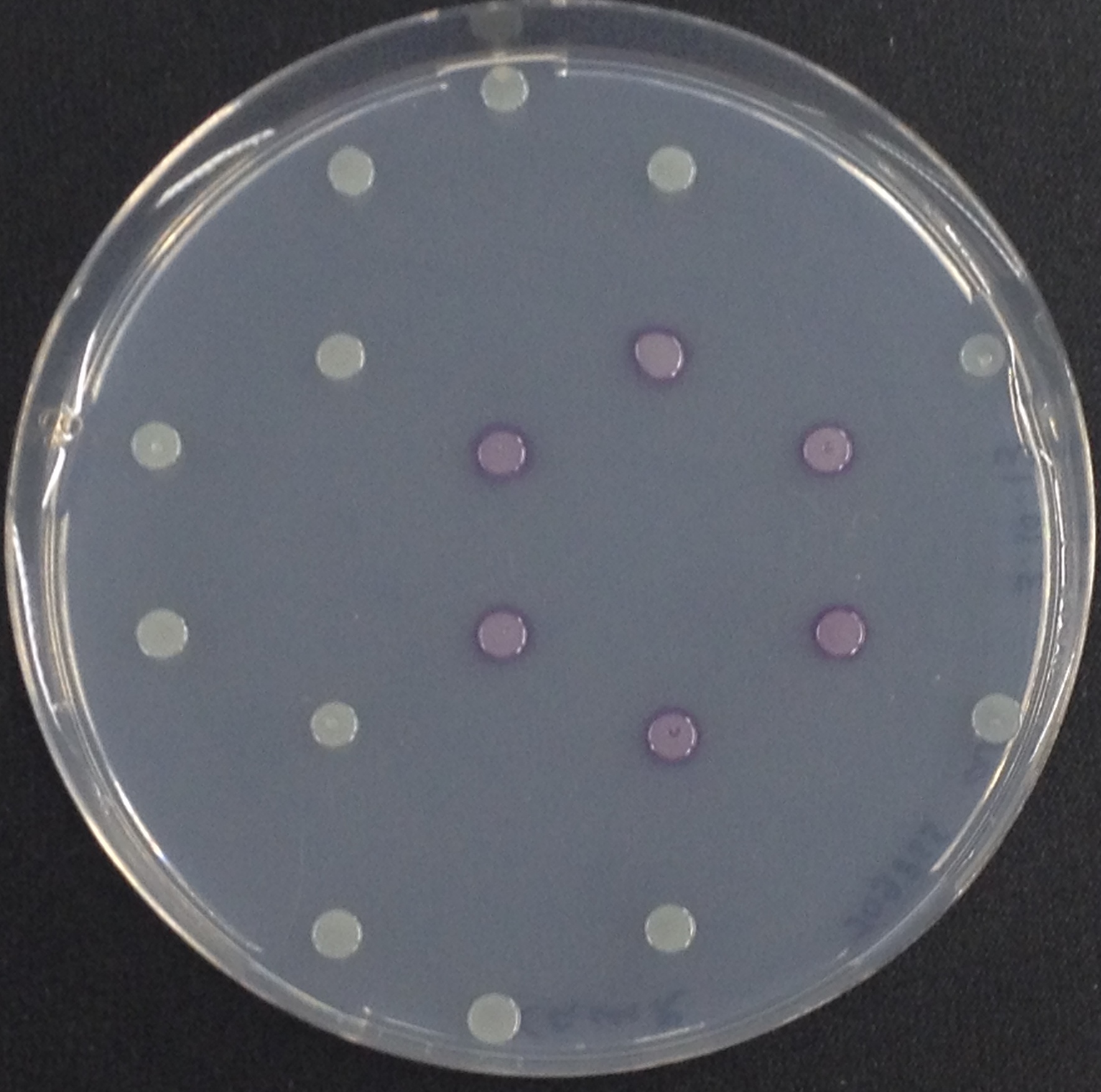
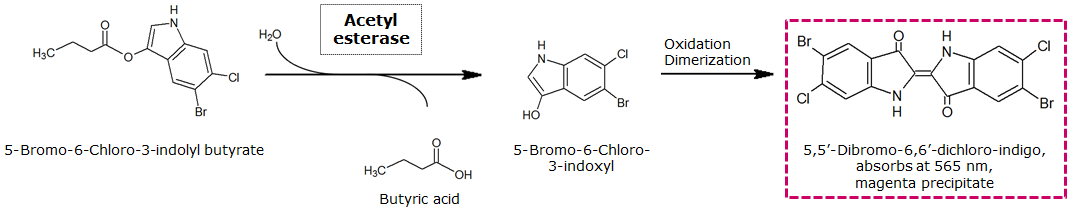
| - | To assess color development after reaction of the enzyme with the chromogenic substrate, a liquid culture of our triple knockout <i>Escherichia coli</i> strain overexpressing Aes was grown until an OD<sub>600</sub> of 0.4 - 0.6 was reached before addition of magenta butyrate to a final concentration of 250 µM. To study the color development in the actual Colisweeper game setup, colonies were plated by pipetting 1.5 µl of triple knockout <i>Escherichia coli</i> liquid culture (OD<sub>600</sub> of 0.4 - 0.6) on an M9 agar plate. Addition of 1.5 µl of 20 mM magenta butyrate onto each colony results in color generation visible after a few minutes at room temperature. | + | To assess color development after reaction of the enzyme with the chromogenic substrate, a liquid culture of our triple knockout <i>Escherichia coli</i> strain overexpressing Aes was grown until an OD<sub>600</sub> of 0.4 - 0.6 was reached before addition of 5-Bromo-6-Chloro-3-indolyl butyrate (magenta butyrate) to a final concentration of 250 µM. To study the color development in the actual Colisweeper game setup, colonies were plated by pipetting 1.5 µl of triple knockout <i>Escherichia coli</i> liquid culture (OD<sub>600</sub> of 0.4 - 0.6) on an M9 agar plate. Addition of 1.5 µl of 20 mM magenta butyrate onto each colony results in color generation visible after a few minutes at room temperature. |
| - | [[File:Aes and Magenta Butyrate.jpeg|thumb|right|200px| <b>Figure 3.</b> Colonies of ''Escherichia coli'' overexpressing the | + | [[File:Aes and Magenta Butyrate.jpeg|thumb|right|200px| <b>Figure 3.</b> Colonies of ''Escherichia coli'' overexpressing the Aes. Colonies produce magenta color after addition of magenta butyrate.]]<br> |
| - | [[File:AesReaction.png|thumb|center|800px| <b>Figure 4.</b> | + | [[File:AesReaction.png|thumb|center|800px| <b>Figure 4.</b> Aes catalyzed hydrolysis reaction of magenta butyrate.]]<br> |
<br><b>Enzyme kinetics</b> | <br><b>Enzyme kinetics</b> | ||
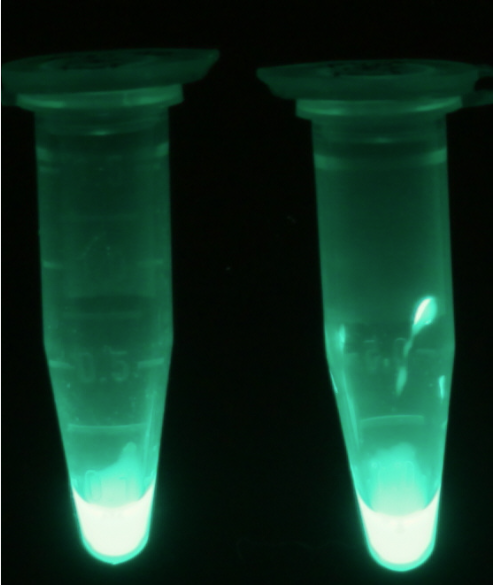
| - | [[File:Aes_fluorescent.png|thumb|right|200px|<b>Figure 5.</b> Cell lysate of <i>Escherichia coli</i> overexpressing | + | [[File:Aes_fluorescent.png|thumb|right|200px|<b>Figure 5.</b> Cell lysate of <i>Escherichia coli</i> overexpressing Aes after reaction with 4-MU-butyrate.]] |
