Team:TU-Eindhoven/KillingMechanism
From 2013.igem.org
(→The Alternative System: The 5-Fluorocytosine/ Cytosine Deaminase System) |
JacquesErnes (Talk | contribs) (→The Alternative System: The 5-Fluorocytosine/ Cytosine Deaminase System) |
||
| (46 intermediate revisions not shown) | |||
| Line 5: | Line 5: | ||
{{:Team:TU-Eindhoven/Template:Abstract}} | {{:Team:TU-Eindhoven/Template:Abstract}} | ||
| - | In order to terminate our device after the CEST MRI has been acquired, we propose the implementation of the HSV-TK/GCV system in our bacteria. The implementation of this system requires that the CEST contrast producing bacteria are | + | In order to terminate our device after the CEST MRI has been acquired, we propose the implementation of the HSV-TK/GCV system in our bacteria. The implementation of this system requires that the CEST contrast producing bacteria are transformed with HSV Thymidine Kinase, so that cell death can be induced when the prodrug Ganciclovir is injected into the system. Directed cell death will only be induced in the cells that express the TK enzyme. This means that by implementing this controllable safety lock we can terminate our bacterial device at any point of the MRI process by administering Ganciclovir to the patient. |
| - | + | We propose to use this system as our killing mechanism, considering that it is well characterized and used in applications related to the principle of our device. However, we also suggest an alternative killing mechanism that could be implemented if the first one was to fail in successfully deactivating and eliminating our device from the patient. | |
{{:Team:TU-Eindhoven/Template:AbstractEnd}} | {{:Team:TU-Eindhoven/Template:AbstractEnd}} | ||
| - | {{:Team:TU-Eindhoven/Template:FloatingImage | position=left | size=1| filename=bacteriaSad.png}}<br> | + | <!--{{:Team:TU-Eindhoven/Template:FloatingImage | position=left | size=1| filename=bacteriaSad.png}}<br>--> |
==The Kill Switch in Detail: The Ganciclovir /Thymidine Kinase System== | ==The Kill Switch in Detail: The Ganciclovir /Thymidine Kinase System== | ||
| - | + | {{:Team:TU-Eindhoven/Template:Float | position=right | size=8 }} | |
| + | {{:Team:TU-Eindhoven/Template:Image | filename=killMechanismI.png}} | ||
| + | {{:Team:TU-Eindhoven/Template:FloatEnd | caption=Cell death induced by the Ganciclovir /Thymidine Kinase system in the CEST protein producing bacteria.| id=kmIFigure }} | ||
| + | A rapidly evolving research area in the field of oncology is the one focused on bacterial based cancer therapies. In this type of therapies, bacteria like ''E. Coli'' and ''Salmonella typhimurium'' are used as tumor targeting systems. These bacteria are used to deliver cytotoxic agents, immunostimulatory cytokine, and genetic material to enable host-cell production of these molecules, and induce cell death in this way. {{:Team:TU-Eindhoven/Template:Ref | id=forbesBact | author=N.S. Forbes | title=Engineering the perfect(bacterial) cancer therapy | journal=Nature | edition= | pages=785-794 | year=2010 }} Using the bacteria for delivering genetic material to the tumor cells is similar to the suicide gene therapy, also known as {{:Team:TU-Eindhoven/Template:Tooltip | text= GDEPT | tooltip=gene-directed enzyme prodrug therapy}}, which is widely used in cancer treatment. {{:Team:TU-Eindhoven/Template:Ref | id=BugajNextGenTher | author=L.J. Bugaj, D.V. Schaffer | title=Bringing next-generation therapeutics to the clinic through synthetic biology | journal=Current Opinion in Chemical Biology | edition=16 | pages=355-361 | year=2012 }} One of the best characterized and used GDEPT systems is the {{:Team:TU-Eindhoven/Template:Tooltip | text= HSV-TK | tooltip=herpes simplex virus Thymidine Kinase}} enzyme with purine nucleoside analog {{:Team:TU-Eindhoven/Template:Tooltip | text= GCV | tooltip=Ganciclovir}} as a prodrug. Systemic administration