Team:Paris Saclay/Notebook/July/30
From 2013.igem.org
(→5 - Gel purification of digestion of BBa_I732017 by EcoRI/PstI) |
|||
| (37 intermediate revisions not shown) | |||
| Line 7: | Line 7: | ||
==='''A - Aerobic/Anaerobic regulation system'''=== | ==='''A - Aerobic/Anaerobic regulation system'''=== | ||
| - | ===='''Objective : obtaining | + | ===='''Objective : obtaining BBa_K1155004, BBa_K1155005, BBa_K1155006'''==== |
| - | ===='''1 - | + | ===='''1 - Inactivation of EcoRI/SpeI used of the digestion of PCR products : NarK, NarG, NirB '''==== |
Xavier | Xavier | ||
| Line 17: | Line 17: | ||
We used 30µL of DNA. | We used 30µL of DNA. | ||
| - | ===='''Objective : obtaining | + | ===='''Objective : obtaining BBa_K1155003, BBa_K1155007'''==== |
| - | ===='''1 - Tranformation of | + | ===='''1 - Tranformation of BBa_K1155003 in DH5α '''==== |
Abdou | Abdou | ||
| Line 25: | Line 25: | ||
Protocol : [[Team:Paris_Saclay/Protocols/Bacterial transformation|Bacterial transformation]] | Protocol : [[Team:Paris_Saclay/Protocols/Bacterial transformation|Bacterial transformation]] | ||
| + | ===='''2 - Digestion of BBa_I732017 by EcoRI/PstI'''==== | ||
| + | |||
| + | Zhou | ||
| + | |||
| + | Used quantities : | ||
| + | * BBa_I732017 : 10µL | ||
| + | * Buffer FD : 3µL | ||
| + | * PstI FD : 1.5µL | ||
| + | * EcoRI FD : 1.5µL | ||
| + | * H2O : 14µL | ||
| + | |||
| + | We let the digestion 1h30 at 37°C. | ||
| + | |||
| + | ===='''3 - Digestion of BBa_I732017 by EcoRI/SpeI'''==== | ||
| + | |||
| + | Abdou | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DE;" | | ||
| + | We used wrong enzymes so we will do the digestion again. | ||
| + | |} | ||
| + | |||
| + | Used quantities : | ||
| + | * BBa_I732017 : 10µL | ||
| + | * Buffer FD : 2µL | ||
| + | * SpeI FD : 1µL | ||
| + | * EcoRI FD : 1µL | ||
| + | * H2O : 6µL | ||
| + | |||
| + | We let the digestion 1h30 at 37°C. | ||
| + | |||
| + | ===='''4 - Electrophoresis to check the digestion of BBa_I732017 by EcoRI/SpeI'''==== | ||
| + | |||
| + | Zhou | ||
| + | |||
| + | {| | ||
| + | | style="width:350px;border:1px solid black;" |[[File:Psgel23007.jpg]] | ||
| + | | style="width:350px;border:1px solid black;vertical-align:top;" | | ||
| + | * Well 1 : 10µL of DNA Ladder | ||
| + | * Well 2 : 10µL of BBa_I732017 digested by EcoRI/SpeI+4µL of 6X loading dye | ||
| + | * Well 3 : 10µL of BBa_I732017 digested by EcoRI/SpeI+4µL of 6X loading dye | ||
| + | * Gel : 1% | ||
| + | |} | ||
| + | |||
| + | Expected size : | ||
| + | * RBS-LacZ : 3090bp | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DEDEDE;" | | ||
| + | We obtain fragments at the right size. We will purify it. | ||
| + | |} | ||
| + | |||
| + | ===='''5 - Gel purification of digestion of BBa_I732017 by EcoRI/SpeI'''==== | ||
| + | |||
| + | Xavier | ||
| + | |||
| + | Protocol : [[Team:Paris_Saclay/Protocols/Gel purification|Gel purification]] | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DEDEDE;" | | ||
| + | We lost our fragments. We will do the digestion again. | ||
| + | |} | ||
==='''A - Aerobic/Anaerobic regulation system / B - PCB sensor system'''=== | ==='''A - Aerobic/Anaerobic regulation system / B - PCB sensor system'''=== | ||
| - | ===='''Objective : obtaining FNR and BphR2 proteins | + | ===='''Objective : obtaining FNR and BphR2 proteins'''==== |