For the kinetics assay, the ''aes'' encoded protein was tested with the fluorescent substrate 4-MU-butyrate. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λ<sub>Ex</sub> 365 nm. | For the kinetics assay, the ''aes'' encoded protein was tested with the fluorescent substrate 4-MU-butyrate. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λ<sub>Ex</sub> 365 nm. | ||
[[File:AES_reaction.png|thumb|center|400px|<b>Figure 6. Enzymatic reaction of Aes with 4-MU-butyrate.</b>]]<br> | [[File:AES_reaction.png|thumb|center|400px|<b>Figure 6. Enzymatic reaction of Aes with 4-MU-butyrate.</b>]]<br> | ||
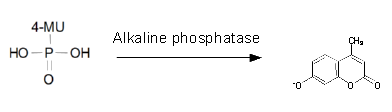
| - | To characterize the | + | To characterize the enzyme, we conducted fluorometric assays to obtain K<sub>m</sub> values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λ<sub>Ex</sub> 365 nm and λ<sub>Em</sub> 445 nm. |
| - | The obtained data was evaluated and | + | The obtained data was evaluated and fitted to Michaelis-Menten-Kinetics with SigmaPlot™. |
| - | [[File:Hydrolase AES.png|thumb|center|400px|<b>Figure 7.</b> Michaelis-Menten-Kinetics of cell lysate from <i>Escherichia coli</i> overexpressing | + | [[File:Hydrolase AES.png|thumb|center|400px|<b>Figure 7.</b> Michaelis-Menten-Kinetics of cell lysate from <i>Escherichia coli</i> overexpressing Aes with the chromogenic substrate: plots velocity versus substrate concentration (10 μL, 30 μL, 100 μL, 200 μL, 400 μL) in 20 mM Tris buffer of pH 8. A kinetic value for K<sub>m</sub> obtained by fitting the raw data to standard the Michaelis Menten equation; K<sub>m</sub> = 31.5 ± 12.5 μM. All assays were carried out in triplicates, results are presented as means.]] |
<br clear="all"/> | <br clear="all"/> | ||
| Line 34: | Line 34: | ||
<h1>Alkaline phosphatase (PhoA)</h1> | <h1>Alkaline phosphatase (PhoA)</h1> | ||
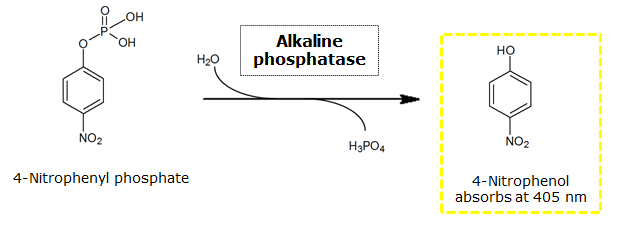
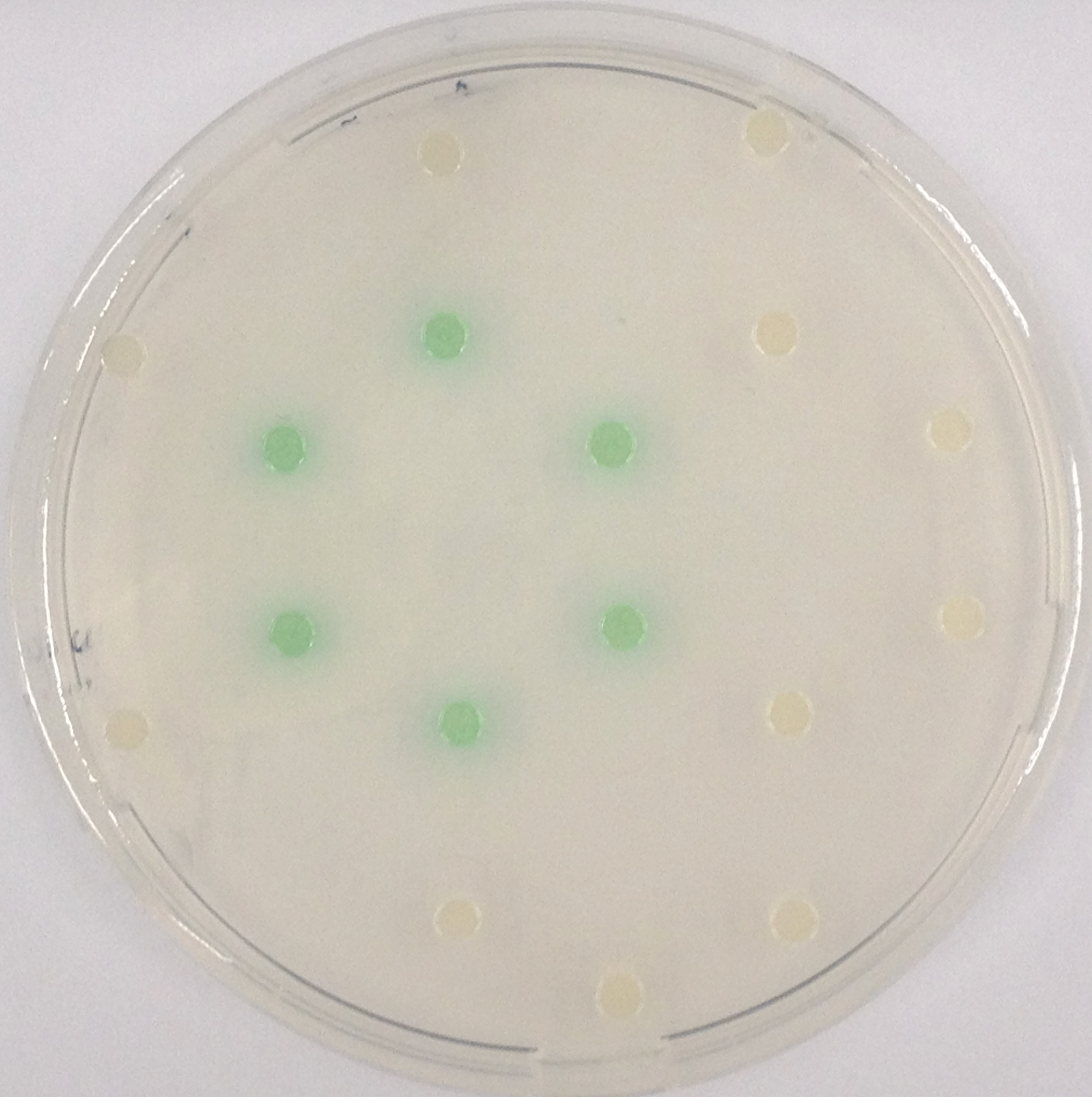
| - | To test the functionality of this | + | To test the functionality of this PhoA, a liquid culture of <i>Escherichia coli</i> overexpressing this hydrolase was grown until an OD<sub>600</sub> of 0.4 - 0.6 was reached before addition of ''p''-nitrophenyl phosphate (pNPP) to a final concentration of 5 µM. This substrate gives rise to yellow color after hydrolysis by the PhoA. In the Colisweeper game, substrates are pipetted onto colonies. Figure 10 shows color development on colonies five minutes after addition of 1.5 µl of a 0.5 M pNPP solution.<br clear="all"/> |
