Team:TU-Munich/Results/GM-Moss
From 2013.igem.org
Creation of transgenic Physcomitrella patens plants
Generation of expression constructs
| Number | Construct name (abbreviation) | BioBrick | Figure | Size [bp] | Lane | GM-Moss |
|---|---|---|---|---|---|---|
| PF-1 | GFP (GFP-cyt) | <partinfo>BBa_K1159311</partinfo> | x | 
| ||
| PF-2 | Igk-GFP (GFP-sec) | <partinfo>BBa_E0040</partinfo> | x | 
| ||
| PF-3 | NanoLuciferase (nLuc-cyt) | <partinfo>BBa_K1159001</partinfo> | x | 
| ||
| PF-4 | Igk-NanoLuciferase (nLuc-sec) | <partinfo>BBa_K1159006</partinfo> | x | 
| ||
| PF-5 | SERK-NanoLuciferase (SERK-nLuc) | <partinfo>BBa_K1159010</partinfo> | x | 
| ||
| PF-6 | NanoLuciferase-Receptor (nLuc-rec) | <partinfo>BBa_K1159015</partinfo> | x | 
| ||
| PF-7 | Erythromycinesterase (EreB-cyt) | <partinfo>BBa_K1159000</partinfo> | x | 
| ||
| PF-8 | Erythromycinesterase-Receptor (EreB-rec) | <partinfo>BBa_K1159014</partinfo> | # | 
| ||
| PF-9 | Ig Kappa Erythromycinesterase (EreB-sec) | <partinfo>BBa_K1159005</partinfo> | # | 
| ||
| PF-10 | Laccase-Receptor (Lac-rec) | <partinfo>BBa_K1159016</partinfo> | # | 
| ||
| PF-11 | Ig Kappa Laccase (Lac-sec) | <partinfo>BBa_K1159007</partinfo> | # | 
| ||
| PF-12 | Catecholdioxygenase (XylE-cyt) | <partinfo>BBa_E0040</partinfo> | x | 
| ||
| PF-13 | DDT-dehydrochlorinase (GST-cyt) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-14 | PP1-Receptor (PP1-rec) | <partinfo>BBa_K1159019</partinfo> | x | 
| ||
| PF-15 | FluA-Receptor (FluA-rec) | <partinfo>BBa_K1159017</partinfo> | x | 
| ||
| PF-16 | Stressinducible_Promoter-RFP (Stress) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-17 | SpyCatcher-Receptor:Spytag-nLuc (Catcher:Tag-nLuc) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-18 | SpyCatcher-Receptor:nLuc-Spytag (Catcher:nLuc-Tag | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-19 | SpyTag-Receptor:SpyCatcher-nLuc (Tag:Catcher-nLuc) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-20 | SpyTag-Receptor:nLuc-SpyCatcher (Tag:nLuc-Catcher) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-21 | Alcohol acetyltransferase I (Banana) | <partinfo>BBa_J45014</partinfo> | # | 
| ||
| PF-22 | Kill-switch with PIF3 (PIF-3) | <partinfo>BBa_E0040</partinfo> | x | no | ||
| PF-23 | Kill-switch with PIF6 (PIF-6) | <partinfo>BBa_E0040</partinfo> | x | no | ||
| PF-24 | Kill-switch with PIF6 (FRET) | <partinfo>BBa_E0040</partinfo> | # | 
| ||
| PF-25 | Kill-switch with PIF3 (FRET) | <partinfo>BBa_E0040</partinfo> | # | 
|
Preparation of linear DNA
In order to transfrom the moss, the DNA including our BioBricks with the nptII selection casette had to be linearized. We used EcoRI for the preparative digestion of our midipreps and simultaneously ran an analytical digest as quality control. Our preparations continued by purifying our digested DNA through ethanol precipitation, measuring the concentration at the NanoDrop and suspending it in different volumes of sterile filtered Ca(NO3)2 buffer for the intended concentration of 0.25 µg/µl .
Transformation of Physcomitrella patens
To transform Physcomitrella patens, the moss material has to be taken from the liquid culture and its cell walls have to be digested with driselase dissolved in mannitol to obtain protoplasts. The protoplasts are isolated by passing the digested material through sieves and the enzyme is washed off with mannitol and then resuspended in mannitol.
