Team:TU-Munich/Project/Bioaccumulation
From 2013.igem.org
m (→The Problem: Algal Blooms) |
|||
| (256 intermediate revisions not shown) | |||
| Line 1: | Line 1: | ||
| - | |||
{{Team:TU-Munich/TUM13_Menu}} | {{Team:TU-Munich/TUM13_Menu}} | ||
| + | {{Team:TU-Munich/TUM13_Style}} | ||
<div id="wikicontent-container"> | <div id="wikicontent-container"> | ||
<div id="wikicontent"> | <div id="wikicontent"> | ||
| - | + | <!-- Star des Inhalts --> | |
| - | <!-- | + | |
==BioAccumulation== | ==BioAccumulation== | ||
| - | |||
| - | |||
| - | |||
| + | [[File:TUM13_General_principle_of_accumulation_of_persistent_chemicals_along_the_food_chain.jpg|thumb|right|350px|'''Figure 1:''' General principle of accumulation of persistent chemicals along the food chain.]] | ||
| + | Bioaccumulation is a difficult concept to define. The [http://toxics.usgs.gov/definitions/bioaccumulation.html U.S. Environmental Protection Agency, 2010] defines bioaccumulation as a “general term describing a process by which chemicals are taken up by an organism either directly from exposure to a contaminated medium or by consumption of food containing the chemical”. The majority of definitions is aimed at the '''build up of persistent chemicals''' such as DDT, PBC or dioxins along the food chain (see figure 1), where the pollutants accumulate in predators until they become toxic. This can lead to the local and global extinction of species and health problems for humans. As most of these persistent chemicals are not biodegradable '''a different approach is necessary'''. | ||
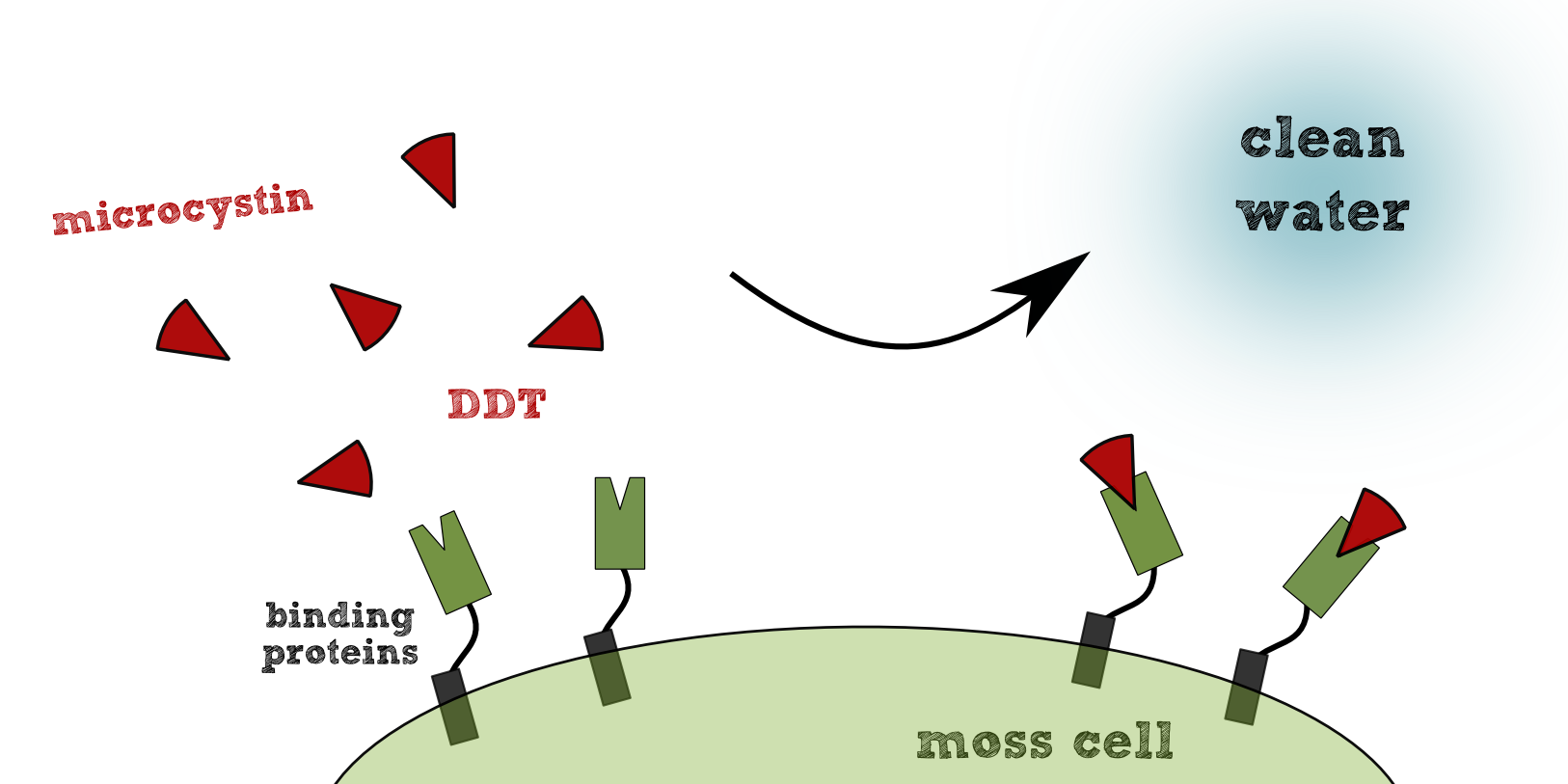
| + | [[File:TUM13 General_principle_bioaccumulation.png|thumb|left|350px|'''Figure 2:''' Principle of our bioaccumulation system.]] | ||
| + | Using our transgenic moos ([https://2013.igem.org/Team:TU-Munich/Project/Physcomitrella ''Physcomitrella patens''] ) as a chassis, we can produce effectors for specific binding of pollutants. For the sustained removal of the target pollutant the effectors must be membrane-anchored, in contrast to the cytoplasmic or secretory localization of biodegrading effectors. The membrane association ensures the fixation of the pollutant to the membrane after successful binding. Internalisation of the effectors either results in metabolism or accumulation of the pollutant in the transgenic plant cell. In case of accumulation, controlled disposal of the plants in the filter would be necessary. | ||
| + | There is a broad range of natural as well as engineered binding proteins available. Natural binding proteins act as a model and initiator in design of new artificial binding proteins combining research in different fields of biotechnology. Lipocalins, natural binding proteins, as base and scaffold for the design of anticalins confirm the popularity of bioaccumulated proteins in red biotechnology [[http://www.ncbi.nlm.nih.gov/pubmed/15676296 Schlehuber et al., 2005]]. The role of Fibrillin in the abscisic acid-mediated photoprotection shows an example of functioned bioaccumulation in a plant [[http://www.pnas.org/content/103/15/6061 Yang et al.,2007]]. The most commonly known binding proteins are antibodies which defend mammals against pathogens and toxins. Besides these natural binding proteins and anticalins there are more and more designed binding proteins such as Affibodies derived from the z-domain of the antibody-binding protein A (Ref) and DARPins that are based on an ankyrin scaffold. One research topic of the [http://www.ws.chemie.tu-muenchen.de chair of analytical chemistry of the TU Munich] is the depletion of algal toxin contaminated water by using selective biofilters based on plantibodies. Plantibodies are plant- produced and derived antibodies which can construct antibody-mediated pathogen resistance as well as change the plant phenotype by immunomodulation [[http://www.ncbi.nlm.nih.gov/pubmed/11950570 Stoger et al.,2002]]. | ||
| + | For our project we exemplarily choose three different binding proteins which each exploit different mechanisms to bind Proteins: | ||
| + | * The anticalin [[#Bioaccumulation_via_binding_proteins| <b>fluA</b>]] as an example for a binding protein. | ||
| + | * The enzyme [[#Glutathione_S-transferase| <b>Gluthathione-S-Transferase</b>]] as an example where the formation of covalent bonds between the target pollutant and another molecule (gluthatione) is catalized. | ||
| + | * The enzyme [[#Protein_Phosphotase_1_-_A_molecular_mop_for_Microcystin| <b>Protein Phosphatase 1</b>]] as example for a target of a binding protein. Here we exploit the inhibitory binding of the pollutant to the enzyme. | ||
| - | + | To sketch the theoretical binding to a membrane anchored protein in our video, we created a short animation of the process using the molecularMaya software: | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | < | + | <html> |
| + | <center><iframe style="box-shadow: 1px 1px 2px rgba(0, 0, 0, 0.2);padding: 5px;margin: 5px;background-color: white;" src="http://player.vimeo.com/video/77974681" width="450" height="255" frameborder="0" webkitAllowFullScreen mozallowfullscreen allowFullScreen></iframe></center></html> | ||
| - | == | + | == Bioaccumulation via binding proteins == |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | === Problem Example: Diclofenac=== | ||
| + | Are pharmaceuticals a blessing or a curse? Respective to the ecological consequences of diclofenac this task must be valued negative. Diclofenac which is a proven, nonsteroidal anti-imflammatory drug (NSAID) is used mostly in human medicine to treat a variety of acute and chronic pain and inflammatory conditions via inhibition of prostaglandin synthesis by inhibiting cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2)[[http://www.ncbi.nlm.nih.gov/pubmed/20470236 Gan,2010]]. Its necessity and importance in medicine research confirm e.g. the development of diclofenac patches for topical treatment of acute impact injuries [[http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1724805/ Predel et al.,2004]] and the main studies about safety and efficiency of the analgesic, anti-inflammatory, and antipyretic properties of diclofenac [[http://www.ncbi.nlm.nih.gov/pubmed/11276273 Morgan et al.,2001]][[http://www.ncbi.nlm.nih.gov/pubmed/15480981 Chan et al.,2004]][[http://www.google.de/url?sa=t&rct=j&q=&esrc=s&source=web&cd=9&ved=0CHsQFjAI&url=http%3A%2F%2Fwww.ncbi.nlm.nih.gov%2Fpubmed%2F22168216&ei=DLI5UqrPNJHSsgaL2YHAAQ&usg=AFQjCNFs7U5eWUrOKgcIuLly-afQqXpwTQ&sig2=ebhoYAq_NdG9v2nuxdB8BQ&bvm=bv.52288139,d.Yms Pavelka,2012]]. The possible resulting vicious circle (see Figure 1) shows the poisoning tragedy in 2005 which led to mass mortality and decline of vultures species especially ''Gyps bengalensis'', ''Gyps indicus'' and ''Gyps tenuirostris'' across the Indian sub-continent. Based on the consumation of diclofenac by animals, vultures have an abnormally high intake in this drug by eating the cadavers of contaminated animals. | ||
| + | === Current solutions=== | ||
| + | The fate of degradation products with their toxicity character and the development of antibiotic resistances plays a major role of the research on the effect of diclofenac in the aquatic environment. Because of the lack of analytical standards and the complex matrix of organic molecules in environmental samples, the degradation products have not yet been identified regarding molecular structure or concentration in aquatic fauna and flora. | ||
| + | The degree of the problem becomes visible in HPLC analysis, which shows in addition to the fast degradation of diclofenac over the irradiation time, a phytotoxicity increase of the increasing degradation product concentrations and a weak inhibition of cell growth. | ||
| - | + | To reduce the existence of PhACs such as diclofenac, one possibility is the usage of sewage treatment plants (STPs). Their removal efficiency depends on the sludge retention time and on the characteristics of the pharmaceutical molecule. Other strategies are the adding of an additional stage to the STP. Another approach is treated in the publications [[http://www.ncbi.nlm.nih.gov/pubmed/16190224 Westerhoff et al. (2005)]][[http://www.ncbi.nlm.nih.gov/pubmed/15984812 Huber et al., (2005)]][[http://www.ncbi.nlm.nih.gov/pubmed/12697241 Ternes et al., (2003)]] which confirm a high removal efficiency up to 95% for these additional stages. Currently developments are the nano-filtration, ozonation and power activated carbon. The importance of knowing the transformation products before applying a new technology is again demonstrated by the fact that ozonation reduces the parental substance diclofenac but cannot mineralize it. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | To sum it up, biodegradation of diclofenac introduces several new problems as '''degradation products are toxic''' and pose new harms to the environment. | ||
| + | ===Bioaccumulation instead of Biodegradation=== | ||
| + | [[File:TUM13_FluA.png|thumb|right|350px|'''Figure 3:''' Structure of fluA(blue) with its ligand fluorescein(green) [[http://www.rcsb.org/pdb/explore/explore.do?structureId=1N0S source:PBD]]]] | ||