[[File:PhoA_PhoA_his_compared_colored.png|thumb|left|200px|<b>Figure 8.</b> Liquid culture from <i>Escherichia coli</i> overexpressing PhoA or PhoA-His respectively after reacting with pNPP. The negative control tube contains liquid culture without pNPP.]] | [[File:PhoA_PhoA_his_compared_colored.png|thumb|left|200px|<b>Figure 8.</b> Liquid culture from <i>Escherichia coli</i> overexpressing PhoA or PhoA-His respectively after reacting with pNPP. The negative control tube contains liquid culture without pNPP.]] | ||
| - | [[File:PhoA and pNPP.jpeg|thumb|right|200px| <b>Figure 10.</b> Colonies of ''Escherichia coli'' overexpressing the ''Citrobacter'' | + | [[File:PhoA and pNPP.jpeg|thumb|right|200px| <b>Figure 10.</b> Colonies of ''Escherichia coli'' overexpressing the ''Citrobacter'' PhoA. Colonies produce yellow color after addition of pNPP.]] |
| - | [[File:PhoAReaction2.png|thumb|600px|center|<b>Figure 9.</b> Reaction of | + | [[File:PhoAReaction2.png|thumb|600px|center|<b>Figure 9.</b> Reaction of PhoA catalyzed hydrolysis of pNPP.]] |
<br clear="all"/> | <br clear="all"/> | ||
| Line 52: | Line 52: | ||
To characterize the enzymes, we conducted fluorometric assays to obtain K<sub>m</sub> values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λ<sub>Ex</sub> 365 nm and λ<sub>Em</sub> 445 nm. | To characterize the enzymes, we conducted fluorometric assays to obtain K<sub>m</sub> values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λ<sub>Ex</sub> 365 nm and λ<sub>Em</sub> 445 nm. | ||
The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™: | The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™: | ||
| - | [[File:PhoA and phoAHIS kinetics.png|frame|center|500px|<b>Figure 14.</b> Michaelis-Menten-Kinetics of | + | [[File:PhoA and phoAHIS kinetics.png|frame|center|500px|<b>Figure 14.</b> Michaelis-Menten-Kinetics of cell lysate from <i>Escherichia coli</i> overexpressing the ''Citrobacter'' PhoA: plots velocity versus substrate concentration (2.5 μL, 5 μL, 10 μL, 30 μL, 60 μL, 120 μL, 240 μL) in 20 mM Tris buffer of pH 8. A kinetic value for K<sub>m</sub> obtained by fitting the raw data to standard the Michaelis Menten equation; K<sub>m</sub> = 105.9 ± 5.3 μM. All assays were carried out in triplicates, results are presented as means.]] |
<br clear="all"/><br> | <br clear="all"/><br> | ||
| Line 59: | Line 59: | ||
<h1>β-Galactosidase (LacZ)</h1> | <h1>β-Galactosidase (LacZ)</h1> | ||
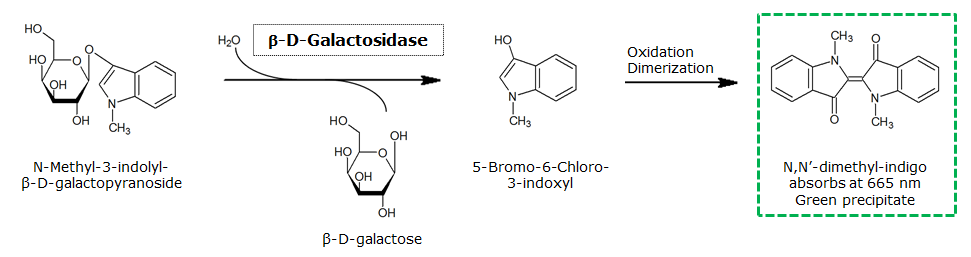