The number of protoplasts is determined with a hemocytometer and the material is suspended in the right amount of 3M medium to adjust the concentration. The linearized and purified DNA is mixed with PEG4000 and the protoplast solution and incubated while regularly mixing. After incubation, the mixture is diluted with 3M medium, centrifuged and resuspended in regeneration medium.
The protoplasts are put into 6-well plates, left in the dark over night and then left for 10 days for the regeneration of the cell walls at standard conditions. After moving the protoplasts onto solid medium covered with a layer of cellophane for three days, they are transferred to solid selection medium plates for two weeks. To ensure stabile integration, repeat the two weeks of selection after a two week release phase.
Trips to Freiburg
We had the great chance to perform our Physcomitrella patens transformations at Prof. Dr.Reski´s lab in Freiburg, with Dr. Wiedemann as our expert instructor. Our first trip started in a great hurry, because we worked on our DNA preparations until the very last minute. Ingmar even pulled an all-nighter to get the DNA ready. We would have missed our intercity bus if it wasn’t for Rosario who drove us to the bus station, all squished together in the “pizza mobile” with the trunk full with our medium bottles and lab equipment. After five hours on the bus, we fell into bed to get some rest, because we had a very long day ahead of us.
We arrived at the Reski lab early in the morning to meet with Dr. Gertrud Wiedemann, who instructed us throughout the day and gave us many tips how to proceed with the moss. Because of the incubation times and because it was our first try, it took us ten hours without a break until we had two boxes stacked with 6 well plates. We quickly went to get some beers and chips and met with the Freiburg iGEM team for a really nice barbecue. When we finally left, public transport wasn’t running anymore, so we didn’t miss the chance to take a midnight sightseeing tour through Freiburg, where Volker showed us around.
Ten days later, Johanna and Andi visited the Freiburg lab to transfer the then regenerated protoplasts onto agar plates and soon after, we came back for our second and final round of transformation. At our first try, we didn’t get enough moss protoplasts, so we worked through two batches and therefore had to prepare another batch of driselase. Our handling had improved, yet it still took twelve hours and again, there was no time left for a break. After we said good bye, we celebrated with a couple of beers and some yummy flammkuchen at the UC uni café of Freiburg. We had learned so much and got much closer to our goal. A big successful step for our team!
Regeneration and Selection of transgenic plants
Characterization of transgenic plants
Investigation of the localization within the moss cell
Idea
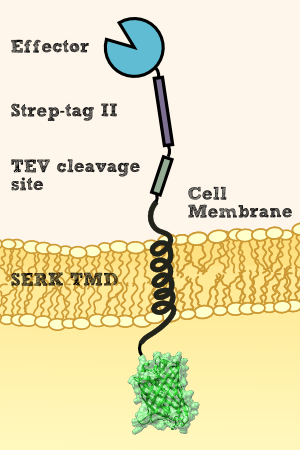
We want to show that our membrane construct, which contains Nanoluciferase fused to a Streptag for purification and a TEV split site to cut of the Nanoluciferase-Streptag-Construct in the Periplasma between cell-membrane and cell-wall by using the TEV-Protease, is expressed and afterwards stocked in the membrane of our moss.
To prove this process, we cut off our Nanoluciferase-Streptag-Construct by using the TEV-Protease. As we hypothetically know, our membrane construct is localized in the membrane and the Nanoluciferase-Streptag-Construct orientated to the Periplasma, protected by the cell-wall of our moss. In conclusion to that, the TEV protease needs to pass the cell-wall to cut off the Nanoluciferase-Streptag-Construct. This process is possible in liquid medium, because the TEV protease just has a molecular weight between 25 kDa and 27 kDa, so it can pass the cell-wall smoothly. After the TEV-Protease has entered the Periplasma of our moss cells, it cuts off the Nanoluciferase-Streptag-Construct at the TEV cleavage site. Again, this construct just has in sum a molecular weight of 20 kDa (Nanoluciferase - 19 kDa, Streptag - 1 kDa), so it can pass the cell-wall due to diffusion to get to the exterior of the moss cell.