===='''1 - PCR of : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I '''==== | ===='''1 - PCR of : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I '''==== | ||
| Line 37: | Line 99: | ||
* Bphr2 Part I : | * Bphr2 Part I : | ||
| - | ** Oligo 54F : | + | ** Oligo 54F : 4µL |
| - | ** Oligo 55R : | + | ** Oligo 55R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA of ''Pseudomonas pseudoalcaligenes'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* Bphr2 Part II : | * Bphr2 Part II : | ||
| - | ** Oligo | + | ** Oligo 56F : 4µL |
| - | ** Oligo | + | ** Oligo 57R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA ''Pseudomonas pseudoalcaligenes'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* RBS-Bphr2 Part I : | * RBS-Bphr2 Part I : | ||
| - | ** Oligo | + | ** Oligo 58F : 4µL |
| - | ** Oligo | + | ** Oligo 57R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA ''Pseudomonas pseudoalcaligenes'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* FNR Part I : | * FNR Part I : | ||
| - | ** Oligo | + | ** Oligo 59F : 4µL |
| - | ** Oligo | + | ** Oligo 60R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA ''Escherichia coli'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* FNR Part II : | * FNR Part II : | ||
| - | ** Oligo | + | ** Oligo 61F : 4µL |
| - | ** Oligo | + | ** Oligo 62R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA ''Escherichia coli'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* RBS-FNR Part I : | * RBS-FNR Part I : | ||
| - | ** Oligo | + | ** Oligo 63F : 4µL |
| - | ** Oligo | + | ** Oligo 62R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA ''Escherichia coli'' : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
* PSB1C3 : | * PSB1C3 : | ||
| - | ** Prefixe | + | ** Prefixe 51 : 4µL |
| - | ** Suffixe | + | ** Suffixe 52 : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
PCR Program : | PCR Program : | ||
| Line 108: | Line 170: | ||
[[File:PsPCRFNR3007.jpg|400px]] | [[File:PsPCRFNR3007.jpg|400px]] | ||
| - | |||
| - | |||
| - | |||
| - | |||
===='''2 - Electrophoresis of the PCR products : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I '''==== | ===='''2 - Electrophoresis of the PCR products : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I '''==== | ||
{| | {| | ||
| - | | style="width:350px;border:1px solid black;" |File: | + | | style="width:350px;border:1px solid black;" |[[File:Psgel13007.jpg]] |
| style="width:350px;border:1px solid black;vertical-align:top;" | | | style="width:350px;border:1px solid black;vertical-align:top;" | | ||
* Well 1 : 6µL of DNA Ladder | * Well 1 : 6µL of DNA Ladder | ||
| Line 122: | Line 180: | ||
* Well 3 : 5µL of PSB1C3+1µl of 6X loading dye | * Well 3 : 5µL of PSB1C3+1µl of 6X loading dye | ||
* Well 4 : 5µL of BphR2 Part I+1µl of 6X loading dye | * Well 4 : 5µL of BphR2 Part I+1µl of 6X loading dye | ||
| - | * Well 5 : | + | * Well 5 : 5µL of BphR2 Part II+1µl of 6X loading dye |
* Well 6 : 5µL of RBS-BphR2 Part I+1µl of 6X loading dye | * Well 6 : 5µL of RBS-BphR2 Part I+1µl of 6X loading dye | ||
* Well 7 : 5µL of FNR Part I+1µl of 6X loading dye | * Well 7 : 5µL of FNR Part I+1µl of 6X loading dye | ||
| Line 137: | Line 195: | ||
* FNR Part II : 200 kb | * FNR Part II : 200 kb | ||
* RBS-FNR Part I : 615 kb | * RBS-FNR Part I : 615 kb | ||
| - | * PSB1C3 : | + | * PSB1C3 : 2070bp |