| + | |||
| + | As biodegradation does not conclusively solve the problem of diclofenac in the aquatic environment, we propose the utilisation of antibodies for accumulation of diclofenac, which would solve the problem of toxic degradation products. | ||
| + | |||
| + | Although an antibody against diclofenac has been developed [[http://www.ncbi.nlm.nih.gov/pubmed/12966990 Deng et al. (2003)]], in vivo verification of successful binding would not be straightforward. Therefore we aimed for a first proof of principle and decided to investigate another substance that is targetable by antibodies for which verification is feasible. One attractive option is the well-known fluorophore fluorescein which was used by the [https://2008.igem.org/Team:FreiburgiGEM team Freiburg 2008]. Fluorescein fluoresces until bound by the binding protein fluA, which makes characterization tractable. The covalent binding of fluA and flourescein is shown in Figure 3. | ||
| + | |||
| + | We submitted an improved version of the Freiburg Biobrick for fluA (see Table 1) and created a transgenic moss plants ([https://2013.igem.org/Team:TU-Munich/Results/GM-Moss PF-15]) with the fluorescein binding anticalin fluA on the extracellular part of the receptor. | ||
| + | |||
| + | Producing engineered binding proteins for medical purpose in the red biotechnology is one of the main research fields in which anticalins play a major role. Lipocalins such as bilin-binding-protein BBP (from ''Pieris brassicae'') can be used for generating molecular pockets with a diversity of shapes and for creating a stable receptor protein for a ligand of choice, so that development of binding proteins against nearly chemical structures with comparable size is possible. Hence it should be possible to target a great variety of pollutants by already available binding proteins. | ||
| + | |||
| + | {|cellspacing="0" border="1" | ||
| + | |+ '''Table 1:''' Variants of the fluorescein binding Anticalin FluA | ||
| + | !Proteinvariant | ||
| + | !K<sub>D</sub> of FluA to fluorescein | ||
| + | !Literature reference | ||
| + | !BioBrick | ||
| + | |- | ||
| + | |FluA | ||
| + | | align=right | 152 nM | ||
| + | |[[http://www.ncbi.nlm.nih.gov/pubmed/10051566 Beste et al., 1999]] | ||
| + | |<partinfo>BBa_K157004</partinfo> | ||
| + | |- | ||
| + | |FluA (R95K) | ||
| + | | align=right | 64 nM | ||
| + | |[[http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005]] | ||
| + | |not available as BioBrick | ||
| + | |- | ||
| + | |FluA (R95K, A45I, S114T) | ||
| + | | align=right | 2 nM | ||
| + | |[[http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005]] | ||
| + | |<partinfo>BBa_K1159002</partinfo> | ||
| + | |} | ||
| + | |||
| + | ==Glutathione-S-transferase == | ||
| + | |||
| + | === Problem: Dichlorodiphenyltrichloroethane (DDT)=== | ||
| + | Since the entry into force of the Stockholm Convention in 2004 the insecticide Dichlordiphenyltrichlorethan (DDT) is only allowed for the abatement of disease-carrying insects such as the malaria carrying ''Anopheles dirus mosquito'' [[http://www.sciencedirect.com/science/article/pii/0965174895000909 La-Aied Prapanthadara et al.,1996]]. This measure was the consequence to the devastating consequences for the ecosystem by the widespread use of DDT, which continuously grew due to it's strong insecticidal properties. Despite the regulation in 2004, DDT still poses an environmental threat today due to its stability and capacity to accumulate in tissue [[http://www.ncbi.nlm.nih.gov/pubmed/11836138 Turusov et al.,2002]]. | ||
| + | |||
| + | === Current Solution === | ||
| + | The simultaneous degradation of mixed insecticides like DDT can be achieved by mixed insecticide enriched isolated cultures in media and soil such as the mixed fungal population (white rot fungus ''Phanerochaete chrysosporium''). The degradation efficiency of DDT can be enhanced by using carbon (mannitol) and nitrogen (sodium nitrate) in the liquid media. Based on the disadvantages which bring along the diverse strategies like chemical treatment, incineration, and landfills the development of the so called soil microflora method leads to a positive effect concerning to the detoxification of pesticides. The poor fungal growth in the media and concomitant biodegradation of insecticides is caused by toxicity exerted by high concentrations of the substrate which explains the low degradation at high concentrations. | ||
| + | |||
| + | === Caltech 2011 Solution: Covalent binding to Gluthathion via Gluthathione-S-Transferase === | ||
| + | |||
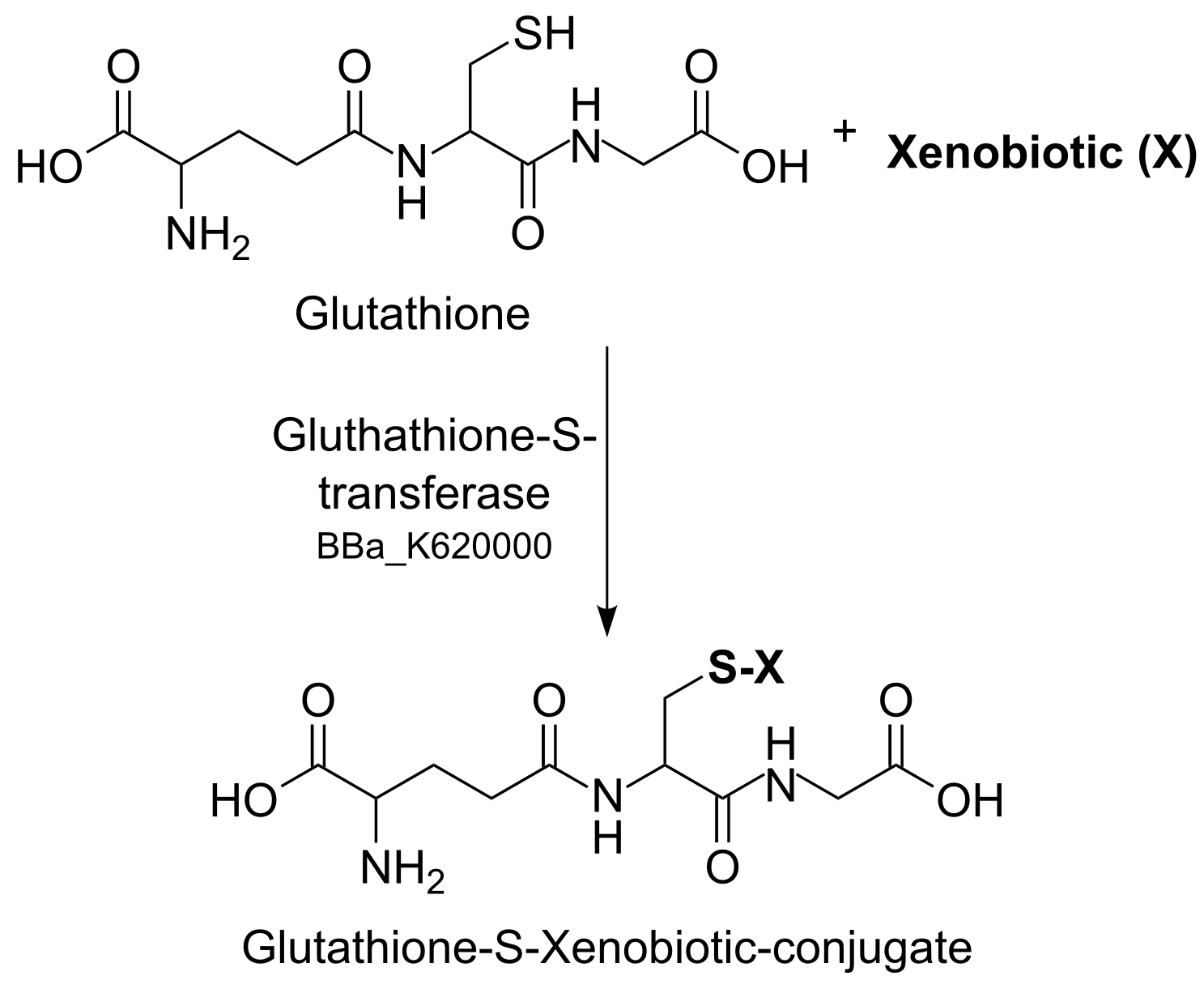
| + | [[File:TUM13_conjugation_of_GSH_and_a_xenobiotic.png|thumb|right|300px| '''Figure 4:''' Conjugation of GSH and a xenobiotic]] | ||
| + | In context to prove the bioaccumulation and to develop an effective methode of removing DDT, we envision the usage of the cytoplasmatic GST 1-1, also known as DDT Dehyrochlorinase. Along with the creation of transgenic ''Physcomitrella patens'' plants ([https://2013.igem.org/Team:TU-Munich/Results/GM-Moss PF-13]) we used the [https://2013.igem.org/Team:TU-Munich/Results/BioBricks biobrick] from the [https://2011.igem.org/Team:Caltech Caltech iGEM team 2011]. This team engineered bacteria which can degrade endocrine-disrupting chemicals such as DDT, synthetic estrogen in bodies of water to less toxic forms. Glutathion S-transferases (GSTs), an eukaryotic and prokaryotic phase II metabolic isozymes-family, catalyze the conjugation of reduced form of gluthatione (GSH, nucleophil) and xenobiotics (electrophil) to gluthatione-S-Conjugate via nucleophilic attack (see Figure 4). | ||
| + | |||
| + | [[File:TUM13_equilibrium reaction of Gluthation.png|thumb|right|300px| '''Figure 5:''' Equilibrium reaction of gluthation]] | ||
| + | |||
| + | The consequence is an increased solubility of the conjugates which leads to the removal of xenobiotics in form of conjugates via vacuole enclosure. The action of the specific transporters and the steady supply of GSH in the equilibrium reaction (see Figure 5) are the limited factors of the detoxification reaction. Along with the detoxification and cell signaling function GST’s act as transport proteins, which gave GST the previous name ligandin. Table 2 shows the 3 different superfamilies with their characteristics. | ||
| + | |||
| + | [[File:TUM13 Structure3D_of_GST.gif|thumb|right|300px| '''Figure 6:''' Molecular structure of Gluthation S-Transferase]] | ||
| + | |||
| + | {|cellspacing="0" border="1" | ||
| + | |+ '''Table 2:''' Superfamilies of GST with their characteristics | ||
| + | !Superfamily of GTS | ||
| + | !Classes based upon their structure | ||
| + | !Sequence homology [%] | ||
| + | |- | ||
| + | |Cytosolic proteins | ||
| + | |alpha, beta, delta, epsilon, zeta, theta, mu, nu, pi, sigma, tau, phi, and omega | ||
| + | | align=right | >40 | ||
| + | |- | ||
| + | |Mitochondrial proteins | ||
| + | |kappa | ||
| + | | align=right | <25 | ||
| + | |- | ||
| + | |microsomal (MAPEG= membrane-associated proteins in eicosanoid and glutathione metabolism) proteins | ||
| + | |subgroups I-IV | ||
| + | | align=right | <25 | ||
| + | |} | ||
| + | |||
| + | [[File:TUM13_Reaction_of_monochlorobimane_with_GSH.png|thumb|right|300px| '''Figure 7:''' Reaction of monochlorobimane with GSH ]] | ||
| + | |||
| + | Generally the three superfamilies differ mostly in structure and sequence as only the cytosolic and the mitochondrial superfamily have a thiorexin like domain in which the glutathione binding site (G-site) is located [[http://www.ncbi.nlm.nih.gov/pubmed/21428697 Oakley A.,2011]]. The helix alpha 2 is the most variable secondary structure. Y-GST is the subgroup which activates glutathione via using tyrosine residues. S/C-GST uses serine/cysteine residues. GST binds the substrate at the hydrophobic H-site of the enzyme and GSH at the hydrophilic G-site which together form the active site of the enzyme (see Figure 6). | ||
| + | In research techniques GST is used as so called GST-tags for separation, elucidation of direct protein-protein interaction and purification of the GST-fusion protein mostly by pull-down assay. So targeting GST with molecule therapeutics represents GTS as an attractive target for drug discovery [[http://www.ncbi.nlm.nih.gov/pubmed/16550164 McIlwain et al.,2006]]. | ||
| + | |||
| + | A mammalian variant of GST, GSTP, plays a major role in cancer- development and potential drug/chemotherapeutic resistance in a majority of tumor cell lines: The inhibition of the pro-apoptotic pathway (JNK pathway) and the overexpression of GSTP in tumor cells lead to escape of apoptosis of the tumor cells mediated by non GSTP- substance-drugs [[http://www.ncbi.nlm.nih.gov/pubmed/8770536 Hayes et al.,1995]] [[http://www.ncbi.nlm.nih.gov/pubmed/20981235 Josephy,2010]] [[http://www.ncbi.nlm.nih.gov/pubmed/10971201 Hayes et al.,2000]] [[http://www.ncbi.nlm.nih.gov/pubmed/12563680 Fraser et al.,2003]]. | ||