After this experimental part, it is necessary to prove the theoretical deliberation. In the following we performed several experiments like SDS-Pages, Luciferase Assays and tried to bind our construct by utilizing different beads.
Experimental Setup for localization process of membrane bound Nanoluciferase
First Step
The reagents which were necessary to perform the first step have been 1,9 ml TEV Protease Buffer, 100 µl TEV Protease and as much genetically modified moss, containing the membrane bound Streptag-Luciferase-Construct, as possible. All reagents have been added to a 2 ml Tube, gently mixed and incubated over night at 4°C.
Second Step
In the second step, the 2 ml tube was centrifuged at 13200 rpm for 5 minutes to make the moss cells form a pellet at the bottom of the tube. Afterwards the supernatant was transferred to a new 2 ml tube to isolate the supernatant where the Nanoluciferase-Streptag-Construct was expected to be found. Two slightly different approaches of Immunoprecipitation have been performed to try to bind the cut-off construct.
First 1 ml of the supernatant was incubated with 20 µl of an antibody called Strepmab-Immo (1 mg/ml), which is able to bind the cut-off construct at the Streptag. After an incubation time of four hours, 100 µl Dynabeads spiked with Protein G have been added to the tube and incubated over night. Protein G is able to bind the Fc-Part of the Strepmab-Immo Antibody. The aim is to bind the Streptag-Nanoluciferase-Construct with the Strepmab-Immo Antibody and the resulting construct with the Protein G anchored magnetic Dynabeads. To get to know if the resulting construct of Protein-G Dynabeads and Strepmab-Immo Antibody are fluorescent active in the same range of the electromagnetic spectrum like the Nanoluciferase, a negative control containing 5 µl Dynabeads and 1 µl Strepmab-Immo Antibody was prepared.
The second approach was to use Biotin-labeled beads to bind the Nanoluciferase-Streptag-Construct. Again 20 µl of Biotin labeled beads have been added to 1 ml of the supernatant, carefully mixed and incubated over night. A negative control containing Phosphate-Bufferd-Saline (PBS) buffer and 5 µl Biotin-labeled beads was established, because of the same reason described above, too.
Third step