| - | The β-Galactosidase is natively expressed in the triple knockout Escherichia coli strain used in Colisweeper. Therefore, this protein catalyzes hydrolysis of the flagging substrate, which is Green-β-D- | + | The β-Galactosidase is natively expressed in the triple knockout Escherichia coli strain used in Colisweeper. Therefore, this protein catalyzes hydrolysis of the flagging substrate, which is N-Methyl-3-indolyl-β-D-Galactopyranoside (Green-β-D-Gal) and produces a green color upon cleavage of the chromophore. |
| - | [[File:LacZ and GreenGal.jpeg|thumb|right|200px| <b>Figure 3.</b> Colonies of our triple knockout strain ''Escherichia coli''. Colonies natively produce LacZ, and addition of Green-β-D- | + | [[File:LacZ and GreenGal.jpeg|thumb|right|200px| <b>Figure 3.</b> Colonies of our triple knockout strain ''Escherichia coli'' in the hexagonal grid setup. Colonies natively produce LacZ, and addition of Green-β-D-Gal gives rise to green color.]] |
[[File:LacZReaction.png|center]] | [[File:LacZReaction.png|center]] | ||
<br clear="all"/> | <br clear="all"/> | ||
| Line 66: | Line 66: | ||
<h1>β-Glucuronidase (GusA)</h1> | <h1>β-Glucuronidase (GusA)</h1> | ||
| - | [[File:GusA_colored.png|frame|right|<b>Figure 1.</b> Liquid culture from <i>Escherichia coli</i> overexpressing | + | [[File:GusA_colored.png|frame|right|<b>Figure 1.</b> Liquid culture from <i>Escherichia coli</i> overexpressing GusA; after reaction with Salmon-Gluc.]] |
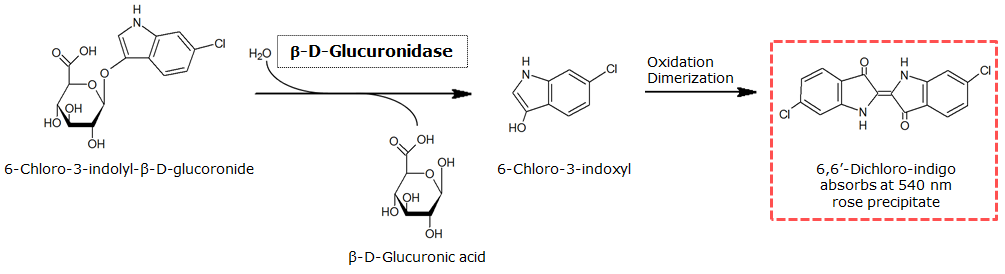
| - | To test the functionality of | + | To test the functionality of GusA, a liquid culture of <i>Escherichia coli</i> overexpressing GusA was grown until an OD<sub>600</sub> of 0.4 - 0.6 was reached before addition of 6-Chloro-3-indolyl-β-glucuronide (Salmon-Gluc), a substrate for GusA which produces a rose/salmon color after cleavage. The final concentration of the substrate in liquid culture was 1 mM and the conversion of this substrate was done by overnight incubation at 37°C. <br clear="all"/> |
| - | [[File:GusAReaction.png|frame|center|<b>Figure 2.</b> Enzymatic reaction of | + | [[File:GusAReaction.png|frame|center|<b>Figure 2.</b> Enzymatic reaction of GusA with the chromogenic substrate Salmon-Gluc.]] |
<br><b>Enzyme kinetics</b> | <br><b>Enzyme kinetics</b> | ||
| - | [[File:GusA_fluorescent.png|thumb|right|<b>Figure 3.</b> Cell lysate from <i>Escherichia coli</i> overexpressing | + | [[File:GusA_fluorescent.png|thumb|right|<b>Figure 3.</b> Cell lysate from <i>Escherichia coli</i> overexpressing GusA, after reaction with 4-MU-β-D-Glucuronide.]] |