of the prodrug Ganciclovir induces cell death only in the cells that express the HSV-Thymidine Kinase enzyme, sparing the life of the cells that do not express this enzyme. {{:Team:TU-Eindhoven/Template:Ref | id=aghiEtAlGCVFlu | author=M. Aghi, C.M. Kramm, T.C. Chou, X.O. Breakefield, E.A. Chiocca | title= Synergistic Anticancer Effects of Ganciclovir/Thymidine Kinase and 5-Fluorocytosine/Cytosine Deaminase Gene Therapies | journal=Journal of the National Cancer Institute | edition=90 | pages=370-380 | year=1998 }} {{:Team:TU-Eindhoven/Template:Figure | id=kmIFigure }} depicts the complete process of cell death induced by the HSV-TK/GCV system. | ||
| - | |||
| - | + | We propose to use this system as our killing mechanism, considering that it is well characterized and used in applications related to the principle of our device. However at this point of our project the ''in vitro'' implementation of this system is not possible and thus we focused on a pharmacokinetics model of the prodrug distribution in the body and the elimination of the CEST protein producing bacteria from the system. The following section describes the main results from the pharmacokinetics model. | |
| - | + | ||
| - | + | ||
| - | + | ||
==The TK/GCV System: Implementation== | ==The TK/GCV System: Implementation== | ||
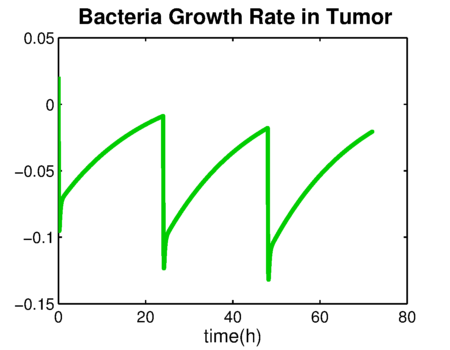
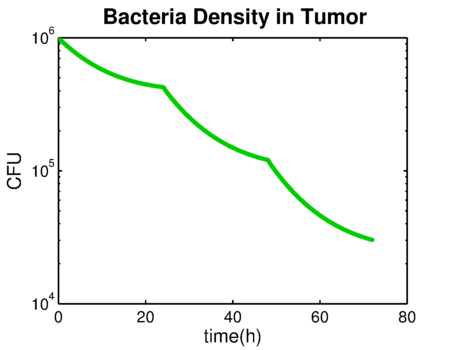
| - | We developed a pharmacokinetics model of the prodrug Ganciclovir distribution and the systemic elimination of the CEST protein production device. The | + | We developed a pharmacokinetics model of the prodrug Ganciclovir distribution and the systemic elimination of the CEST protein production device. The model and results can be found in the [[Team:TU-Eindhoven/PBPK | Drylab section]] of our wiki. The results from the pharmacokinetics model indicate that when administrating 5mg/L of Ganciclovir intravenously to the patient per day, the drug reaches its highest concentration on the tumor region of approximately .23mg/L in 50 hours since treatment. This concentration gradually decreases over the following hours after administration of the prodrug. The total elimination rate of the Ganciclovir is 1.2748 h<sup>-1</sup>. Furthermore, the elimination of bacteria due to the effect of the Ganciclovir is depicted in the following plots ({{:Team:TU-Eindhoven/Template:Figure | id=bacteriaGrowthRate}} and {{:Team:TU-Eindhoven/Template:Figure | id=bacteriaDensity}}). |
| - | + | {{:Team:TU-Eindhoven/Template:Float | position=left| size=6 }} | |
| - | + | {{:Team:TU-Eindhoven/Template:Image | filename=bacteriaGrowthRate.png}} | |
| + | {{:Team:TU-Eindhoven/Template:FloatEnd | caption=Growth Rate of Bacteria under GCV administration | id=bacteriaGrowthRate }} | ||
| + | |||
| + | {{:Team:TU-Eindhoven/Template:Float | position=right| size=6 }} | ||
| + | {{:Team:TU-Eindhoven/Template:Image | filename=bacteriaDensity.png}} | ||
| + | {{:Team:TU-Eindhoven/Template:FloatEnd | caption=Bacteria Density in Tumor under GCV administration | id=bacteriaDensity }} | ||
==The Alternative System: The 5-Fluorocytosine/ Cytosine Deaminase System== | ==The Alternative System: The 5-Fluorocytosine/ Cytosine Deaminase System== | ||
| - | |||
| - | {{:Team:TU-Eindhoven/Template:Float | position= | + | {{:Team:TU-Eindhoven/Template:Float | position=left | size=8 }} |
{{:Team:TU-Eindhoven/Template:Image | filename=killMechanismII.png}} | {{:Team:TU-Eindhoven/Template:Image | filename=killMechanismII.png}} | ||
| - | {{:Team:TU-Eindhoven/Template:FloatEnd | caption= | + | {{:Team:TU-Eindhoven/Template:FloatEnd | caption=Cell death induced by the 5-Fluorocytosine/ Cytosine Deaminase system in the CEST protein producing bacteria. | id=kmIFigure1 }} |
| + | |||
