Team:Paris Saclay/Notebook/August/9
From 2013.igem.org
(Difference between revisions)
CarolineMir (Talk | contribs) (→1 - Tranduction of Km in MG1655Z1) |
|||
| Line 7: | Line 7: | ||
==='''A - Aerobic/Anaerobic regulation system'''=== | ==='''A - Aerobic/Anaerobic regulation system'''=== | ||
| - | ===='''Objective : characterize | + | ===='''Objective : characterize BBa_K1155000, BBa_K1155004, BBa_K1155005, BBa_K1155006'''==== |
===='''1 - Tranduction of Km in MG1655Z1 ==== | ===='''1 - Tranduction of Km in MG1655Z1 ==== | ||
| Line 32: | Line 32: | ||
We used kanamycine antibiotic. | We used kanamycine antibiotic. | ||
| - | ===='''Objective : obtaining | + | ===='''Objective : obtaining BBa_K1155007'''==== |
| - | ====1 - Extraction of | + | ====1 - Extraction of BBa_K115007 from DH5α==== |
Abdou | Abdou | ||
| Line 43: | Line 43: | ||
Nanodrop | Nanodrop | ||
| - | * | + | * BBa_K1155007 in clone 10 : 38ng/µl |
| - | * | + | * BBa_K1155007 in clone 14 : 48.5ng/µl |
| - | * | + | * BBa_K1155007 in clone 15 : 52 ng/µl |
{| | {| | ||
Revision as of 09:19, 30 September 2013
Notebook : August 9
Lab work
A - Aerobic/Anaerobic regulation system
Objective : characterize BBa_K1155000, BBa_K1155004, BBa_K1155005, BBa_K1155006
1 - Tranduction of Km in MG1655Z1
Abdou, Anaïs, Damir, Nadia, XioaJing
|
We observed lysis areas. We will continue the transduction protocol. |
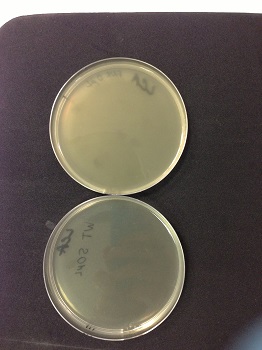
|
Picture: lysed cells comparison.
|
Protocol : Transduction
Our Mutant bacteria was called BW : Δfnr::Km. Our wild type bacteria was called MG1655Z1. We used kanamycine antibiotic.
Objective : obtaining BBa_K1155007
1 - Extraction of BBa_K115007 from DH5α
Abdou
Protocol : Hight copy plamid extraction
We used colonies number 10, 14 and 15.
Nanodrop
- BBa_K1155007 in clone 10 : 38ng/µl
- BBa_K1155007 in clone 14 : 48.5ng/µl
- BBa_K1155007 in clone 15 : 52 ng/µl
|
The extraction was good. We will sequence our plasmid. |
A - Aerobic/Anaerobic regulation system / B - PCB sensing system
Objective : Obtaining FNR and BphR2 proteins
1 - Electrophoresis of the PCR of BphR2 Part I, BphR2 Part II, RBS_BphR2 Part I, FNR Part I, FNR Part II, RBS_FNR Part I to check the gel purification
| IMAGE |
|
Expected size :
- BphR2 Part I :
- BphR2 Part II :
- RBS-BphR2 Part I :
- FNR Part I :
- FNR Part II :
- RBS-FNR PartI :
|
We lost all our PCR fragments. We will do the PCR again. |
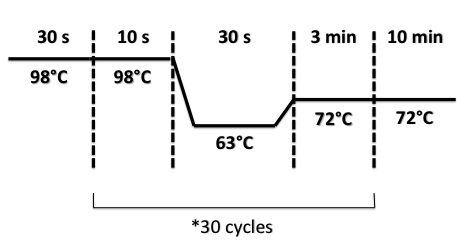
2 - PCR of BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I
Anaïs, Damir, Nadia, XiaoJing
Used quantities :
- Bphr2 Part I :
- Oligo 54F : 1µL
- Oligo 55R : 1µL
- Buffer Phusion : 10µL
- DNA of Pseudomonas pseudoalcaligenes : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
- Bphr2 Part II :
- Oligo 56F : 1µL
- Oligo 57R : 1µL
- Buffer Phusion : 10µL
- DNA Pseudomonas pseudoalcaligenes : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
- RBS-Bphr2 Part I :
- Oligo 58F : 1µL
- Oligo 57R : 1µL
- Buffer Phusion : 10µL
- DNA Pseudomonas pseudoalcaligenes : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
- FNR Part I :
- Oligo 59F : 1µL
- Oligo 60R : 1µL
- Buffer Phusion : 10µL
- DNA Escherichia coli : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
- FNR Part II :
- Oligo 61F : 1µL
- Oligo 62R : 1µL
- Buffer Phusion : 10µL
- DNA Escherichia coli : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
- RBS-FNR Part I :
- Oligo 63F : 1µL
- Oligo 62R : 1µL
- Buffer Phusion : 10µL
- DNA Escherichia coli : 1µL
- dNTP : 1µL
- Phusion : 0.5µL
- H2O : 35.5µL
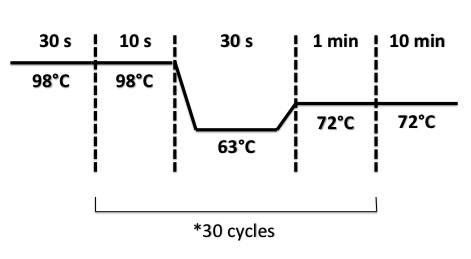
PCR Program :
- BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I :
- FNR Part I, FNR Part II, RBS-FNR Part I :
| Previous day | Back to calendar | Next day |
 "
"