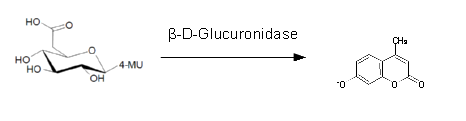
For the kinetics assay, we tested both ''gusA'' and ''gusA''-HIS encoded proteins with the fluorescent substrate 4-MU-β-D-Glucuronide. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λ<sub>Ex</sub> 365 nm. | For the kinetics assay, we tested both ''gusA'' and ''gusA''-HIS encoded proteins with the fluorescent substrate 4-MU-β-D-Glucuronide. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λ<sub>Ex</sub> 365 nm. | ||
| - | [[File:gusa_fluorescent_reaction.png|frame|center|<b>Figure 4.</b> Enzymatic reaction of | + | [[File:gusa_fluorescent_reaction.png|frame|center|<b>Figure 4.</b> Enzymatic reaction of GusA with the fluorogenic substrate 4-MU-β-D-Glucuronide.]] |
To characterize the enzymes, we conducted fluorometric assays to obtain K<sub>m</sub> values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λ<sub>Ex</sub> 365 nm and λ<sub>Em</sub> 445 nm. | To characterize the enzymes, we conducted fluorometric assays to obtain K<sub>m</sub> values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λ<sub>Ex</sub> 365 nm and λ<sub>Em</sub> 445 nm. | ||
The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™: | The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™: | ||
| Line 80: | Line 80: | ||
<h1>β-N-Acetylglucosaminidase (NagZ)</h1> | <h1>β-N-Acetylglucosaminidase (NagZ)</h1> | ||
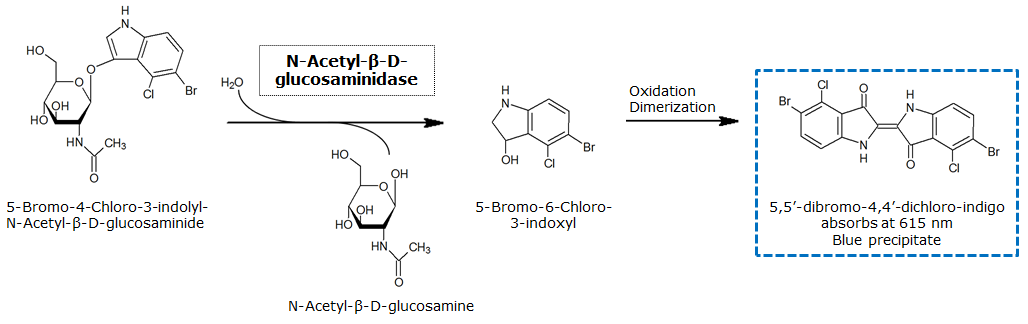
| - | In Colisweeper, the | + | In Colisweeper, the NagZ is constitutively expressed in the mine cells. Addition of 5-Bromo-4-Chloro-3-indolyl-β-N-acetylglucosaminide (X-GluNAc) would result in a blue color precipitate, as shown in Figure Bla. However, addition of the X-GluNAc substrate that we have did not result in any color production. Control assays with ''p''-nitrophenyl-β-N-acetylglucosaminide (pNP-GluNAc) on the other hand gave rise to yellow color, which confirms that NagZ is expressed in our mine cells. |
[[File:NagZReaction.png|frame|center|<b>Figure 2.</b> β-N-Acetylglucosaminidase catalyzed hydrolysis of the chromogenic substrate X-β-N-acetylglucosaminide.]] | [[File:NagZReaction.png|frame|center|<b>Figure 2.</b> β-N-Acetylglucosaminidase catalyzed hydrolysis of the chromogenic substrate X-β-N-acetylglucosaminide.]] | ||
<br clear="all"/> | <br clear="all"/> | ||
Revision as of 03:11, 5 October 2013
Contents |
Enzyme-substrate reactions