| + | Similar to the HSV-TK/GCV GDEPT system, is the ''Escherichia coli'' {{:Team:TU-Eindhoven/Template:Tooltip | text= CD | tooltip=cytosine deaminase}} enzyme with prodrug 5-fluorocytosine. Like the prodrug Ganciclovir, systemic administration of the prodrug 5-fluorocytosine induces cell death only in the cells that express the cytosine deaminase enzyme. Cell death is induced when the CD deaminates the prodrug 5-fluorocytosine, forming 5-fluoro-uracil, which is further metabolized into {{:Team:TU-Eindhoven/Template:Tooltip | text= FdUMP | tooltip=5-fluoro-28-deoxyuridine-58-monophosphate }}.<!--5-fluorouracil is then metabolized into 5-fluoro-28-deoxyuridine-58-monophosphate (FdUMP), 5- fluoro-28-deoxyuridine-58-triphosphate (FdUTP), and 5- fluorouridine-58-triphosphate (FUTP).--> FdUMP inhibits the enzyme thymidylate synthetase which converts dUMP into {{:Team:TU-Eindhoven/Template:Tooltip | text= dTMP | tooltip=thymidylate }}. The inhibition of the enzyme causes the inhibition of DNA synthesis, leading to cell death. {{:Team:TU-Eindhoven/Template:RefAgain | id=aghiEtAlGCVFlu }} Figure 2 shows a detailed graphic explanation of the complete process. | ||
| + | |||
| + | Given that the chassis of our device is'' E. coli'', the implementation of the CD/5’fluorocytosine system on our device could be an alternative to the HSV-TK/GCV system. We didn’t explore this alternative further but we will consider it when testing the kill switch ''in vitro'' in case the HSV-TK/GCV is not successful in inducing cell death in our CEST protein producing bacteria. | ||
==References== | ==References== | ||
| Line 48: | Line 55: | ||
{{:Team:TU-Eindhoven/Template:UseReferencing}} | {{:Team:TU-Eindhoven/Template:UseReferencing}} | ||
{{:Team:TU-Eindhoven/Template:UseFigures}} | {{:Team:TU-Eindhoven/Template:UseFigures}} | ||
| + | <html> | ||
| + | <script language="javascript"> | ||
| + | $('.tue-toc-well').hide(); | ||
| + | </script> | ||
| + | </html> | ||
Latest revision as of 10:59, 26 October 2013



Contents |
The Kill Switch as a Safety Lock


We propose to use this system as our killing mechanism, considering that it is well characterized and used in applications related to the principle of our device. However, we also suggest an alternative killing mechanism that could be implemented if the first one was to fail in successfully deactivating and eliminating our device from the patient.
The Kill Switch in Detail: The Ganciclovir /Thymidine Kinase System
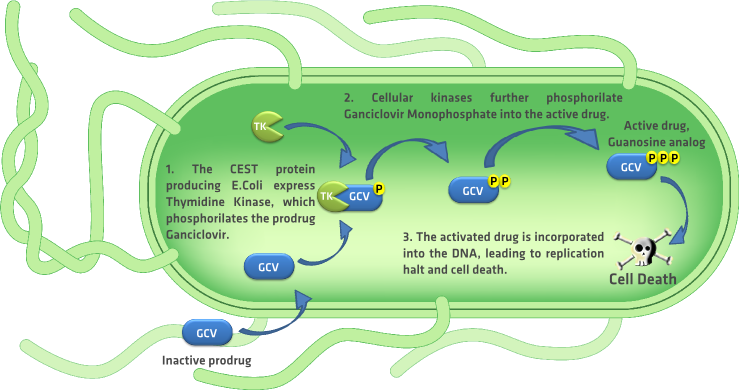
A rapidly evolving research area in the field of oncology is the one focused on bacterial based cancer therapies. In this type of therapies, bacteria like E. Coli and Salmonella typhimurium are used as tumor targeting systems. These bacteria are used to deliver cytotoxic agents, immunostimulatory cytokine, and genetic material to enable host-cell production of these molecules, and induce cell death in this way. forbesBactN.S. Forbes, Engineering the perfect(bacterial) cancer therapy. Nature , 785-794 (2010) Using the bacteria for delivering genetic material to the tumor cells is similar to the suicide gene therapy, also known as GDEPT, which is widely used in cancer treatment. BugajNextGenTherL.J. Bugaj, D.V. Schaffer, Bringing next-generation therapeutics to the clinic through synthetic biology. Current Opinion in Chemical Biology 16, 355-361 (2012) One of the best characterized and used GDEPT systems is the HSV-TK enzyme with purine nucleoside analog GCV as a prodrug. Systemic administration of the prodrug Ganciclovir induces cell death only in the cells that express the HSV-Thymidine Kinase enzyme, sparing the life of the cells that do not express this enzyme. aghiEtAlGCVFluM. Aghi, C.M. Kramm, T.C. Chou, X.O. Breakefield, E.A. Chiocca, Synergistic Anticancer Effects of Ganciclovir/Thymidine Kinase and 5-Fluorocytosine/Cytosine Deaminase Gene Therapies. Journal of the National Cancer Institute 90, 370-380 (1998) depicts the complete process of cell death induced by the HSV-TK/GCV system.
