Team:TU-Delft/SUMOPeptideProduction
From 2013.igem.org

The era of antibiotics are now outdated as the microorganisms started gaining new resistance mechanism to evade the effects of antibiotics. Nowadays more and more researches are focused towards new antimicrobial that can be more effective against these pathogens.
The antimicrobial peptides (AMP's), natural or de-novo are becoming more popular due to their potential to kill or restrict the growth of microorganisms. They form a part of chemical defense against predators. All organisms, including higher order eukaryotes have some antimicrobial peptides that acts against specific strains of microorganisms.They are usually charged and interact with the opposite charges present on the membrane thus boring a hole through the membrane subsequently lysing the cell.Unlike, antibiotics they do not interfere in the genetic, transcriptional or translational machinery of the cells.
We chose 3 AMP's that are mostly produced in the skins of toads and frogs. They are magainin from Xenopus laevis , signiferin from Crinia signifera and maximin-H5 from Bombina maxima. The reasons for choosing these AMP's could be attributed to their target specific nature, charge and length of peptides. They do not affect humans, which makes them a suitable candidate as pharmaceuticals.
The recombinant production of these peptides in E. coli would help large scale production of these peptides and administer them as antimicrobials. But, the major hurdle in producing them in vivo is the formation of inclusion bodies as they are charged. This could be overcome by making protein fusion with SUMO (Small Ubiquitin like Modifier), MBP (Maltose Binding Protein), Trx (Thioredoxin) etc.
In this project, a SUMO-peptide fusion was opted as a suitable expression system where the enzyme Ulp cleaving the fusion is also expressed and controlled by a negative transcriptional cascade. This fusion increases the solubility of the peptide and reduces inclusion bodies formation(Figure 1).
The in-vivo production of peptides at sufficient quantities is crucial to kill the MRSA. To prove that we produce enough peptides we carried out the following characterization experiments.
Experiments: SUMO-Peptide Production
Aim: Increasing solubility of peptide with Sumo Fusion.
Description
The peptide by itself is not soluble in the cytoplasm but making a fusion of peptide with Small Ubiquitin like Modifiers (SUMO) will increase the solubility of the peptide, thus increasing the cytoplasmic fraction of the peptide.
A gene was constructed in such a way that the SUMO-peptide production was driven by the strong T7 phage promoter. This gene containing plasmid was harboured in a BL21(DE3) strain that has lac promoter driven T7 polymerase. Upon induction by IPTG the SUMO peptide fusion is produced as a soluble protein fraction.
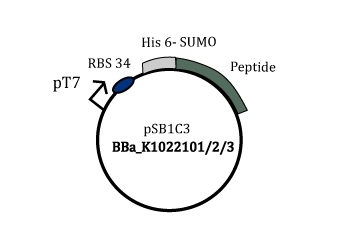
Figure 1: Part BBa_K1022101/2/3
 "
"