To generate visible output by adding substrates to colonies in Colisweeper, we made use of orthogonal enzyme-substrate reactions. A set of chromogenic substrates was chosen to produce different colors depending on the abundant hydrolases and thereby to uncover the identity of each colony to the player.
Chromogenic substrates incorporate a chromophore whose absorbance properties change after the enzyme reaction, and the color signal produced then is directly related to the enzyme-catalyzed reaction.
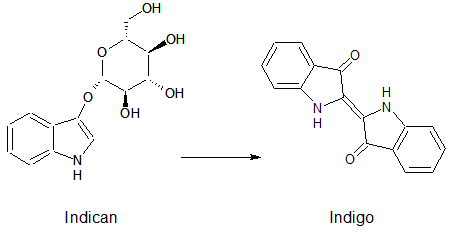
Indican is a chromogenic glycoside hydrolase substrate which belongs to a family of natural glycosides found in plants. Cleavage of the glycosidic bond forms an unstable hydroxyindole intermediate, which dimerizes by oxidation to form indigo as a blue precipitate:
Numerous enzyme substrates have been designed following this natural product example, giving rise to many colored phenols that are used to detect enzyme activities.
As shown in Figure 1, some of the hydrolases used in the Colisweeper reporter system can catalyze hydrolysis of various substrates, with different chromophores that give rise to a wide range of colors. This variety of substrates and color outputs enables change of positions and function of these enzymes. Other possible substrates that can be used for the enzymes of the Colisweeper reporter system can be found in the Hydrolases section.
The set of enzyme-substrate pairs chosen for Colisweeper is described below.
Acetyl esterase (Aes)
To assess color development after reaction of the enzyme with the chromogenic substrate, a liquid culture of our triple knockout Escherichia coli strain overexpressing Aes was grown until an OD600 of 0.4 - 0.6 was reached before addition of 5-Bromo-6-Chloro-3-indolyl butyrate (magenta butyrate) to a final concentration of 250 µM. To study the color development in the actual Colisweeper game setup, colonies were plated by pipetting 1.5 µl of triple knockout Escherichia coli liquid culture (OD600 of 0.4 - 0.6) on an M9 agar plate. Addition of 1.5 µl of 20 mM magenta butyrate onto each colony results in color generation visible after a few minutes at room temperature.
Enzyme kinetics
For the kinetics assay, the aes encoded protein was tested with the fluorescent substrate 4-MU-butyrate. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λEx 365 nm.
To characterize the enzyme, we conducted fluorometric assays to obtain Km values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λEx 365 nm and λEm 445 nm. The obtained data was evaluated and fitted to Michaelis-Menten-Kinetics with SigmaPlot™.