| + | To avoid the time- and labor-intensive method HPLC following derivatization with 2-nitrobenzoic acid we used the common sensitive technique with monochlorobimane to measure GSH as a proof of principle. The adding of monochlorobimane to the culture medium leads to the conjugation of GSH to monochlorobimane catalyzed by DDT Dehyrochlorinase (see Figure 7). The GSH-monochlorobimane conjugate can be measured fluorometrically [[http://www.ncbi.nlm.nih.gov/pubmed/11038270 Kamencic et al.,2000]]. | ||
| + | |||
| + | ==Protein Phosphotase 1 – A molecular mop for Microcystin== | ||
| + | |||
| + | === The Problem: Algal Blooms === | ||
| + | [[File:TUM13_Johannas_Stuff.png|thumb|center|900px| '''Figure 8:''' Eutrophication]] | ||
| + | Which phenomenon implicates the common name “Florida red tide”? This term is used in annually along Florida waters where species known as ''Karenia brevis'' leads to a red colored alga bloom along the Florida coast. Algal blooms are a worldwide environmental problem! This becomes evident with the appointment of 10.000 people for cleaning up the algal bloom in Beijing for the Olympic discipline “sailing regatta” in 2008. Depending on the cause, bacteria algal can bloom at concentrations of hundreds to thousands of cells per milliliter. An officially recognized threshold level isn’t public. Possible diverse factors which are the reason for the high affecting of bloom formation are listed followed: | ||
| + | *Light intensity: cyanobacteria which form surface blooms have a higher tolerance concerning high light intensities. This leads to lower growth rate than other phytoplankton organisms. | ||
| + | *Gas vacuoles: Gas vesicles give the cyanobacteria a lower density than water. | ||
| + | *Growth rate: Slow growth rates require long water retention times to enable a bloom. | ||
| + | *Phosphorous and nitrogen: A low ratio of these substance may favour a development of cyanobacteria blooms. | ||
| + | *Temperature: maximum growth rates are attained by the most cyanobacteria above 25°C. Thus much bloom formation is during the summer. | ||
| + | A possible cascade which is caused by the release of nutrients in the aquatic ecosystems is shown in Figure 8. Another result confirms the spread and the persistence of cholera through algal blooms [[http://www.ncbi.nlm.nih.gov/pubmed/8155853 Epstein PR.,1993]]. Currently the so called harmful algal blooms are an additive for the detection of bacteria spread and their caused health effects to fauna and flora [[http://www.ncbi.nlm.nih.gov/pubmed/21135906 Ferrante et al.,2010]] [[http://www.ncbi.nlm.nih.gov/pubmed/17298369 Barlaan EA et al.,2007]]. The reduction of the uptake of toxic methylmercury in freshwater food webs by algal blooms confirms a positive reverse of algal blooms [[http://www.ncbi.nlm.nih.gov/pubmed/11904388 Pickhardt PC. et al.,2002]]. | ||
| + | |||
| + | === Current Solution to combat Algal blooms === | ||
| + | It is well known that cyanobacteria produce most Toxins such as microcystin under species-specific conditions which are most favorable for their growth. As a result most studies concerning this topic try to tackle the problem from this point of view. For the investigated species, they found out that the toxin production increased under light optimum condition of the relative bacteria species and on low and high pH. The usage of zinc was necessary for the increase of growth and toxin production. Dependent on nitrogen fixing respectively non-nitrogen fixing species the nitrogen and phosphorous rate in media was unnecessary respectively necessary concerning to the toxin production. Evidences of the role of plasmid, multi-enzyme complexes and peptide synthetase gene involvement in toxin production were also confirmed: e.g. knockout experiments showed that peptide synthetase genes are responsible for microcysteine production. | ||
| + | |||
| + | Major problems are still the lack of knowledge regarding to occurrence and toxicity of cyanobacterial products like the diversity of LPS structures which cause allergic as well as toxic human health problems, the extremely weak scientific basis and poorly developed procedures for valuation on health effects. The TDI (tolerable daily intake) is one of the key features on which the toxicology studies are aimed. Based on analytic and sampling problems or inadequate scientific data safe practice guidelines can be assisted for the reducing of exposure. | ||
| + | |||
| + | The human exposure to the toxins is a major point to combat the human health risk. The WHO guideline values base upon a number of assumptions which change locally or nationally according to the present situation. To fill the lack of knowledge of parental variants the HPLC method is commonly used also for the determination of references. Current treatment processes used at water surface plants are coagulation, sand filtration, and clarification. | ||
| + | |||
| + | The negative site of these methods is the ineffective removal and destruction of cyanotoxins. To combat this problem treatment systems like carbon- and membrane filtration were developed. External and internal accumulation of toxins is another research topic since the inhibition of Mustard seedling development by of microcystein-LR in aquatic root solution. | ||
| + | |||
| + | === Dundee 2013 Solution to combat Algal blooms=== | ||
| + | |||
| + | [[File:TUM13_PP1.png|thumb|right|350px| '''Figure 9:''' interaction of PP1 and microcystin [[http://www.rcsb.org/pdb/explore/explore.do?structureId=2BDX source: PDB]]]] | ||
| + | |||
| + | Due to the deleterious results from the toxin release of algal blooms many risk assesments were have been developed [[http://www.ncbi.nlm.nih.gov/pubmed/10499991 Schaeffer et al., 1999]]. The effects of microcystin for the aquatic and terrestrial ecosystem have been proven to include the modulation of proliferation, apoptosis and proliferation of spermatogenic cells in vivo [[http://www.ncbi.nlm.nih.gov/pubmed/24025782 Zhou et al.,2013]], which makes it an attractive target for our project. | ||
| + | |||
| + | For the bioaccumulation of microcystin, it is possible to use the enzyme PP1. Mycrocystin can inhibit the function of PP1 by binding to it, this interaction of microcystin, in form of a heptapeptide structure, with three distinct regions of the PP1c is depicted in Figure 9. Hence PP1 can act as natural binding partner for mycrocystin. | ||
| + | |||
| + | As the Dundee iGEM team 2013 [https://2013.igem.org/Team:Dundee Team Dundee] was already working on this subject we contacted them and they kindly provided us with the gene that encodes PP1. After converting the human PP1, to <nowiki>RFC</nowiki> 25 and constructing some expression plasmids we produced transgenic ''phytomitrella patens'' ([https://2013.igem.org/Team:TU-Munich/Results/GM-Moss PF-14]) with PP1 and a biobrick of PP1 receptor. | ||
==References:== | ==References:== | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
<!-- Ab hier richtige Referenzen einfügen --> | <!-- Ab hier richtige Referenzen einfügen --> | ||
| - | |||
| - | |||
| + | [[http://toxics.usgs.gov/definitions/bioaccumulation.html Definition of Bioaccumulation, U.S. Department of the Interior | U.S. Geological Survey,2013]] Definiton of Bioaccumulation | ||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/15676296 Schlehuber and Skerra,2005]] Schlehuber S, Skerra A.(2005)Lipocalins in drug discovery: from natural ligand-binding proteins to "anticalins".''Drug Discov Today''10(1):23-33 | ||
| + | [[http://www.pnas.org/content/103/15/6061 Yang et al.,2006]] Yang Y.,Sulpice R.,Himmelbach A.,Meinhard M.,Christmann A., Grill E. Fibrillin expression is regulated by abscisic acid response regulators and is involved in abscisic acid-mediated photoprotection ''PNAS'' 6061–6066 | ||
| - | + | [[http://www.ws.chemie.tu-muenchen.de/ Institute of Hydrochemistry and Chair for Analytical Chemistry ]] | |
| - | + | [[http://www.ncbi.nlm.nih.gov/pubmed/11950570 Stoger et al.,2002]] Stoger E, Sack M, Fischer R, Christou P.(2002) Plantibodies: applications, advantages and bottlenecks ''Curr Opin Biotechnol.'' 13(2):161-6. | |