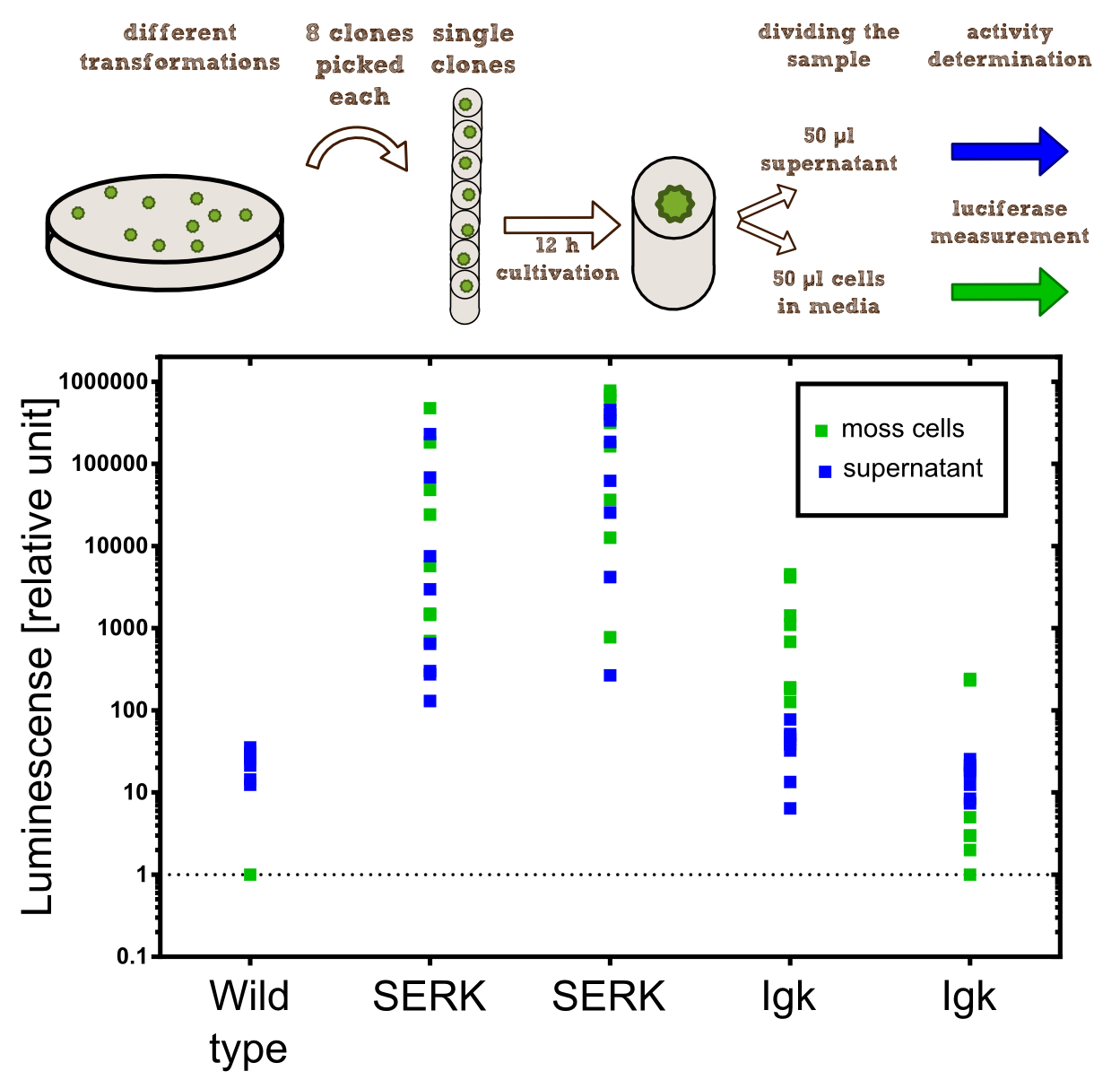
In the third step both approaches have been centrifuged at 13200 rpm for two minutes to discard the beads. Each supernatant was transferred to a separate 2 ml tube, so we could measure if the beads have bound the antibody-nanoluciferase-streptag and nanoluciferase-streptag construct by comparing the supernatants with the samples containing the beads. The beads have been washed three times with 2 ml of PBS-Buffer. After measuring the Luciferase activity in each of the four samples in a 96 well plate with 20 µl of each sample mixed with 80 µl of Luciferase-Substrate-Buffer using a plate reader, we clamped that the Luciferase activity in the supernatant was at its limit, whereas every bead-sample showed no significant Luciferase activity.
Fourth step
The conclusion of step three was that the beads have not been able to bind the cut-off construct, but, because of the significant Luciferase activity in both supernatants, it stands to reason that the membrane construct was expressed by the moss in a significant way. To get to know more about the cut-off construct we started concentrating cut-off construct in the supernatants to a volume of 150 µl.
Fifth step
After the cut-off construct in the supernatants was concentrated, we performed a SDS-Page with three samples to get to know how big the cut-off construct was. Each sample was prepared by mixing 15 µl supernatant with 7 µl of Sodiumdodecylsulfat and cooked at 95°C. Afterwards, the samples have been applied to a standard SDS-Gel and unseamed. The result of the SDS-Page proves the existence of the cut-off Nanoluciferase-Streptag-Construct at about 20 kDa. The second band at about 150 kDa refers to the used Antibody in step 1.
Sixth step
A standard Western Blot was performed, using two different Antibodies to label the cut-off Luciferase-Streptag-construct. First we labeled the cut-off construct with Strepmab Classic. Afterwards we labeled the Strepmab Classic with an Antibody fused to the enzyme Alkaline Phosphatase to bind the Strepmab Classic Antibody. By starting the reaction process of the Alkaline Phosphatase using NBP and BCIP as substrate and AP-Buffer, we tried to make the cut-off construct visible on the PVDF membrane. On the Western Blot was no significant band seen.