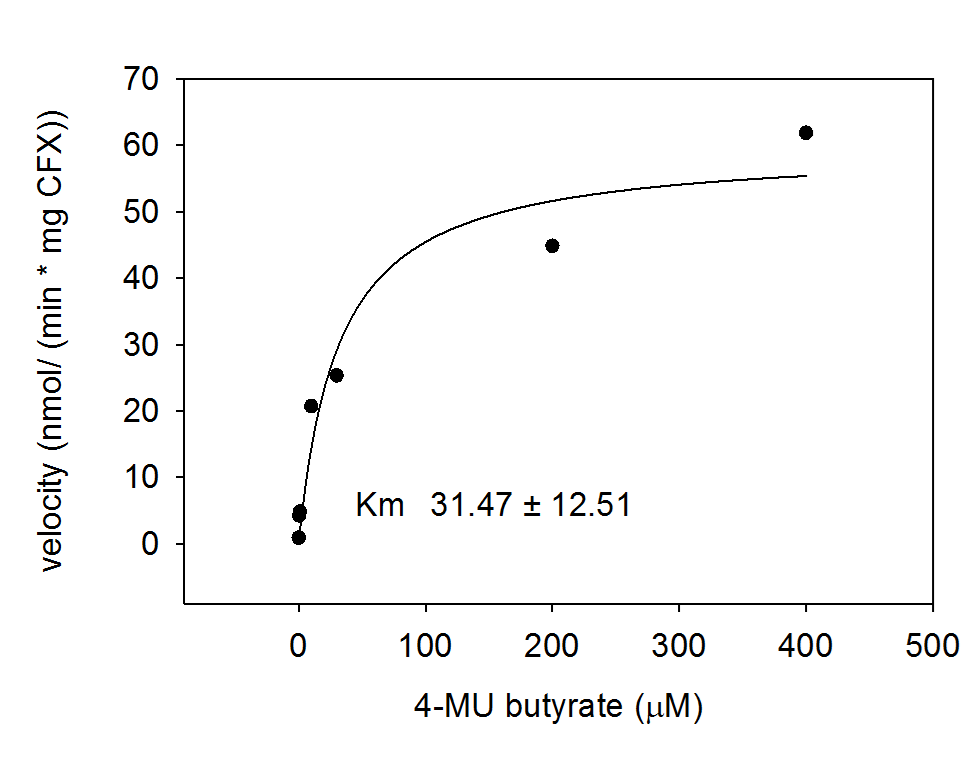
Alkaline phosphatase (PhoA)
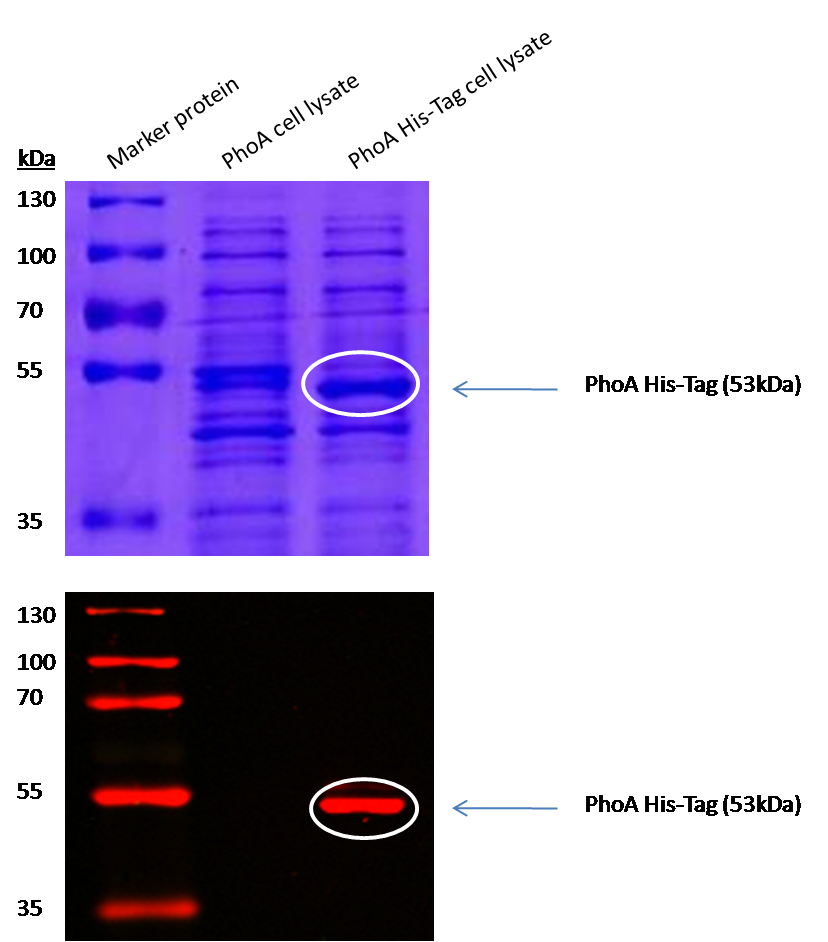
To test the functionality of this PhoA, a liquid culture of Escherichia coli overexpressing this hydrolase was grown until an OD600 of 0.4 - 0.6 was reached before addition of p-nitrophenyl phosphate (pNPP) to a final concentration of 5 µM. This substrate gives rise to yellow color after hydrolysis by the PhoA. In the Colisweeper game, substrates are pipetted onto colonies. Figure 10 shows color development on colonies five minutes after addition of 1.5 µl of a 0.5 M pNPP solution.