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/20470236 Gan, 2010]] Gan TJ.(2010) Diclofenac: an update on its mechanism of action and safety profile ''Curr Med Res Opin''26(7):1715-31. doi: 10.1185/03007995.2010.486301 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1724805/ Predel et al.,2004]] H Predel, R Koll, H Pabst, R Dieter, G Gallacchi, B Giannetti, M Bulitta, J Heidecker, and E Mueller(2004)Diclofenac patch for topical treatment of acute impact injuries: a randomised, double blind, placebo controlled, multicentre study ''Br J Sports Med'' 38(3): 318–323. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/11276273 Moragan et al.,2001]] Morgan GJ Jr, Kaine J, DeLapp R, Palmer R (2001)Treatment of elderly patients with nabumetone or diclofenac: gastrointestinal safety profile ''J Clin Gastroenterol''32(4):310-4. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/15480981 Chan et al.,2004]] Chan FK, Hung LC, Suen BY, Wong VW, Hui AJ, Wu JC, Leung WK, Lee YT, To KF, Chung SC, Sung JJ.(2004) Celecoxib versus diclofenac plus omeprazole in high-risk arthritis patients: results of a randomized double-blind trial ''Gastroenterology'' 127(4):1038-43. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/22168216 Pavelka,2012]] Pavelka K.(2012) A comparison of the therapeutic efficacy of diclofenac in osteoarthritis: a systematic review of randomised controlled trials ''Curr Med Res Opin.''28(1):163-78. doi: 10.1185/03007995.2011.649848. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/16190224 Westerhoff et al. (2005)]] Westerhoff, P., Yoon, Y., Snyder, S., and Wert, E. (2005). Fate of endocrine- disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. ''Environ Sci Technol'', 39(17):6649–63. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/15984812 Huber et al., (2005)]] Huber, M. M., G ̈obel, A., Joss, A., Hermann, N., L ̈offler, D., McArdell, C. S., Ried, A., Siegrist, H., Ternes, T. A., and von Gunten, U. (2005). Oxidation of pharmaceuticals during ozonation of municipal wastewater effluents: a pilot study. ''Environ Sci Technol'', 39(11):4290–9. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/12697241 Ternes et al., (2003)]] Ternes, T. A., Stu ̈ber, J., Herrmann, N., McDowell, D., Ried, A., Kampmann, M., and Teiser, B. (2003). Ozonation: a tool for removal of pharmaceuticals, contrast media and musk fragrances from wastewater? ''Water Res'', 37(8):1976–82. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/12966990 Deng et al. (2003)]] Deng, A., Himmelsbach, M., Zhu, Q.-Z., Frey, S., Sengl, M., Buchberger, W., Niessner, R., and Knopp, D. (2003). Residue analysis of the pharmaceutical diclofenac in different water types using elisa and gc-ms. ''Environ Sci Technol'', 37(15):3422–9. | ||
| + | |||
| + | [[https://2008.igem.org/Team:Freiburg Homepage iGEM team Freiburg 2008]] | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/10051566 Beste et al., 1999]] Beste G, Schmidt FS, Stibora T, Skerra A. (1999) Small antibody-like proteins with prescribed ligand specificities derived from the lipocalin fold. ''PNAS'', 96(5):1898-903. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005]] Vopel S, Mühlbach H, Skerra A. (2005) Rational engineering of a fluorescein-binding anticalin for improved ligand affinity. ''Biol. Chem.'', 386(11):1097-104. | ||
| + | |||
| + | [[http://www.sciencedirect.com/science/article/pii/0965174895000909 La-Aied Prapanthadara et al.,1996]] La-Aied Prapanthadara,Surangchit Koottathe,Nongkran Promtet,Janet Hemingway†,Albert J., Ketterman (1996) Purification and characterization of a major glutathione S-transferase from the mosquito Anopheles dirus (Species B) ''Insect Biochemistry and Molecular Biology''Volume 26, Issue 3 Pages 277–285 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/11836138 Turusov et al.,2002 ]] Turusov V, Rakitsky V, Tomatis L.(2002)Dichlorodiphenyltrichloroethane (DDT): ubiquity, persistence, and risks''Environ Health Perspect.''110(2):125-8 | ||
| + | |||
| + | [[https://2011.igem.org/Team:Caltech Homepage iGEM team Caltech 2011]] | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/21428697 Oakley,2006]] Oakley A.(2011)''Drug Metab Rev''43(2):138-51. doi: 10.3109/03602532.2011.558093 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/16550164 McIlwain et al.,2006]] McIlwain CC, Townsend DM, Tew KD(2006)Glutathione S-transferase polymorphisms: cancer incidence and therapy ''Oncogene''25(11):1639-48 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/8770536 Hayes et al.,1995]]Hayes JD, Pulford DJ.(1995) The glutathione S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance.''Crit Rev Biochem Mol Biol.''30(6):445-600 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/20981235 Josephy, 2010]] Josephy PD.(2010)Genetic variations in human glutathione transferase enzymes: significance for pharmacology and toxicology.''Hum Genomics Proteomics'' 2010:876940. doi: 10.4061/2010/876940 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/10971201 Hayes et al.,2000]]Hayes JD, Strange RC.(2000)Glutathione S-transferase polymorphisms and their biological consequences''Pharmacology'' 61(3):154-66 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/12563680 Fraser et al., 2003]]Fraser PA, Ding WZ, Mohseni M, Treadwell EL, Dooley MA, St Clair EW, Gilkeson GS, Cooper GS. (2003),Glutathione S-transferase M null homozygosity and risk of systemic lupus erythematosus associated with sun exposure: a possible gene-environment interaction for autoimmunity''J Rheumatol''30(2):276-82 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/11038270 Kamencic et al.,2000]] Kamencic H, Lyon A, Paterson PG, Juurlink BH.(2000)Monochlorobimane fluorometric method to measure tissue glutathione.''Anal Biochem'' 286(1):35-7 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/8155853 Epstein PR.,1993]] Epstein PR.(1993),Algal blooms in the spread and persistence of Cholera ''Biosystems'' 31(2-3):209-21 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/21135906 Ferrante M. et al., 2010]] Ferrante M, Ledda C, Cunsolo MA, Fiore M, Fallico R, Sciacca S, Oliveri Conti GM (2010) Harmful algal blooms in Italy and their health effects in the Population ''Ig Sanita Pubbl.'' 66(5):649-58 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/17298369 Barlaan EA et al.,2007]] Barlaan EA, Furukawa S, Takeuchi K.(2007) Detection of bacteria associated with harmful algal blooms from coastal and microcosm environments using electronic microarrays ''Environ Microbiol''9(3):690-702. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/11904388 Pickhardt PC et al.,2002 ]] Pickhardt PC, Folt CL, Chen CY, Klaue B, Blum JD.(2002) Algal blooms reduce the uptake of toxic methylmercury in freshwater food webs ''Proc Natl Acad Sci U S A.''99(7):4419-23. Epub 2002 Mar 19. | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/10499991 Schaeffer et al.,1995]] Schaeffer DJ, Malpas PB, Barton LL.(1995),Risk assessment of microcystin in dietary Aphanizomenon flos-aquae,Ecotoxicol Environ Saf.,44(1):73-80 | ||
| + | |||
| + | [[http://www.ncbi.nlm.nih.gov/pubmed/24025782 Zhou et al.,2013]] Zhou Y, Chen Y, Yuan M, Xiang Z, Han X.(2013)In vivo study on the effects of microcystin-LR on the apoptosis, proliferation and differentiation of rat testicular spermatogenic cells of male rats injected i.p. with Toxins ''J Toxicol Sci.''38(5):661-70 | ||
| + | |||
| + | [[https://2013.igem.org/Team:Dundee Homepage iGEM team Dundee 2013]] | ||
| + | |||
| + | [[http://www.birdlife.org.uk/action/science/species/asia_vulture_crisis/index.html "Asia vulture crisis",Birdlife international]] | ||
| + | |||
| + | [[http://www.bfarm.de/SharedDocs/1_Downloads/EN/vigilance/csp/c-d/diclofenac.pdf;jsessionid=7EF043741A9EE95F1EEF8C14A116BBD7.1_cid332?__blob=publicationFile Core safty profil Diclofenac (2011)]] | ||
| + | |||
| + | [[http://www.nabu.de/tiereundpflanzen/voegel/international/03530.html "Katastrophales Geiersterben in Indien"(2005),''NABU'']] | ||
| + | |||
| + | [[http://www.spiegel.de/wissenschaft/natur/insektengift-ddt-wie-die-malaria-wunderwaffe-bauern-in-die-armut-treibt-a-768654.html Koch L.,2011]]Koch L.(2011)Insektengift DDT: Wie die Malaria-Wunderwaffe Bauern in die Armut treibt ''Spiegel online'' | ||
| + | |||
| + | Chorus I.,Bartram J.(1999),Toxic Cyanobacteria in Water:A guide to their public health consequences,monitoring and Management,E & FN Spon, an imprint of Routledge 11 New Fetter Lane, London EC4P 4EE | ||
| + | |||
| + | Jiskra,M.,Fate of the pharmaceutical diclofenac in the aquatic Environment,Term paper 21. In Biogeochemistry and Pollutant Dynamics | ||
| + | |||
| + | Kulshrestha,Kumari,Simultaneous Degradation of Mixed Insecticides by Mixed Fungal Culture Isolated from Sewage Sludge(2010),J. Agric. Food Chem., 58, 11852–11856 DOI:10.1021/jf1028202 | ||
| + | |||
| + | Tomlinson,T.G.,Control by D.D.T of Flies Breeding in Percolating Sewage Filters(1945),Water pollution research laboratory,Minworth Birmingham,Nature | ||
| + | |||
| + | <!-- Ende des Inhalts --> | ||
</div> | </div> | ||
</div> | </div> | ||
| - | {{Team:TU-Munich/ | + | {{Team:TU-Munich/TUM13_Footer}} |
| - | + | ||
Latest revision as of 03:47, 29 October 2013
BioAccumulation
Bioaccumulation is a difficult concept to define. The [http://toxics.usgs.gov/definitions/bioaccumulation.html U.S. Environmental Protection Agency, 2010] defines bioaccumulation as a “general term describing a process by which chemicals are taken up by an organism either directly from exposure to a contaminated medium or by consumption of food containing the chemical”. The majority of definitions is aimed at the build up of persistent chemicals such as DDT, PBC or dioxins along the food chain (see figure 1), where the pollutants accumulate in predators until they become toxic. This can lead to the local and global extinction of species and health problems for humans. As most of these persistent chemicals are not biodegradable a different approach is necessary.