We propose to use this system as our killing mechanism, considering that it is well characterized and used in applications related to the principle of our device. However at this point of our project the in vitro implementation of this system is not possible and thus we focused on a pharmacokinetics model of the prodrug distribution in the body and the elimination of the CEST protein producing bacteria from the system. The following section describes the main results from the pharmacokinetics model.
The TK/GCV System: Implementation
We developed a pharmacokinetics model of the prodrug Ganciclovir distribution and the systemic elimination of the CEST protein production device. The model and results can be found in the Drylab section of our wiki. The results from the pharmacokinetics model indicate that when administrating 5mg/L of Ganciclovir intravenously to the patient per day, the drug reaches its highest concentration on the tumor region of approximately .23mg/L in 50 hours since treatment. This concentration gradually decreases over the following hours after administration of the prodrug. The total elimination rate of the Ganciclovir is 1.2748 h-1. Furthermore, the elimination of bacteria due to the effect of the Ganciclovir is depicted in the following plots ( and ).
The Alternative System: The 5-Fluorocytosine/ Cytosine Deaminase System
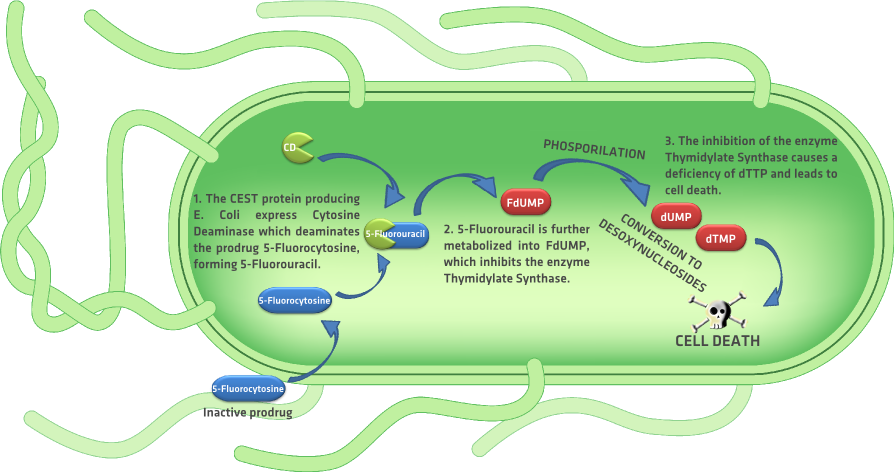
Similar to the HSV-TK/GCV GDEPT system, is the Escherichia coli CD enzyme with prodrug 5-fluorocytosine. Like the prodrug Ganciclovir, systemic administration of the prodrug 5-fluorocytosine induces cell death only in the cells that express the cytosine deaminase enzyme. Cell death is induced when the CD deaminates the prodrug 5-fluorocytosine, forming 5-fluoro-uracil, which is further metabolized into FdUMP. FdUMP inhibits the enzyme thymidylate synthetase which converts dUMP into dTMP. The inhibition of the enzyme causes the inhibition of DNA synthesis, leading to cell death. aghiEtAlGCVFlu Figure 2 shows a detailed graphic explanation of the complete process.
Given that the chassis of our device is E. coli, the implementation of the CD/5’fluorocytosine system on our device could be an alternative to the HSV-TK/GCV system. We didn’t explore this alternative further but we will consider it when testing the kill switch in vitro in case the HSV-TK/GCV is not successful in inducing cell death in our CEST protein producing bacteria.
References
 "
"