{| | {| | ||
| Line 150: | Line 208: | ||
Used quantities : | Used quantities : | ||
| - | ** Oligo 54F : | + | ** Oligo 54F : 4µL |
| - | ** Oligo 55R : | + | ** Oligo 55R : 4µL |
| - | ** Buffer | + | ** Buffer Dream Taq : 40µL |
| - | ** DNA : | + | ** DNA : 2µL |
| - | ** dNTP : | + | ** dNTP : 4µL |
| - | ** | + | ** Dream Taq : 1µL |
| - | ** H2O : | + | ** H2O : 146µL |
PCR Program : | PCR Program : | ||
| Line 164: | Line 222: | ||
===='''4 - Gel purification of PCR of : RBS-BphR2 Part I'''==== | ===='''4 - Gel purification of PCR of : RBS-BphR2 Part I'''==== | ||
| - | + | Xavier | |
| + | |||
| + | Protocol : [[Team:Paris_Saclay/Protocols/Gel purification|Gel purification]] | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DEDEDE;" | | ||
| + | We lost our fragments. We will do the PCR again. | ||
| + | |} | ||
| + | |||
| Line 174: | Line 240: | ||
Abdou, Xavier | Abdou, Xavier | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DE;" | | ||
| + | The transformation was good. We will make a Colony PCR. | ||
| + | |} | ||
| + | |||
| + | We used 25 different colonies. | ||
Used quantities : | Used quantities : | ||
| + | * Oligo 43F : 0.5µL | ||
| + | * Oligo 44R : 0.5µL | ||
| + | * Buffer Dream Taq : 35µL | ||
| + | * DNA : 2µL | ||
| + | * dNTP : 0.5µL | ||
| + | * Dream Taq : 0.25µL | ||
| + | * H2O : 25µL | ||
| + | ===='''2 - Electrophoresis of the Colony PCR of Bba_K1155002 in DH5α'''==== | ||
| + | Abdou, Xavier | ||
| + | |||
| + | {| | ||
| + | | style="width:350px;border:1px solid black;" |[[File:Psgel3007.jpg]] | ||
| + | | style="width:350px;border:1px solid black;vertical-align:top;" | | ||
| + | * Well 1 : 6µL of DNA Ladder | ||
| + | * Well 2 to 26 : 5µL of Bba_K1155002+1µl of 6X loading dye | ||
| + | * Gel : 0.8% | ||
| + | |} | ||
| + | |||
| + | Expected sizes : | ||
| + | * Bba_K1155002 : 169bp | ||
| + | |||
| + | {| | ||
| + | | style="border:1px solid black;padding:5px;background-color:#DEDEDE;" | | ||
| + | We obtain fragments at the right size. We will sequence it. | ||
| + | |} | ||
| + | |||
| + | |||
| + | {| border="1" align="center" | ||
| + | |[[Team:Paris Saclay/Notebook/July/26|<big>Previous day</big>]] | ||
| + | |||
| + | |[[Team:Paris_Saclay/Notebook|<big>Back to calendar</big>]] | ||
| + | |||
| + | |[[Team:Paris Saclay/Notebook/July/31|<big>Next day</big>]] | ||
| + | |} | ||
{{Team:Paris_Saclay/incl_fin}} | {{Team:Paris_Saclay/incl_fin}} | ||
Latest revision as of 00:58, 5 October 2013
Notebook : July 30
Lab work
A - Aerobic/Anaerobic regulation system
Objective : obtaining BBa_K1155004, BBa_K1155005, BBa_K1155006
1 - Inactivation of EcoRI/SpeI used of the digestion of PCR products : NarK, NarG, NirB
Xavier
Protocol : Ethanol precipitation
We used 30µL of DNA.
Objective : obtaining BBa_K1155003, BBa_K1155007
1 - Tranformation of BBa_K1155003 in DH5α
Abdou
Protocol : Bacterial transformation
2 - Digestion of BBa_I732017 by EcoRI/PstI
Zhou
Used quantities :
- BBa_I732017 : 10µL
- Buffer FD : 3µL
- PstI FD : 1.5µL
- EcoRI FD : 1.5µL
- H2O : 14µL
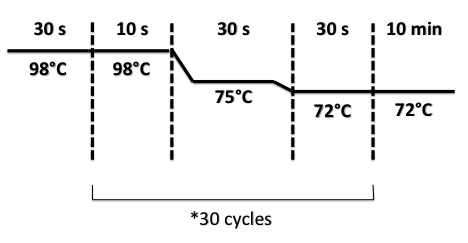
We let the digestion 1h30 at 37°C.
3 - Digestion of BBa_I732017 by EcoRI/SpeI
Abdou
|
We used wrong enzymes so we will do the digestion again. |
Used quantities :
- BBa_I732017 : 10µL
- Buffer FD : 2µL
- SpeI FD : 1µL
- EcoRI FD : 1µL
- H2O : 6µL
We let the digestion 1h30 at 37°C.