Using our transgenic moos (Physcomitrella patens ) as a chassis, we can produce effectors for specific binding of pollutants. For the sustained removal of the target pollutant the effectors must be membrane-anchored, in contrast to the cytoplasmic or secretory localization of biodegrading effectors. The membrane association ensures the fixation of the pollutant to the membrane after successful binding. Internalisation of the effectors either results in metabolism or accumulation of the pollutant in the transgenic plant cell. In case of accumulation, controlled disposal of the plants in the filter would be necessary.
There is a broad range of natural as well as engineered binding proteins available. Natural binding proteins act as a model and initiator in design of new artificial binding proteins combining research in different fields of biotechnology. Lipocalins, natural binding proteins, as base and scaffold for the design of anticalins confirm the popularity of bioaccumulated proteins in red biotechnology http://www.ncbi.nlm.nih.gov/pubmed/15676296 Schlehuber et al., 2005. The role of Fibrillin in the abscisic acid-mediated photoprotection shows an example of functioned bioaccumulation in a plant http://www.pnas.org/content/103/15/6061 Yang et al.,2007. The most commonly known binding proteins are antibodies which defend mammals against pathogens and toxins. Besides these natural binding proteins and anticalins there are more and more designed binding proteins such as Affibodies derived from the z-domain of the antibody-binding protein A (Ref) and DARPins that are based on an ankyrin scaffold. One research topic of the [http://www.ws.chemie.tu-muenchen.de chair of analytical chemistry of the TU Munich] is the depletion of algal toxin contaminated water by using selective biofilters based on plantibodies. Plantibodies are plant- produced and derived antibodies which can construct antibody-mediated pathogen resistance as well as change the plant phenotype by immunomodulation http://www.ncbi.nlm.nih.gov/pubmed/11950570 Stoger et al.,2002.
For our project we exemplarily choose three different binding proteins which each exploit different mechanisms to bind Proteins:
- The anticalin fluA as an example for a binding protein.
- The enzyme Gluthathione-S-Transferase as an example where the formation of covalent bonds between the target pollutant and another molecule (gluthatione) is catalized.
- The enzyme Protein Phosphatase 1 as example for a target of a binding protein. Here we exploit the inhibitory binding of the pollutant to the enzyme.
To sketch the theoretical binding to a membrane anchored protein in our video, we created a short animation of the process using the molecularMaya software:
Bioaccumulation via binding proteins
Problem Example: Diclofenac
Are pharmaceuticals a blessing or a curse? Respective to the ecological consequences of diclofenac this task must be valued negative. Diclofenac which is a proven, nonsteroidal anti-imflammatory drug (NSAID) is used mostly in human medicine to treat a variety of acute and chronic pain and inflammatory conditions via inhibition of prostaglandin synthesis by inhibiting cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2)http://www.ncbi.nlm.nih.gov/pubmed/20470236 Gan,2010. Its necessity and importance in medicine research confirm e.g. the development of diclofenac patches for topical treatment of acute impact injuries http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1724805/ Predel et al.,2004 and the main studies about safety and efficiency of the analgesic, anti-inflammatory, and antipyretic properties of diclofenac http://www.ncbi.nlm.nih.gov/pubmed/11276273 Morgan et al.,2001http://www.ncbi.nlm.nih.gov/pubmed/15480981 Chan et al.,2004http://www.google.de/url?sa=t&rct=j&q=&esrc=s&source=web&cd=9&ved=0CHsQFjAI&url=http://www.ncbi.nlm.nih.gov/pubmed/22168216&ei=DLI5UqrPNJHSsgaL2YHAAQ&usg=AFQjCNFs7U5eWUrOKgcIuLly-afQqXpwTQ&sig2=ebhoYAq_NdG9v2nuxdB8BQ&bvm=bv.52288139,d.Yms Pavelka,2012. The possible resulting vicious circle (see Figure 1) shows the poisoning tragedy in 2005 which led to mass mortality and decline of vultures species especially Gyps bengalensis, Gyps indicus and Gyps tenuirostris across the Indian sub-continent. Based on the consumation of diclofenac by animals, vultures have an abnormally high intake in this drug by eating the cadavers of contaminated animals.
Current solutions
The fate of degradation products with their toxicity character and the development of antibiotic resistances plays a major role of the research on the effect of diclofenac in the aquatic environment. Because of the lack of analytical standards and the complex matrix of organic molecules in environmental samples, the degradation products have not yet been identified regarding molecular structure or concentration in aquatic fauna and flora.
The degree of the problem becomes visible in HPLC analysis, which shows in addition to the fast degradation of diclofenac over the irradiation time, a phytotoxicity increase of the increasing degradation product concentrations and a weak inhibition of cell growth.
To reduce the existence of PhACs such as diclofenac, one possibility is the usage of sewage treatment plants (STPs). Their removal efficiency depends on the sludge retention time and on the characteristics of the pharmaceutical molecule. Other strategies are the adding of an additional stage to the STP. Another approach is treated in the publications http://www.ncbi.nlm.nih.gov/pubmed/16190224 Westerhoff et al. (2005)http://www.ncbi.nlm.nih.gov/pubmed/15984812 Huber et al., (2005)http://www.ncbi.nlm.nih.gov/pubmed/12697241 Ternes et al., (2003) which confirm a high removal efficiency up to 95% for these additional stages. Currently developments are the nano-filtration, ozonation and power activated carbon. The importance of knowing the transformation products before applying a new technology is again demonstrated by the fact that ozonation reduces the parental substance diclofenac but cannot mineralize it.
To sum it up, biodegradation of diclofenac introduces several new problems as degradation products are toxic and pose new harms to the environment.
Bioaccumulation instead of Biodegradation
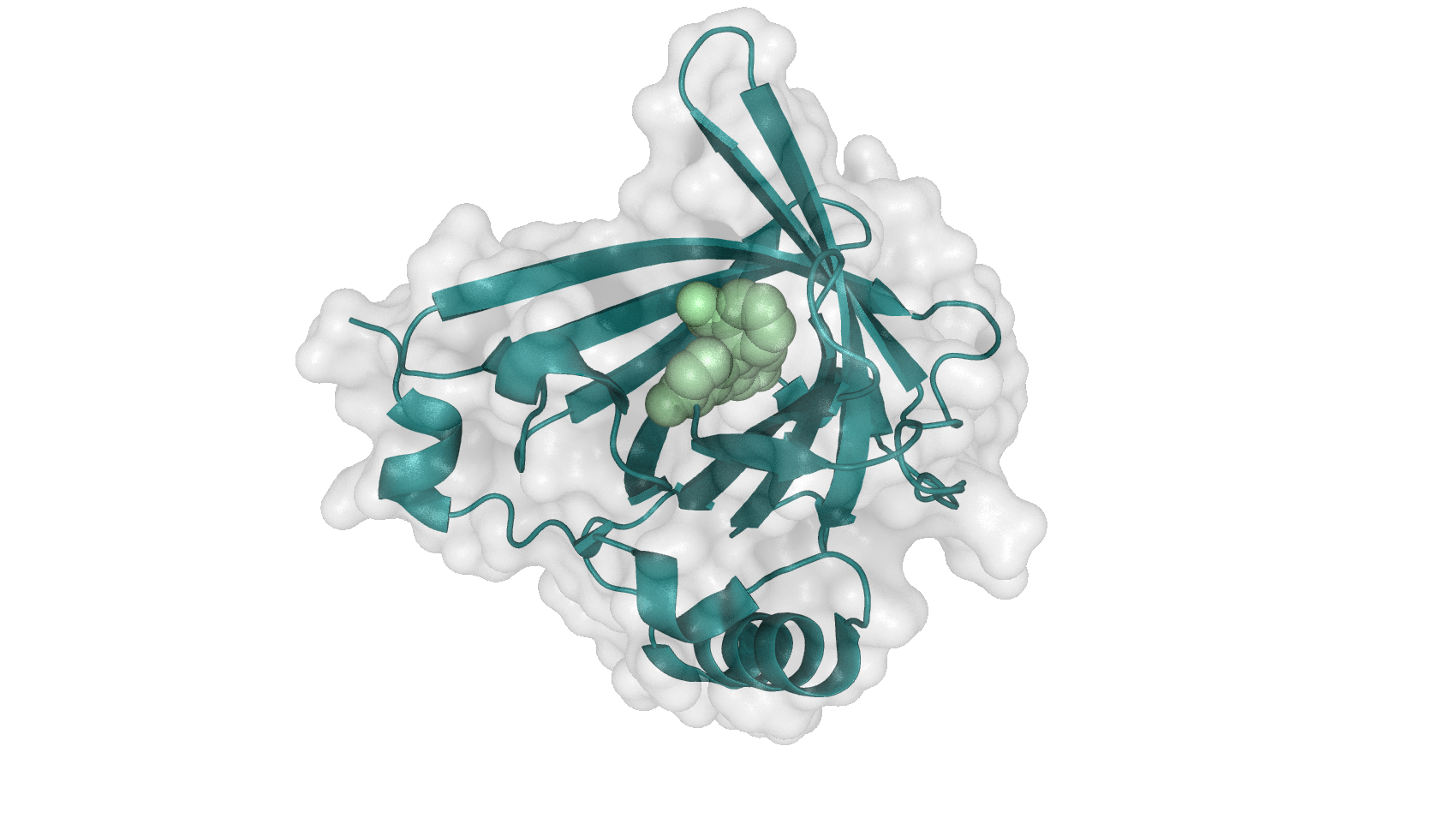
As biodegradation does not conclusively solve the problem of diclofenac in the aquatic environment, we propose the utilisation of antibodies for accumulation of diclofenac, which would solve the problem of toxic degradation products.