Conclusion
Investigation of different Signal peptides for secretion of effector proteins
Characterization of intracellular localization of recombinant proteins
Characterization of Erythromycine B esterase in the cytoplasm of recombinant moss
In order to check the activity of the erythromycine esterase B we pursued two different approaches: on the one hand we produced the enzyme recombinant in Escherichia coli and purified it. The purified protein was used for in vitro activity tests determining the depletion rates. The reaction was stopped at predetermined times by addition of three volumes acetonitrile and freezing in liquid nitrogen. On the other hand we added the substrate erythromycin at a concentration of 0.2 g L-1 to 3 mL of a polyclonal suspension of transgenic moss plants expressing the erythromycin esterase B in the cytoplasm. After 24 h of incubation at normal growth conditions 100 µL of the supernatant were used for further analysis.
Both samples were analyzed by liquid chromatography coupled to mass spectrometry (UPLC/MS/MS). The samples were measured at a Waters Xevo TQ-S using a Waters Acquity BEH C18 2,1 x 100mm column (1.7 µm, 130 Å pore size) and the following method: Solvent A: 0.1 % formic acid in water, B: 0.1 % formic acid in ACN and a linear gradient of 99 % A 0.5 min isocratic to 99 % B in 6 min. Column temperature 40 °C, flux 0.4 mL min-1, maximal pressure 703 bar. The tuning was executed with solutions consisting of the pure educt at concentrations of approximately 0.1 mgL-1.
In part A of figure seven the liquid chromatography chromatogram and the associated molecular mass for pure erythromycin as a standard is shown highlighted in red at a retention time of 3.65 min and a mass of ~734.3 g mol-1.
Part B of figure seven shows the corresponding LCMS spectra of purified erythromycin esterase B incubated for 0 min and 5 min with erythromycin. After 0 min of incubation the red highlighted peak with a retention time of 3.68 min and a mass of ~734.3 g mol-1 shows the initial presence of the substrate. However even merely mixing the substrate and enzyme was sufficient to degrade a small amount of substrate which is shown by the emerging peaks at 4.19 and 4.30 min retention time. The chromatogram of the sample taken after five minutes of incubation no longer shows the characteristic substrate peak at 3.68 min indicating that the whole amount of substrate was successfully degraded by the purified enzyme.
Part C of figure seven shows the corresponding LCMS spectra of the in vivo degradation experiment with transgenic moss expressing erythromycin esterase B in the cytoplasm. After 24 h of incubation at normal growth conditions the chromatogram of the sample containing the wild type moss as a negative control exclusively shows the characteristic peak for the substrate highlighted in red. The chromatogram of the sample in which the transgenic moss was utilized in contrast shows the clear degradation of the substrate because only the peaks at retention times of 4.19 and 4.30 min appear representing the degradation product as explained for part B of this figure.
To summarize this experiment demonstrates that our transgenic moss successfully degraded the environmental pollutant erythromycin!
References:
http://www.ncbi.nlm.nih.gov/pubmed/6327079 Edens et al., 1984
- http://www.ncbi.nlm.nih.gov/pubmed/6327079 Edens et al., 1984 Edens, L., Bom, I., Ledeboer, A. M., Maat, J., Toonen, M. Y., Visser, C., and Verrips, C. T. (1984). Synthesis and processing of the plant protein thaumatin in yeast. Cell, 37(2):629–33.
- http://www.plant-biotech.net/paper/CurrGenet_2003_hohe.pdf Hohe, A., T. Egener, J. Lucht, H. Holtorf, C. Reinhard, G. Schween, R. Reski (2004): An improved and highly standardised transformation procedure allows efficient production of single and multiple targeted gene knockouts in a moss, Physcomitrella patens. Current Genet. 44, 339-347.
 "
"













































AutoAnnotator:
Follow us:
Address:
iGEM Team TU-Munich
Emil-Erlenmeyer-Forum 5
85354 Freising, Germany
Email: igem@wzw.tum.de
Phone: +49 8161 71-4351