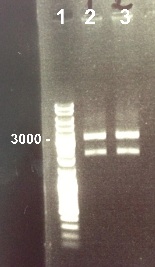
4 - Electrophoresis to check the digestion of BBa_I732017 by EcoRI/SpeI
Zhou
|
|
Expected size :
- RBS-LacZ : 3090bp
|
We obtain fragments at the right size. We will purify it. |
5 - Gel purification of digestion of BBa_I732017 by EcoRI/SpeI
Xavier
Protocol : Gel purification
|
We lost our fragments. We will do the digestion again. |
A - Aerobic/Anaerobic regulation system / B - PCB sensor system
Objective : obtaining FNR and BphR2 proteins
1 - PCR of : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I
Abdou, Xavier
Used quantities :
- Bphr2 Part I :
- Oligo 54F : 4µL
- Oligo 55R : 4µL
- Buffer Dream Taq : 40µL
- DNA of Pseudomonas pseudoalcaligenes : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
- Bphr2 Part II :
- Oligo 56F : 4µL
- Oligo 57R : 4µL
- Buffer Dream Taq : 40µL
- DNA Pseudomonas pseudoalcaligenes : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
- RBS-Bphr2 Part I :
- Oligo 58F : 4µL
- Oligo 57R : 4µL
- Buffer Dream Taq : 40µL
- DNA Pseudomonas pseudoalcaligenes : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
- FNR Part I :
- Oligo 59F : 4µL
- Oligo 60R : 4µL
- Buffer Dream Taq : 40µL
- DNA Escherichia coli : 2µL
- dNTP : 4µL
- Dream taq : 1µL
- H2O : 146µL
- FNR Part II :
- Oligo 61F : 4µL
- Oligo 62R : 4µL
- Buffer Dream Taq : 40µL
- DNA Escherichia coli : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
- RBS-FNR Part I :
- Oligo 63F : 4µL
- Oligo 62R : 4µL
- Buffer Dream Taq : 40µL
- DNA Escherichia coli : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
- PSB1C3 :
- Prefixe 51 : 4µL
- Suffixe 52 : 4µL
- Buffer Dream Taq : 40µL
- DNA : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
PCR Program :
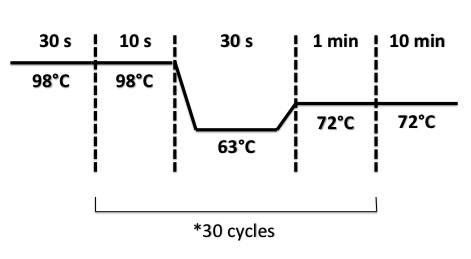
- BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I :
- FNR Part I, FNR Part II, RBS-FNR Part I :
2 - Electrophoresis of the PCR products : BphR2 Part I, BphR2 Part II, RBS-BphR2 Part I, FNR Part I, FNR Part II, RBS-FNR Part I
Expected size
- BphR2 Part I : 178 kb
- BphR2 Part II : 790 kb
- RBS-BphR2 Part I : 197 kb
- FNR Part I : 597 kb
- FNR Part II : 200 kb
- RBS-FNR Part I : 615 kb
- PSB1C3 : 2070bp
|
We can't see RBS-BphR2 Part I fragments at the good size. We will make the PCR again with a new PCR Program. We obtain BphR2 Part I, BphR2 Part II, FNR Part I, Fnr Part II and RBS-FNR Part I frangments at the right size thanks to the PCR. We will purify it. |
3 - PCR of : RBS-BphR2 Part I
Abdou, Xavier
Used quantities :
- Oligo 54F : 4µL
- Oligo 55R : 4µL
- Buffer Dream Taq : 40µL
- DNA : 2µL
- dNTP : 4µL
- Dream Taq : 1µL
- H2O : 146µL
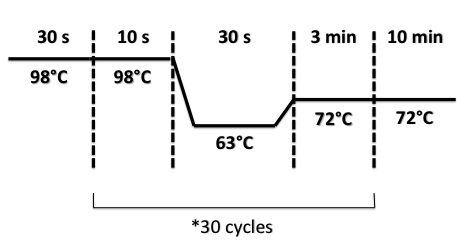
PCR Program :
4 - Gel purification of PCR of : RBS-BphR2 Part I
Xavier
Protocol : Gel purification
|
We lost our fragments. We will do the PCR again. |
B - PCB sensor system
Objective : obtaining Bba_K1155002
1 - Colony PCR of Bba_K1155002 in DH5α
Abdou, Xavier
|
The transformation was good. We will make a Colony PCR. |
We used 25 different colonies.
Used quantities :
- Oligo 43F : 0.5µL
- Oligo 44R : 0.5µL
- Buffer Dream Taq : 35µL
- DNA : 2µL
- dNTP : 0.5µL
- Dream Taq : 0.25µL
- H2O : 25µL
2 - Electrophoresis of the Colony PCR of Bba_K1155002 in DH5α
Abdou, Xavier
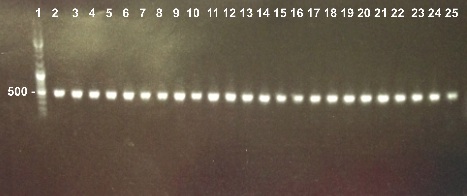
|
|
Expected sizes :
- Bba_K1155002 : 169bp
|
We obtain fragments at the right size. We will sequence it. |
| Previous day | Back to calendar | Next day |
 "
"