Although an antibody against diclofenac has been developed http://www.ncbi.nlm.nih.gov/pubmed/12966990 Deng et al. (2003), in vivo verification of successful binding would not be straightforward. Therefore we aimed for a first proof of principle and decided to investigate another substance that is targetable by antibodies for which verification is feasible. One attractive option is the well-known fluorophore fluorescein which was used by the team Freiburg 2008. Fluorescein fluoresces until bound by the binding protein fluA, which makes characterization tractable. The covalent binding of fluA and flourescein is shown in Figure 3.
We submitted an improved version of the Freiburg Biobrick for fluA (see Table 1) and created a transgenic moss plants (PF-15) with the fluorescein binding anticalin fluA on the extracellular part of the receptor.
Producing engineered binding proteins for medical purpose in the red biotechnology is one of the main research fields in which anticalins play a major role. Lipocalins such as bilin-binding-protein BBP (from Pieris brassicae) can be used for generating molecular pockets with a diversity of shapes and for creating a stable receptor protein for a ligand of choice, so that development of binding proteins against nearly chemical structures with comparable size is possible. Hence it should be possible to target a great variety of pollutants by already available binding proteins.
| Proteinvariant | KD of FluA to fluorescein | Literature reference | BioBrick |
|---|---|---|---|
| FluA | 152 nM | http://www.ncbi.nlm.nih.gov/pubmed/10051566 Beste et al., 1999 | <partinfo>BBa_K157004</partinfo> |
| FluA (R95K) | 64 nM | http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005 | not available as BioBrick |
| FluA (R95K, A45I, S114T) | 2 nM | http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005 | <partinfo>BBa_K1159002</partinfo> |
Glutathione-S-transferase
Problem: Dichlorodiphenyltrichloroethane (DDT)
Since the entry into force of the Stockholm Convention in 2004 the insecticide Dichlordiphenyltrichlorethan (DDT) is only allowed for the abatement of disease-carrying insects such as the malaria carrying Anopheles dirus mosquito http://www.sciencedirect.com/science/article/pii/0965174895000909 La-Aied Prapanthadara et al.,1996. This measure was the consequence to the devastating consequences for the ecosystem by the widespread use of DDT, which continuously grew due to it's strong insecticidal properties. Despite the regulation in 2004, DDT still poses an environmental threat today due to its stability and capacity to accumulate in tissue http://www.ncbi.nlm.nih.gov/pubmed/11836138 Turusov et al.,2002.
Current Solution
The simultaneous degradation of mixed insecticides like DDT can be achieved by mixed insecticide enriched isolated cultures in media and soil such as the mixed fungal population (white rot fungus Phanerochaete chrysosporium). The degradation efficiency of DDT can be enhanced by using carbon (mannitol) and nitrogen (sodium nitrate) in the liquid media. Based on the disadvantages which bring along the diverse strategies like chemical treatment, incineration, and landfills the development of the so called soil microflora method leads to a positive effect concerning to the detoxification of pesticides. The poor fungal growth in the media and concomitant biodegradation of insecticides is caused by toxicity exerted by high concentrations of the substrate which explains the low degradation at high concentrations.
Caltech 2011 Solution: Covalent binding to Gluthathion via Gluthathione-S-Transferase
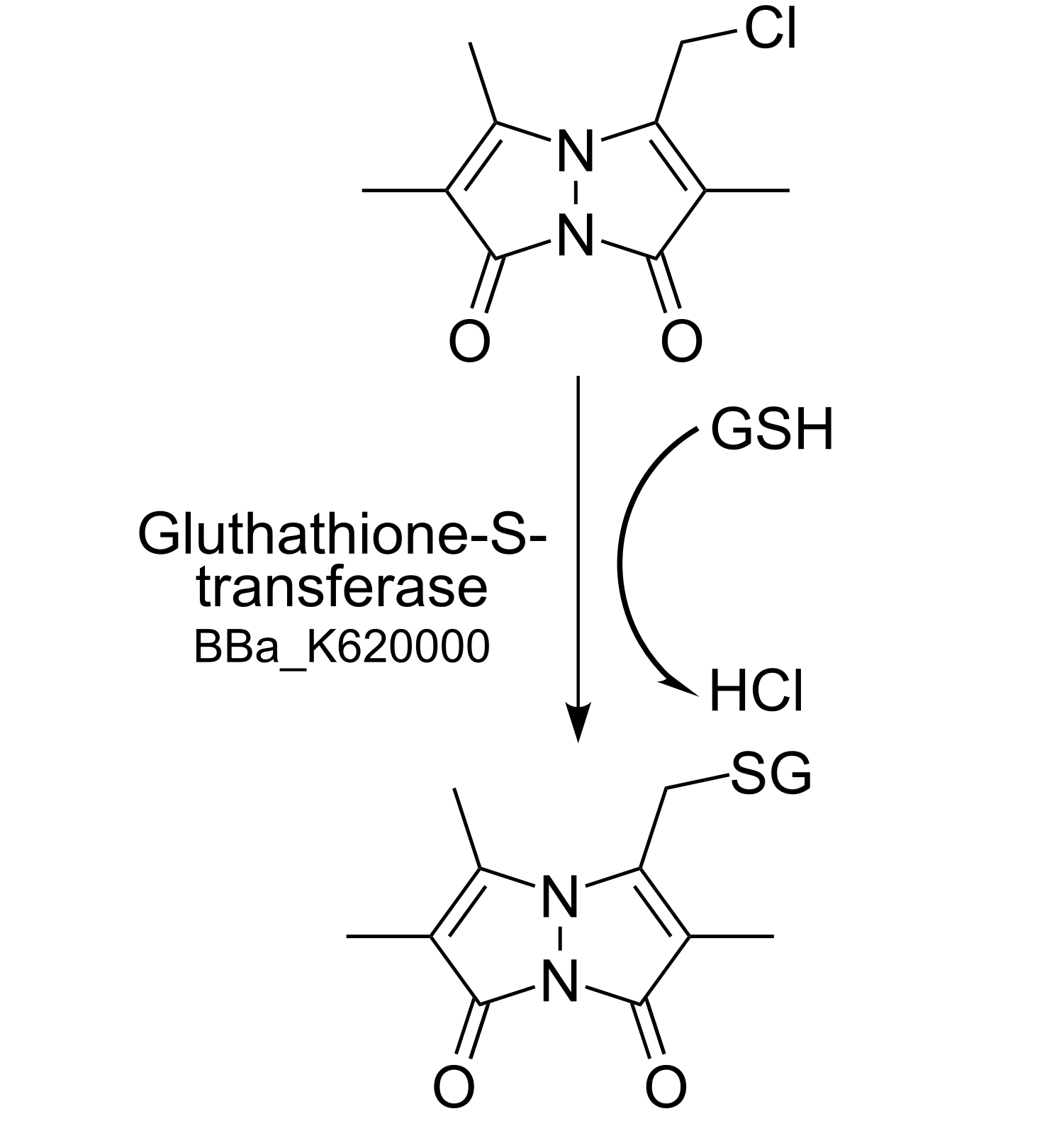
In context to prove the bioaccumulation and to develop an effective methode of removing DDT, we envision the usage of the cytoplasmatic GST 1-1, also known as DDT Dehyrochlorinase. Along with the creation of transgenic Physcomitrella patens plants (PF-13) we used the biobrick from the Caltech iGEM team 2011. This team engineered bacteria which can degrade endocrine-disrupting chemicals such as DDT, synthetic estrogen in bodies of water to less toxic forms. Glutathion S-transferases (GSTs), an eukaryotic and prokaryotic phase II metabolic isozymes-family, catalyze the conjugation of reduced form of gluthatione (GSH, nucleophil) and xenobiotics (electrophil) to gluthatione-S-Conjugate via nucleophilic attack (see Figure 4).
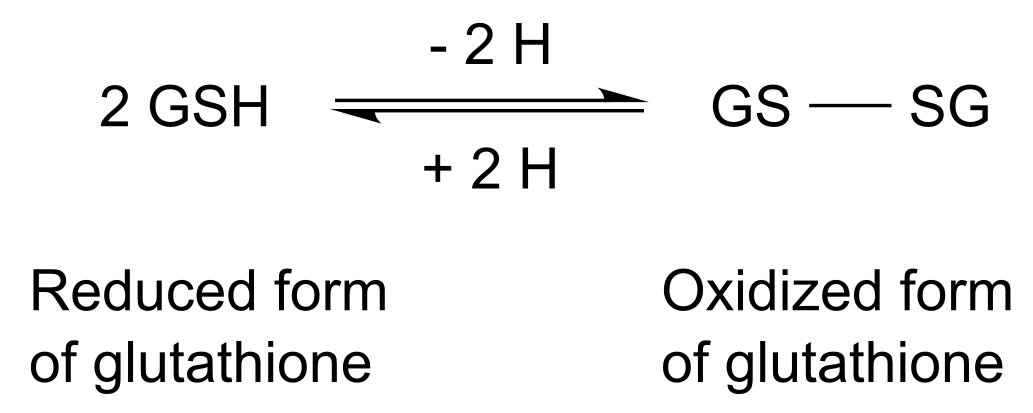
The consequence is an increased solubility of the conjugates which leads to the removal of xenobiotics in form of conjugates via vacuole enclosure. The action of the specific transporters and the steady supply of GSH in the equilibrium reaction (see Figure 5) are the limited factors of the detoxification reaction. Along with the detoxification and cell signaling function GST’s act as transport proteins, which gave GST the previous name ligandin. Table 2 shows the 3 different superfamilies with their characteristics.
| Superfamily of GTS | Classes based upon their structure | Sequence homology [%] |
|---|---|---|
| Cytosolic proteins | alpha, beta, delta, epsilon, zeta, theta, mu, nu, pi, sigma, tau, phi, and omega | >40 |
| Mitochondrial proteins | kappa | <25 |
| microsomal (MAPEG= membrane-associated proteins in eicosanoid and glutathione metabolism) proteins | subgroups I-IV | <25 |
Generally the three superfamilies differ mostly in structure and sequence as only the cytosolic and the mitochondrial superfamily have a thiorexin like domain in which the glutathione binding site (G-site) is located http://www.ncbi.nlm.nih.gov/pubmed/21428697 Oakley A.,2011. The helix alpha 2 is the most variable secondary structure. Y-GST is the subgroup which activates glutathione via using tyrosine residues. S/C-GST uses serine/cysteine residues. GST binds the substrate at the hydrophobic H-site of the enzyme and GSH at the hydrophilic G-site which together form the active site of the enzyme (see Figure 6). In research techniques GST is used as so called GST-tags for separation, elucidation of direct protein-protein interaction and purification of the GST-fusion protein mostly by pull-down assay. So targeting GST with molecule therapeutics represents GTS as an attractive target for drug discovery http://www.ncbi.nlm.nih.gov/pubmed/16550164 McIlwain et al.,2006.