Characterization of PhoA-HIS tagged protein
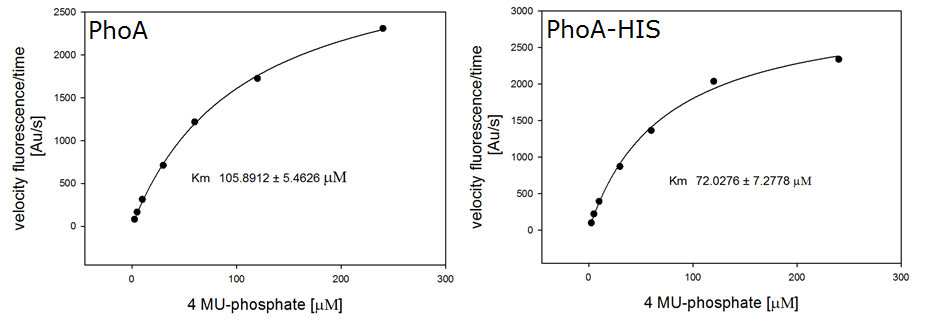
Enzyme kinetics
For the kinetics assay, we tested both the Citrobacter phoA and phoA-HIS encoded proteins with the fluorescent substrate 4-MU-phosphate. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λEx 365 nm.
To characterize the enzymes, we conducted fluorometric assays to obtain Km values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λEx 365 nm and λEm 445 nm. The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™:
β-Galactosidase (LacZ)
The β-Galactosidase is natively expressed in the triple knockout Escherichia coli strain used in Colisweeper. Therefore, this protein catalyzes hydrolysis of the flagging substrate, which is N-Methyl-3-indolyl-β-D-Galactopyranoside (Green-β-D-Gal) and produces a green color upon cleavage of the chromophore.
β-Glucuronidase (GusA)
To test the functionality of GusA, a liquid culture of Escherichia coli overexpressing GusA was grown until an OD600 of 0.4 - 0.6 was reached before addition of 6-Chloro-3-indolyl-β-glucuronide (Salmon-Gluc), a substrate for GusA which produces a rose/salmon color after cleavage. The final concentration of the substrate in liquid culture was 1 mM and the conversion of this substrate was done by overnight incubation at 37°C.
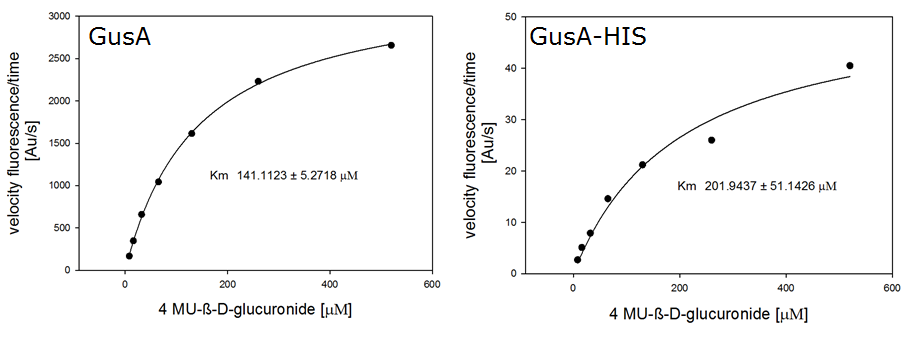
Enzyme kinetics
For the kinetics assay, we tested both gusA and gusA-HIS encoded proteins with the fluorescent substrate 4-MU-β-D-Glucuronide. The picture on the right was taken with a common single lens reflex camera mounted on a dark hood at λEx 365 nm.
To characterize the enzymes, we conducted fluorometric assays to obtain Km values. Bacterial cells were harvested and lysed, and the cell free extract (CFX) was then collected for the fluorometric assay. The properly diluted CFX was measured on a 96 well plate in triplicates per substrate concentration. A plate reader took measurements at λEx 365 nm and λEm 445 nm. The obtained data was evaluated and finally fitted to Michaelis-Menten-Kinetics with SigmaPlot™:
β-N-Acetylglucosaminidase (NagZ)
In Colisweeper, the NagZ is constitutively expressed in the mine cells. Addition of 5-Bromo-4-Chloro-3-indolyl-β-N-acetylglucosaminide (X-GluNAc) would result in a blue color precipitate, as shown in Figure Bla. However, addition of the X-GluNAc substrate that we have did not result in any color production. Control assays with p-nitrophenyl-β-N-acetylglucosaminide (pNP-GluNAc) on the other hand gave rise to yellow color, which confirms that NagZ is expressed in our mine cells.
 "
"