A mammalian variant of GST, GSTP, plays a major role in cancer- development and potential drug/chemotherapeutic resistance in a majority of tumor cell lines: The inhibition of the pro-apoptotic pathway (JNK pathway) and the overexpression of GSTP in tumor cells lead to escape of apoptosis of the tumor cells mediated by non GSTP- substance-drugs http://www.ncbi.nlm.nih.gov/pubmed/8770536 Hayes et al.,1995 http://www.ncbi.nlm.nih.gov/pubmed/20981235 Josephy,2010 http://www.ncbi.nlm.nih.gov/pubmed/10971201 Hayes et al.,2000 http://www.ncbi.nlm.nih.gov/pubmed/12563680 Fraser et al.,2003. To avoid the time- and labor-intensive method HPLC following derivatization with 2-nitrobenzoic acid we used the common sensitive technique with monochlorobimane to measure GSH as a proof of principle. The adding of monochlorobimane to the culture medium leads to the conjugation of GSH to monochlorobimane catalyzed by DDT Dehyrochlorinase (see Figure 7). The GSH-monochlorobimane conjugate can be measured fluorometrically http://www.ncbi.nlm.nih.gov/pubmed/11038270 Kamencic et al.,2000.
Protein Phosphotase 1 – A molecular mop for Microcystin
The Problem: Algal Blooms
Which phenomenon implicates the common name “Florida red tide”? This term is used in annually along Florida waters where species known as Karenia brevis leads to a red colored alga bloom along the Florida coast. Algal blooms are a worldwide environmental problem! This becomes evident with the appointment of 10.000 people for cleaning up the algal bloom in Beijing for the Olympic discipline “sailing regatta” in 2008. Depending on the cause, bacteria algal can bloom at concentrations of hundreds to thousands of cells per milliliter. An officially recognized threshold level isn’t public. Possible diverse factors which are the reason for the high affecting of bloom formation are listed followed:
- Light intensity: cyanobacteria which form surface blooms have a higher tolerance concerning high light intensities. This leads to lower growth rate than other phytoplankton organisms.
- Gas vacuoles: Gas vesicles give the cyanobacteria a lower density than water.
- Growth rate: Slow growth rates require long water retention times to enable a bloom.
- Phosphorous and nitrogen: A low ratio of these substance may favour a development of cyanobacteria blooms.
- Temperature: maximum growth rates are attained by the most cyanobacteria above 25°C. Thus much bloom formation is during the summer.
A possible cascade which is caused by the release of nutrients in the aquatic ecosystems is shown in Figure 8. Another result confirms the spread and the persistence of cholera through algal blooms http://www.ncbi.nlm.nih.gov/pubmed/8155853 Epstein PR.,1993. Currently the so called harmful algal blooms are an additive for the detection of bacteria spread and their caused health effects to fauna and flora http://www.ncbi.nlm.nih.gov/pubmed/21135906 Ferrante et al.,2010 http://www.ncbi.nlm.nih.gov/pubmed/17298369 Barlaan EA et al.,2007. The reduction of the uptake of toxic methylmercury in freshwater food webs by algal blooms confirms a positive reverse of algal blooms http://www.ncbi.nlm.nih.gov/pubmed/11904388 Pickhardt PC. et al.,2002.
Current Solution to combat Algal blooms
It is well known that cyanobacteria produce most Toxins such as microcystin under species-specific conditions which are most favorable for their growth. As a result most studies concerning this topic try to tackle the problem from this point of view. For the investigated species, they found out that the toxin production increased under light optimum condition of the relative bacteria species and on low and high pH. The usage of zinc was necessary for the increase of growth and toxin production. Dependent on nitrogen fixing respectively non-nitrogen fixing species the nitrogen and phosphorous rate in media was unnecessary respectively necessary concerning to the toxin production. Evidences of the role of plasmid, multi-enzyme complexes and peptide synthetase gene involvement in toxin production were also confirmed: e.g. knockout experiments showed that peptide synthetase genes are responsible for microcysteine production.
Major problems are still the lack of knowledge regarding to occurrence and toxicity of cyanobacterial products like the diversity of LPS structures which cause allergic as well as toxic human health problems, the extremely weak scientific basis and poorly developed procedures for valuation on health effects. The TDI (tolerable daily intake) is one of the key features on which the toxicology studies are aimed. Based on analytic and sampling problems or inadequate scientific data safe practice guidelines can be assisted for the reducing of exposure.
The human exposure to the toxins is a major point to combat the human health risk. The WHO guideline values base upon a number of assumptions which change locally or nationally according to the present situation. To fill the lack of knowledge of parental variants the HPLC method is commonly used also for the determination of references. Current treatment processes used at water surface plants are coagulation, sand filtration, and clarification.
The negative site of these methods is the ineffective removal and destruction of cyanotoxins. To combat this problem treatment systems like carbon- and membrane filtration were developed. External and internal accumulation of toxins is another research topic since the inhibition of Mustard seedling development by of microcystein-LR in aquatic root solution.
Dundee 2013 Solution to combat Algal blooms
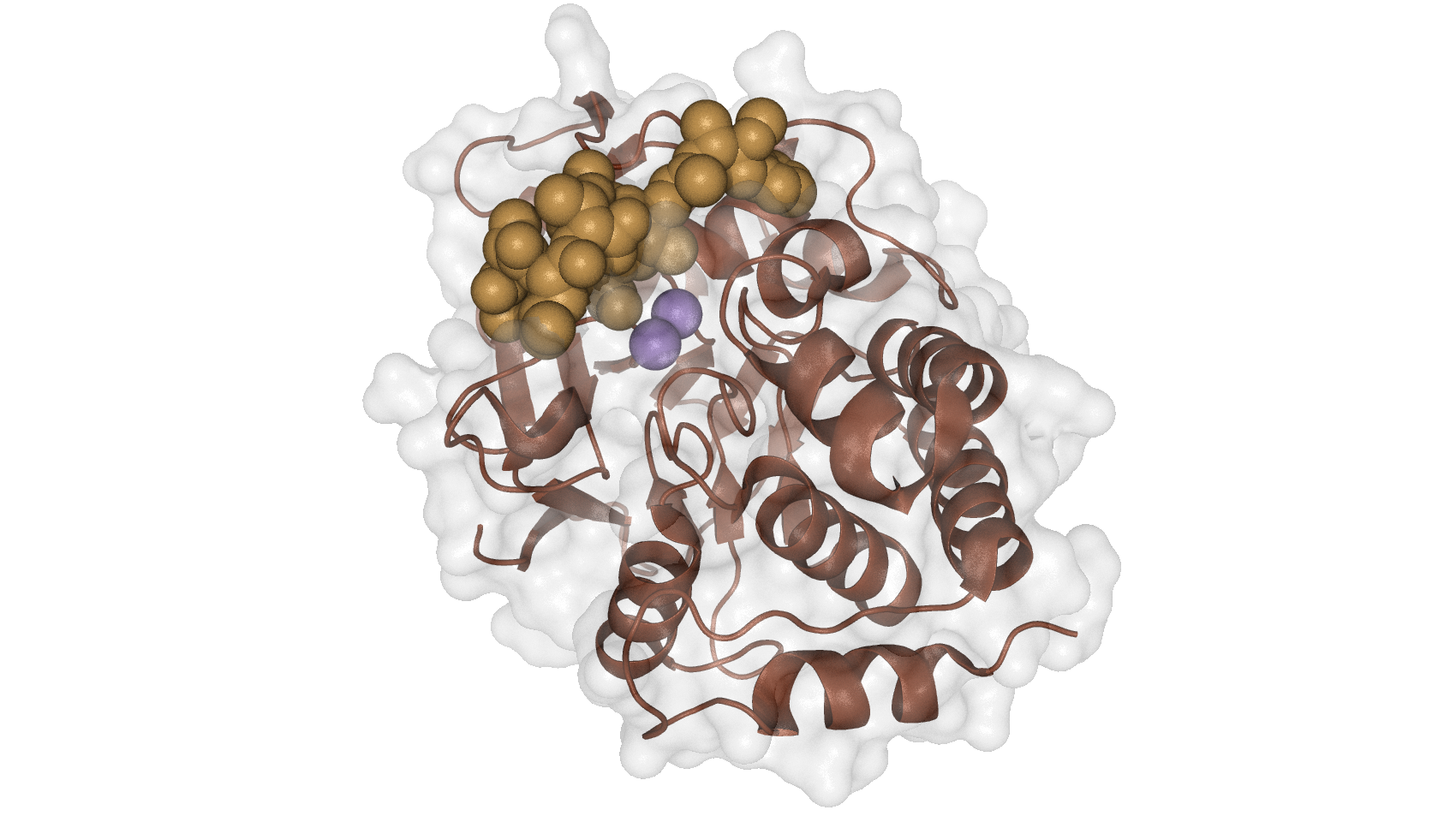
Due to the deleterious results from the toxin release of algal blooms many risk assesments were have been developed http://www.ncbi.nlm.nih.gov/pubmed/10499991 Schaeffer et al., 1999. The effects of microcystin for the aquatic and terrestrial ecosystem have been proven to include the modulation of proliferation, apoptosis and proliferation of spermatogenic cells in vivo http://www.ncbi.nlm.nih.gov/pubmed/24025782 Zhou et al.,2013, which makes it an attractive target for our project.
For the bioaccumulation of microcystin, it is possible to use the enzyme PP1. Mycrocystin can inhibit the function of PP1 by binding to it, this interaction of microcystin, in form of a heptapeptide structure, with three distinct regions of the PP1c is depicted in Figure 9. Hence PP1 can act as natural binding partner for mycrocystin.
As the Dundee iGEM team 2013 Team Dundee was already working on this subject we contacted them and they kindly provided us with the gene that encodes PP1. After converting the human PP1, to RFC 25 and constructing some expression plasmids we produced transgenic phytomitrella patens (PF-14) with PP1 and a biobrick of PP1 receptor.
References:
U.S. Geological Survey,2013 Definiton of Bioaccumulation
http://www.ncbi.nlm.nih.gov/pubmed/15676296 Schlehuber and Skerra,2005 Schlehuber S, Skerra A.(2005)Lipocalins in drug discovery: from natural ligand-binding proteins to "anticalins".Drug Discov Today10(1):23-33
http://www.pnas.org/content/103/15/6061 Yang et al.,2006 Yang Y.,Sulpice R.,Himmelbach A.,Meinhard M.,Christmann A., Grill E. Fibrillin expression is regulated by abscisic acid response regulators and is involved in abscisic acid-mediated photoprotection PNAS 6061–6066
http://www.ws.chemie.tu-muenchen.de/ Institute of Hydrochemistry and Chair for Analytical Chemistry
http://www.ncbi.nlm.nih.gov/pubmed/11950570 Stoger et al.,2002 Stoger E, Sack M, Fischer R, Christou P.(2002) Plantibodies: applications, advantages and bottlenecks Curr Opin Biotechnol. 13(2):161-6.
http://www.ncbi.nlm.nih.gov/pubmed/20470236 Gan, 2010 Gan TJ.(2010) Diclofenac: an update on its mechanism of action and safety profile Curr Med Res Opin26(7):1715-31. doi: 10.1185/03007995.2010.486301
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1724805/ Predel et al.,2004 H Predel, R Koll, H Pabst, R Dieter, G Gallacchi, B Giannetti, M Bulitta, J Heidecker, and E Mueller(2004)Diclofenac patch for topical treatment of acute impact injuries: a randomised, double blind, placebo controlled, multicentre study Br J Sports Med 38(3): 318–323.
http://www.ncbi.nlm.nih.gov/pubmed/11276273 Moragan et al.,2001 Morgan GJ Jr, Kaine J, DeLapp R, Palmer R (2001)Treatment of elderly patients with nabumetone or diclofenac: gastrointestinal safety profile J Clin Gastroenterol32(4):310-4.
http://www.ncbi.nlm.nih.gov/pubmed/15480981 Chan et al.,2004 Chan FK, Hung LC, Suen BY, Wong VW, Hui AJ, Wu JC, Leung WK, Lee YT, To KF, Chung SC, Sung JJ.(2004) Celecoxib versus diclofenac plus omeprazole in high-risk arthritis patients: results of a randomized double-blind trial Gastroenterology 127(4):1038-43.
http://www.ncbi.nlm.nih.gov/pubmed/22168216 Pavelka,2012 Pavelka K.(2012) A comparison of the therapeutic efficacy of diclofenac in osteoarthritis: a systematic review of randomised controlled trials Curr Med Res Opin.28(1):163-78. doi: 10.1185/03007995.2011.649848.
http://www.ncbi.nlm.nih.gov/pubmed/16190224 Westerhoff et al. (2005) Westerhoff, P., Yoon, Y., Snyder, S., and Wert, E. (2005). Fate of endocrine- disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. Environ Sci Technol, 39(17):6649–63.
http://www.ncbi.nlm.nih.gov/pubmed/15984812 Huber et al., (2005) Huber, M. M., G ̈obel, A., Joss, A., Hermann, N., L ̈offler, D., McArdell, C. S., Ried, A., Siegrist, H., Ternes, T. A., and von Gunten, U. (2005). Oxidation of pharmaceuticals during ozonation of municipal wastewater effluents: a pilot study. Environ Sci Technol, 39(11):4290–9.
http://www.ncbi.nlm.nih.gov/pubmed/12697241 Ternes et al., (2003) Ternes, T. A., Stu ̈ber, J., Herrmann, N., McDowell, D., Ried, A., Kampmann, M., and Teiser, B. (2003). Ozonation: a tool for removal of pharmaceuticals, contrast media and musk fragrances from wastewater? Water Res, 37(8):1976–82.
http://www.ncbi.nlm.nih.gov/pubmed/12966990 Deng et al. (2003) Deng, A., Himmelsbach, M., Zhu, Q.-Z., Frey, S., Sengl, M., Buchberger, W., Niessner, R., and Knopp, D. (2003). Residue analysis of the pharmaceutical diclofenac in different water types using elisa and gc-ms. Environ Sci Technol, 37(15):3422–9.
[Homepage iGEM team Freiburg 2008]
http://www.ncbi.nlm.nih.gov/pubmed/10051566 Beste et al., 1999 Beste G, Schmidt FS, Stibora T, Skerra A. (1999) Small antibody-like proteins with prescribed ligand specificities derived from the lipocalin fold. PNAS, 96(5):1898-903.
http://www.ncbi.nlm.nih.gov/pubmed/16307475 Vopel et al., 2005 Vopel S, Mühlbach H, Skerra A. (2005) Rational engineering of a fluorescein-binding anticalin for improved ligand affinity. Biol. Chem., 386(11):1097-104.
http://www.sciencedirect.com/science/article/pii/0965174895000909 La-Aied Prapanthadara et al.,1996 La-Aied Prapanthadara,Surangchit Koottathe,Nongkran Promtet,Janet Hemingway†,Albert J., Ketterman (1996) Purification and characterization of a major glutathione S-transferase from the mosquito Anopheles dirus (Species B) Insect Biochemistry and Molecular BiologyVolume 26, Issue 3 Pages 277–285
http://www.ncbi.nlm.nih.gov/pubmed/11836138 Turusov et al.,2002 Turusov V, Rakitsky V, Tomatis L.(2002)Dichlorodiphenyltrichloroethane (DDT): ubiquity, persistence, and risksEnviron Health Perspect.110(2):125-8
[Homepage iGEM team Caltech 2011]
http://www.ncbi.nlm.nih.gov/pubmed/21428697 Oakley,2006 Oakley A.(2011)Drug Metab Rev43(2):138-51. doi: 10.3109/03602532.2011.558093
http://www.ncbi.nlm.nih.gov/pubmed/16550164 McIlwain et al.,2006 McIlwain CC, Townsend DM, Tew KD(2006)Glutathione S-transferase polymorphisms: cancer incidence and therapy Oncogene25(11):1639-48
http://www.ncbi.nlm.nih.gov/pubmed/8770536 Hayes et al.,1995Hayes JD, Pulford DJ.(1995) The glutathione S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance.Crit Rev Biochem Mol Biol.30(6):445-600
http://www.ncbi.nlm.nih.gov/pubmed/20981235 Josephy, 2010 Josephy PD.(2010)Genetic variations in human glutathione transferase enzymes: significance for pharmacology and toxicology.Hum Genomics Proteomics 2010:876940. doi: 10.4061/2010/876940
http://www.ncbi.nlm.nih.gov/pubmed/10971201 Hayes et al.,2000Hayes JD, Strange RC.(2000)Glutathione S-transferase polymorphisms and their biological consequencesPharmacology 61(3):154-66
http://www.ncbi.nlm.nih.gov/pubmed/12563680 Fraser et al., 2003Fraser PA, Ding WZ, Mohseni M, Treadwell EL, Dooley MA, St Clair EW, Gilkeson GS, Cooper GS. (2003),Glutathione S-transferase M null homozygosity and risk of systemic lupus erythematosus associated with sun exposure: a possible gene-environment interaction for autoimmunityJ Rheumatol30(2):276-82
http://www.ncbi.nlm.nih.gov/pubmed/11038270 Kamencic et al.,2000 Kamencic H, Lyon A, Paterson PG, Juurlink BH.(2000)Monochlorobimane fluorometric method to measure tissue glutathione.Anal Biochem 286(1):35-7
http://www.ncbi.nlm.nih.gov/pubmed/8155853 Epstein PR.,1993 Epstein PR.(1993),Algal blooms in the spread and persistence of Cholera Biosystems 31(2-3):209-21
http://www.ncbi.nlm.nih.gov/pubmed/21135906 Ferrante M. et al., 2010 Ferrante M, Ledda C, Cunsolo MA, Fiore M, Fallico R, Sciacca S, Oliveri Conti GM (2010) Harmful algal blooms in Italy and their health effects in the Population Ig Sanita Pubbl. 66(5):649-58
http://www.ncbi.nlm.nih.gov/pubmed/17298369 Barlaan EA et al.,2007 Barlaan EA, Furukawa S, Takeuchi K.(2007) Detection of bacteria associated with harmful algal blooms from coastal and microcosm environments using electronic microarrays Environ Microbiol9(3):690-702.
http://www.ncbi.nlm.nih.gov/pubmed/11904388 Pickhardt PC et al.,2002 Pickhardt PC, Folt CL, Chen CY, Klaue B, Blum JD.(2002) Algal blooms reduce the uptake of toxic methylmercury in freshwater food webs Proc Natl Acad Sci U S A.99(7):4419-23. Epub 2002 Mar 19.
http://www.ncbi.nlm.nih.gov/pubmed/10499991 Schaeffer et al.,1995 Schaeffer DJ, Malpas PB, Barton LL.(1995),Risk assessment of microcystin in dietary Aphanizomenon flos-aquae,Ecotoxicol Environ Saf.,44(1):73-80
http://www.ncbi.nlm.nih.gov/pubmed/24025782 Zhou et al.,2013 Zhou Y, Chen Y, Yuan M, Xiang Z, Han X.(2013)In vivo study on the effects of microcystin-LR on the apoptosis, proliferation and differentiation of rat testicular spermatogenic cells of male rats injected i.p. with Toxins J Toxicol Sci.38(5):661-70
[Homepage iGEM team Dundee 2013]
[[http://www.nabu.de/tiereundpflanzen/voegel/international/03530.html "Katastrophales Geiersterben in Indien"(2005),NABU]]
http://www.spiegel.de/wissenschaft/natur/insektengift-ddt-wie-die-malaria-wunderwaffe-bauern-in-die-armut-treibt-a-768654.html Koch L.,2011Koch L.(2011)Insektengift DDT: Wie die Malaria-Wunderwaffe Bauern in die Armut treibt Spiegel online
Chorus I.,Bartram J.(1999),Toxic Cyanobacteria in Water:A guide to their public health consequences,monitoring and Management,E & FN Spon, an imprint of Routledge 11 New Fetter Lane, London EC4P 4EE
Jiskra,M.,Fate of the pharmaceutical diclofenac in the aquatic Environment,Term paper 21. In Biogeochemistry and Pollutant Dynamics
Kulshrestha,Kumari,Simultaneous Degradation of Mixed Insecticides by Mixed Fungal Culture Isolated from Sewage Sludge(2010),J. Agric. Food Chem., 58, 11852–11856 DOI:10.1021/jf1028202
Tomlinson,T.G.,Control by D.D.T of Flies Breeding in Percolating Sewage Filters(1945),Water pollution research laboratory,Minworth Birmingham,Nature
 "
"




AutoAnnotator:
Follow us:
Address:
iGEM Team TU-Munich
Emil-Erlenmeyer-Forum 5
85354 Freising, Germany
Email: igem@wzw.tum.de
Phone: +49 8161 71-4351